Abstract
Purpose
To investigate changes in the incidence of postoperative infections in the surgical department of a teaching hospital.
Methods
During the 30-year period from September 1987 to August 2017, 11,568 gastroenterological surgical procedures were performed in our surgical department. This 30-year period was divided into seven periods (A–G), ranging from 2 to 7 years each and based on the infection control methods used in each period. We then compared the rates of incisional surgical site infection (SSI) and organ/space SSI; remote infection (RI) including respiratory tract infection (RTI), intravascular catheter-related infection, and urinary tract infection (UTI); and antibiotic-associated colitis caused by methicillin-resistant Staphylococcus aureus (MRSA) enteritis or Clostridioides (Clostridium) difficile-associated disease (CDAD) among the seven periods.
Results
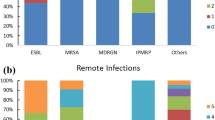
In periods B (September 1990–August 1997) and E (November 2004–July 2007), when a unique antibiotic therapy devised in our department was in use, MRSA was isolated from only 0.3% and 0.4% of surgical patients, respectively, and these rates were significantly lower than those in the other periods (p < 0.05). The rate of CDAD increased during period F (August 2007–July 2014), but in period G (August 2014–August 2017), restrictions were placed on the use of antibiotics with a strong anti-anaerobic action and, in this period, the rate of CDAD was only 0.04%, which was significantly lower than that in period F (p < 0.05).
Conclusions
Limiting the use of antibiotics that tend to disrupt the intestinal flora may reduce the rates of MRSA infection and CDAD after gastroenterological surgery.
Similar content being viewed by others
References
Kusachi S, Sumiyama Y, Nagao J, Kawai K, Arima Y, Yoshida Y, et al. New methods of control against postoperative methicillin-resistant Staphylococcus aureus infection. Surg Today. 1999;29:724–9.
Mangram AJ, Horan TC, Pearson ML, Silver LC, Jarvis WR, Centers for Disease Control and Prevention (CDC) Hospital Infection Control Practices Advisory Committee. Guideline for prevention of surgical site infection, 1999. Am J Infect Control. 1999;27:97–132.
Kiribayashi T, Kusachi S, Watanabe M, Nishimuta H, Hagiwara O, Saida Y. Countermeasures against methicillin-resistant Staphylococcus aureus transmission non-screening preemptive isolation and cohorting of patients with respiratory tract devices. Toho J Med. 2017;3:34–40.
Kusachi S, Nagao J, Saida Y, Watanabe M, Nakamura Y, Asai K, et al. Twenty years of countermeasures against postoperative methicillin-resistant Staphylococcus aureus infections. Surg Today. 2011;41:630–6.
Evans HL, Milburn ML, Hughes MG, Smith RL, Chong TW, Raymond DP, et al. Nature of Gram-negative rod antibiotic resistance during antibiotic rotation. Surg Infect (Larchmt). 2005;6:223–31.
Gruson D, Hilbert G, Vargas F, Valentino R, Bui N, Pereyre S, et al. Strategy of antibiotic rotation: long-term effect on incidence and susceptibilities of Gram-negative bacilli responsible for ventilator-associated pneumonia. Crit Care Med. 2003;31(7):1908–14.
Craig WA. Pharmacokinetic/pharmacodynamic parameters: rationale for antibacterial dosing of mice and men. Clin Infect Dis. 1998;26:1–10.
Li C, Kuti JL, Nightingale CH, Mansfield DL, Dana A, Nicolau DP. Population pharmacokinetics and pharmacodynamics of piperacillin/tazobactam in patients with complicated intra-abdominal infection. J Antimicrob Chemother. 2005;56:388–95.
Niitsuma T, Kusachi S, Takesue Y, Mikamo H, Asai K, Watanabe M. Current status of postoperative infections after digestive surgery in Japan: the Japan Postoperative Infectious Complications Survey in 2015. Ann Gastroenterol Surg. 2019;00:1–9. https://doi.org/10.1002/ags3.12236.
Furuya-Kanamori L, Stone JC, Clark J, McKenzie SJ, Yakob L, Paterson DL, et al. Comorbidities, exposure to medications, and the risk of community-acquired Clostridium difficile infection: a systematic review and meta-analysis. Infect Control Hosp Epidemiol. 2015;36:132–41.
Eze P, Balsells E, Kyaw MH, Nair H. Risk factors for Clostridium difficile infections—an overview of the evidence base and challenges in data synthesis. J Glob Health. 2017;7:010417.
Kusachi S, Sumiyama Y, Nagao J, Arima Y, