Abstract
Purposes
The lymph node ratio (LNR) has been considered a better prognostic factor than traditional N staging in patients with gastric cancer (GC), but its accuracy is unclear for those who receive neoadjuvant therapy (NAT). We aimed to compare the node ratio (Nr) staging with the ypN staging and to thereby develop a modified staging system incorporating Nr staging.
Methods
A total of 1791 patients who underwent gastrectomy after NAT in the Surveillance, Epidemiology, and End Results database were retrospectively analyzed. ypTNrM staging was established based on the overall survival (OS).
Results
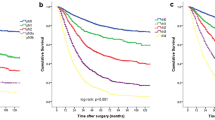
The Nr staging was generated using 0.2 and 0.5 as the cutoff values of LNR and represented patients with more homogeneous OS compared with ypN staging. The 5-year OS rates for ypTNrM stages IA, IB, II, IIIA, and IIIB were 70.2%, 54.2%, 36.0%, 21.2%, and 6.6%, respectively, compared with 58.8%, 39.1%, and 21.6% for ypTNM stages I, II, and III, respectively. Compared with the ypTNM staging system, the ypTNrM staging system had a lower misclassification rate (3.0% vs. 50.9%) and better prognostic predictive power (C-index: 0.645 vs. 0.589, P < 0.001).
Conclusions
The ypTNrM staging system incorporating Nr staging may provide a more accurate assessment in the clinical decision-making process for GC after NAT.




Similar content being viewed by others
References
Bray F, Ferlay J, Soerjomataram I, Siegel RL, Torre LA, Jemal A. Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. Cancer J Clin. 2018;68:394–424.
Pyo JH, Lee H, Min BH, Lee JH, Choi MG, Lee JH, et al. Long-term outcome of endoscopic resection vs surgery for early gastric cancer: a non-inferiority-matched cohort study. Am J Gastroenterol. 2016;111:240–9.
Songun I, Putter H, Kranenbarg EM, Sasako M, van de Velde CJ. Surgical treatment of gastric cancer: 15-year follow-up results of the randomised nationwide Dutch D1D2 trial. Lancet Oncol. 2010;11:439–49.
Ajani JA, Lee J, Sano T, Janjigian YY, Fan D, Song S. Gastric adenocarcinoma. Nat Rev Dis Primers. 2017;3:17036.
Ajani JA, D'Amico TA, Bentrem DJ, Chao J, Corvera C, Das P, et al. Gastric cancer, version 2.2020, NCCN clinical practice guidelines in oncology. J Natl Compr Cancer Netw Jnccn. 2020:MS-11–3.
Cunningham D, Allum WH, Stenning SP, Thompson JN, Van de Velde CJ, Nicolson M, et al. Perioperative chemotherapy versus surgery alone for resectable gastroesophageal cancer. N Engl J Med. 2006;355:11–20.
Ychou M, Boige V, Pignon JP, Conroy T, Bouché O, Lebreton G, et al. Perioperative chemotherapy compared with surgery alone for resectable gastroesophageal adenocarcinoma: an FNCLCC and FFCD multicenter phase III trial. J Clin Oncol. 2011;29:1715–21.
Al-Batran SE, Homann N, Pauligk C, Goetze TO, Meiler J, Kasper S, et al. Perioperative chemotherapy with fluorouracil plus leucovorin, oxaliplatin, and docetaxel versus fluorouracil or capecitabine plus cisplatin and epirubicin for locally advanced, resectable gastric or gastro-oesophageal junction adenocarcinoma (FLOT4): a randomised, phase 2/3 trial. Lancet. 2019;393:1948–57.
Kakar S, Pawlik T, Allen P. AJCC cancer staging manual. 8th ed. New York: Springer-Verlag; 2017.
Li Z, Wang Y, Shan F, Ying X, Wu Z, Xue K, et al. ypTNM staging after neoadjuvant chemotherapy in the Chinese gastric cancer population: an evaluation on the prognostic value of the AJCC eighth edition cancer staging system. Gastric Cancer. 2018;21:977–87.
