Abstract
Purposes
Numerous indicators have been discussed as predictive markers for the incidence of chronic allograft dysfunction (CLAD) after lung transplantation (LTX). The aim of this study was to evaluate whether or not the preoperative prognostic nutrition index (PNI) correlated with the development of CLAD.
Method
This study is a single-center and retrospective cohort study. Forty-six patients underwent cadaveric lung transplantation between 2000 and 2016 at our institution. The primary endpoint of this study was the CLAD-free survival of the patients.
Result
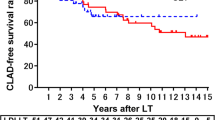
CLAD was diagnosed in 11 patients (23%) during the follow-up period. Potential risk factors included recipient factors, donor factors, number of HLA mismatches, operation-related factors, and preoperative blood test results, including the preoperative PNI. The patients with a higher PNI showed a longer CLAD-free survival after LTX than those with lower values according to univariate and multivariate analyses (p = 0.01, 0.04, respectively). The 5-year CLAD-free survival rates in the higher-PNI patients and lower-PNI patients were 94% and 62%, respectively.
Conclusion
We found that a lower preoperative PNI of the recipient was significantly associated with a higher incidence rate of CLAD. The preoperative PNI may, therefore, be useful as a predictor of the development of CLAD.



Similar content being viewed by others
References
Yusen RD, Christie JD, Edwards LB, Kucheryavaya AY, Benden C, Dipchand AI, et al. The registry of the international society for heart and lung transplantation: thirtieth adult lung and heart-lung transplant report—2013; focus theme: age. J Hear Lung Transplant. 2013;32:965–78. https://doi.org/10.1016/j.healun.2013.08.007.
Gries CJ, Bhadriraju S, Edelman JD, Goss CH, Raghu G, Mulligan MS. Obese patients with idiopathic pulmonary fibrosis have a higher 90-day mortality risk with bilateral lung transplantation. J Hear Lung Transplant. 2015;34:241–6. https://doi.org/10.1016/j.healun.2014.09.031.
Pacelli F, Bossola M, Rosa F, Tortorelli AP, Papa V, Doglietto GB. Is malnutrition still a risk factor of postoperative complications in gastric cancer surgery? Clin Nutr. 2008;27:398–407.
Nozoe T, Kimura Y, Ishida M, Saeki H, Korenaga D, Sugimachi K. Correlation of pre-operative nutritional condition with post-operative complications in surgical treatment for oesophageal carcinoma. Eur J Surg Oncol. 2002;28:396–400.
Nozoe T, Kohno M, Iguchi T, Mori E, Maeda T, Matsukuma A, et al. The prognostic nutritional index can be a prognostic indicator in colorectal carcinoma. Surg Today. 2012;42:532–5.
Kim CY, Kim SY, Song JH, Kim YS, Jeong SJ, Lee JG, et al. Usefulness of the preoperative prognostic nutritional index score as a predictor of the outcomes of lung transplantation: a single-institution experience. Clin Nutr. 2019;38:2423–9. https://doi.org/10.1016/j.clnu.2018.10.027.
Yamamoto H, Sugimoto S, Soh J, Shiotani T, Miyoshi K, Otani S, et al. The prognostic nutritional index is correlated negatively with the lung allocation score and predicts survival after both cadaveric and living-donor lobar lung transplantation. Surg Today. 2021. https://doi.org/10.1007/s00595-021-02244-2.
Verleden GM, Vos R, Vanaudenaerde B, Dupont L, Yserbyt J, Van Raemdonck D, et al. Current views on chronic rejection after lung transplantation. Transpl Int. 2015;28:1131–9.
Vos R, Verleden SE, Verleden GM. Chronic lung allograft dysfunction: evolving practice. Curr Opin Organ Transplant. 2015;20:483–91.
Hsu J, Krishnan A, Lin CT, Shah PD, Broderick SR, Higgins RSD, et al. Sarcopenia of the psoas muscles is associated with poor outcomes following lung transplantation. Ann Thorac Surg. 2019;107:1082–8. https://doi.org/10.1016/j.athoracsur.2018.10.006.
Inoue M, Minami M, Wada N, Nakagiri T, Funaki S, Kawamura T, et al. Results of surveillance bronchoscopy after cadaveric lung transplantation: a Japanese single-institution study. Transplant Proc. 2014;46:944–7. https://doi.org/10.1016/j.transproceed.2013.10.055.
Stewart S, Fishbein MC, Snell GI, Berry GJ, Boehler A, Burke MM, et al. Revision of the 1996 working formulation for the standardization of nomenclature in the diagnosis of lung rejection. J Hear Lung Transplant. 2007;26:1229–42.
Goh BKP, Kam JH, Lee SY, Chan CY, Allen JC, Jeyaraj P, et al. Significance of neutrophil-to-lymphocyte ratio, platelet-to-lymphocyte ratio and prognostic nutrition index as preoperative predictors of early mortality after liver resection for huge (≥ 10 cm) hepatocellular carcinoma. J Surg Oncol. 2016;113:621–7.
Verleden GM, Raghu G, Meyer KC, Glanville AR, Corris P. A new classification system for chronic lung allograft dysfunction. J Hear Lung Transplant. 2014;33:127–33. https://doi.org/10.1016/j.healun.2013.10.022.
Suzuki Y, Okabayashi K, Hasegawa H, Tsuruta M, Shigeta K, Kondo T, et al. Comparison of preoperative inflammation-based prognostic scores in patients with colorectal cancer. Ann Surg. 2018;267:527–31.
Chen QJ, Qu HJ, Li DZ, Li XM, Zhu JJ, Xiang Y, et al. Prognostic nutritional index predicts clinical outcome in patients with acute ST-segment elevation myocardial infarction undergoing primary percutaneous coronary intervention. Sci Rep. 2017;7:1–8.
Hori S, Ichikawa K, Morizawa Y, Gotoh D, Itami Y, Nakai Y, et al. Clinical significance of postoperative nutritional status as a prognostic factor in kidney transplant recipients. Transplant Proc. 2019;51:1763–72. https://doi.org/10.1016/j.transproceed.2019.04.047.
Lu K, Li H, Chen Y, Wu B, Zhang J, Huang M, et al. Can the preoperative nutritional risk score be a predictor of the outcomes in critically ill patients of lung transplantation: a retrospective study. Ann Transl Med. 2020;8:40–40.
Halpern AL, Boshier PR, White AM, Houk AK, Helmkamp L, Mitchell JD, et al. A comparison of frailty measures at listing to predict outcomes after lung transplantation. Ann Thorac Surg. 2020;109:233–40. https://doi.org/10.1016/j.athoracsur.2019.07.040.
Chamogeorgakis T, Mason DP, Murthy SC, Thuita L, Raymond DP, Pettersson GB, et al. Impact of nutritional state on lung transplant outcomes. J Hear Lung Transplant. 2013;32:693–700.
Kelm DJ, Bonnes SL, Jensen MD, Eiken PW, Hathcock MA, Kremers WK, et al. Pre-transplant wasting (as measured by muscle index) is a novel prognostic indicator in lung transplantation. Clin Transplant. 2016;30:247–55.
Beaudart C, Sanchez-Rodriguez D, Locquet M, Reginster JY, Lengelé L, Bruyère O. Malnutrition as a strong predictor of the onset of sarcopenia. Nutrients. 2019;11:1–13.
Yamada Y, Langner T, Inci I, Benden C, Schuurmans M, Weder W, et al. Impact of human leukocyte antigen mismatch on lung transplant outcome. Interact Cardiovasc Thorac Surg. 2018;26:859–64.
Young KA, Dilling DF. The future of lung transplantation. Chest. 2019;155:465–73. https://doi.org/10.1016/j.chest.2018.08.1036.
Levy L, Huszti E, Tikkanen J, Ghany R, Klement W, Ahmed M, et al. The impact of first untreated subclinical minimal acute rejection on risk for chronic lung allograft dysfunction or death after lung transplantation. Am J Transplant. 2020;20:241–9.
Lauzurica R, Pastor MC, Bayés B, Hernández JM, Bonet J, Doladé M, et al. Pretransplant inflammation: a risk factor for delayed graft function? J Nephrol. 2008;21:221–8.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors have no conflicts of interest to declare.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Kanou, T., Minami, M., Funaki, S. et al. Importance of the preoperative prognostic nutritional index score as a predictor of chronic lung allograft dysfunction after lung transplantation: a Japanese single-institution study. Surg Today 51, 1946–1952 (2021). https://doi.org/10.1007/s00595-021-02285-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00595-021-02285-7