Lin JX, Yoon C, Desiderio J, Yi BC, Li P, Zheng CH, et al. Development and validation of a staging system for gastric adenocarcinoma after neoadjuvant chemotherapy and gastrectomy with D2 lymphadenectomy. Br J Surg. 2019;106:1187–96.
Zhong Q, Chen QY, Parisi A, Ma YB, Lin GT, Desiderio J, et al. Modified ypTNM staging classification for gastric cancer after neoadjuvant therapy: a multi-institutional study. Oncologist. 2021;26:e99–110.
In H, Ravetch E, Langdon-Embry M, Palis B, Ajani JA, Hofstetter WL, et al. The newly proposed clinical and post-neoadjuvant treatment staging classifications for gastric adenocarcinoma for the American Joint Committee on Cancer (AJCC) staging. Gastric Cancer. 2018;21:1–9.
Inoue K, Nakane Y, Iiyama H, Sato M, Kanbara T, Nakai K, et al. The superiority of ratio-based lymph node staging in gastric carcinoma. Ann Surg Oncol. 2002;9:27–34.
Marchet A, Mocellin S, Ambrosi A, Morgagni P, Garcea D, Marrelli D, et al. The ratio between metastatic and examined lymph nodes (N ratio) is an independent prognostic factor in gastric cancer regardless of the type of lymphadenectomy: results from an Italian multicentric study in 1853 patients. Ann Surg. 2007;245:543–52.
Xu DZ, Geng QR, Long ZJ, Zhan YQ, Li W, Zhou ZW, et al. Positive lymph node ratio is an independent prognostic factor in gastric cancer after D2 resection regardless of the examined number of lymph nodes. Ann Surg Oncol. 2009;16:319–26.
Wang J, Dang P, Raut CP, Pandalai PK, Maduekwe UN, Rattner DW, et al. Comparison of a lymph node ratio-based staging system with the 7th AJCC system for gastric cancer: analysis of 18,043 patients from the SEER database. Ann Surg. 2012;255:478.
Kong SH, Lee HJ, Ahn HS, Kim JW, Kim WH, Lee KU, et al. Stage migration effect on survival in gastric cancer surgery with extended lymphadenectomy: the reappraisal of positive lymph node ratio as a proper N-staging. Ann Surg. 2012;255:50–8.
Yamashita K, Hosoda K, Ema A, Watanabe M. Lymph node ratio as a novel and simple prognostic factor in advanced gastric cancer. Eur J Surg Oncol. 2016;42:1253–60.
Surveillance E, and End Results (SEER) Program (www.seer.cancer.gov) SEER*Stat Database. Incidence-SEER 18 Regs Research Data + Hurricane Katrina Impacted Louisiana Cases (with additional treatment fields), Nov 2018 Sub(1975–2016 varying), National Cancer Institute, DCCPS, Surveillance Research Program, released April 2019, based on the November 2018 submission.
Chen QY, Zhong Q, Wang W, Chen S, Li P, Xie JW, et al. Prognosis of young survivors of gastric cancer in China and the U.S.: determining long-term outcomes based on conditional survival. Oncologist. 2018. https://doi.org/10.1634/theoncologist.2018-0220.
Hu C, Xing Y, Cormier JN, Chang GJ. The validity of cancer specific mortality within the surveillence, epidemiology, and end results registry. J Surg Res. 2011;165:270.
Sun Z, Zhu GL, Lu C, Guo PT, Huang BJ, Li K, et al. A novel subclassification of pT2 gastric cancers according to the depth of muscularis propria invasion: superficial muscularis propria versus deep muscularis propria/subserosa. Ann Surg. 2009;249:768–75.
Zhao L, Claggett B, Tian L, Uno H, Pfeffer MA, Solomon SD, et al. On the restricted mean survival time curve in survival analysis. Biometrics. 2016;72:215–21.
Ben-Aharon O, Magnezi R, Leshno M, Goldstein DA. Median survival or mean survival: Which measure is the most appropriate for patients, physicians, and policymakers? Oncologist. 2019;24:1469–78.
Harrell FE, Califf RM, Pryor DB, Lee KL, Rosati RA. Evaluating the yield of medical tests. JAMA. 1982;247:2543–6.
Harrell FE, Lee KL, Mark DB. Multivariable prognostic models: issues in developing models, evaluating assumptions and adequacy, and measuring and reducing errors. Stat Med. 1996;15:361–87.
Awad AM. Properties of the Akaike information criterion. Microelectron Reliab. 1996;36:457–64.
Heagerty PJ, Lumley T, Pepe MS. Biometrics. Time-dependent ROC curves for censored survival data and a diagnostic marker. Biometrics. 2000;56:337–44.
Lemeshow S, Hosmer DW Jr. A review of goodness of fit statistics for use in the development of logistic regression models. Am J Epidemiol. 1982;115:92–106.
Vickers AJ, Elkin EB. Decision curve analysis: a novel method for evaluating prediction models. Med Decis Making. 2006;26:565.
Siegel RL, Miller KD, Jemal A. Cancer statistics, 2018. Cancer J Clin. 2018;68:7–30.
Tey J, Lu JJ. Gastric cancer. In: Lee NY, Riaz N, Lu JJ, editors. Target volume delineation for conformal and intensity-modulated radiation therapy. Medical radiology. New York: Springer; 2014. p. 261–73.
Stahl M, Walz MK, Stuschke M, Lehmann N, Meyer HJ, Riera-Knorrenschild J, et al. Phase III comparison of preoperative chemotherapy compared with chemoradiotherapy in patients with locally advanced adenocarcinoma of the esophagogastric junction. J Clin Oncol. 2009;27:851–6.
van Hagen P, Hulshof MC, van Lanschot JJ, Steyerberg EW, van Berge Henegouwen MI, Wijnhoven BP, et al. Preoperative chemoradiotherapy for esophageal or junctional cancer. N Engl J Med. 2012;366:2074–84.
Cats A, Jansen EPM, van Grieken NCT, Sikorska K, Lind P, Nordsmark M, et al. Chemotherapy versus chemoradiotherapy after surgery and preoperative chemotherapy for resectable gastric cancer (CRITICS): an international, open-label, randomised phase 3 trial. Lancet Oncol. 2018;19:616–28.
Okines A, Verheij M, Allum W, Cunningham D, Cervantes A. Gastric cancer: ESMO Clinical Practice Guidelines for diagnosis, treatment and follow-up. Ann Oncol. 2010;21(5):50.
Ajani JA, D'Amico TA, Bentrem DJ, Chao J, Collier S, Corvera C, et al. Gastric Cancer, Version 4.2020, NCCN Clinical Practice Guidelines in Oncology. J Natl Compr Cancer Netw Jnccn. 2020:MS-11–3.
Zhou Y, Zhang J, Cao S, Li Y. The evaluation of metastatic lymph node ratio staging system in gastric cancer. Gastric Cancer. 2013;16:309–17.
Association JGC. Japanese gastric cancer treatment guidelines 2014 (ver. 4). Gastric Cancer. 2017;20:1–19.
Ajani JA, D'Amico TA, Bentrem DJ, Chao J, Corvera C, Das P, et al. Gastric cancer, version 1.2019, NCCN clinical practice guidelines in oncology. J Natl Compr Cancer Netw Jnccn. 2019:MS-11–2.
Becker K, Reim D, Novotny A, zum Büschenfelde CM, Engel J, Friess H, et al. Proposal for a multifactorial prognostic score that accurately classifies 3 groups of gastric carcinoma patients with different outcomes after neoadjuvant chemotherapy and surgery. Ann Surg. 2012;256:1002.
Smyth EC, Fassan M, Cunningham D, Allum WH, Okines AF, Lampis A, et al. Effect of pathologic tumor response and nodal status on survival in the medical research council adjuvant gastric infusional chemotherapy trial. J Clin Oncol. 2016;34:2721–7.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
There are no conflicts of interest or financial ties to disclose from any authors.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Chen, JX., Sun, JW., Wang, Y. et al. Lymph node ratio-based the ypTNrM staging system for gastric cancer after neoadjuvant therapy: a large population-based study. Surg Today 52, 783–794 (2022). https://doi.org/10.1007/s00595-021-02386-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00595-021-02386-3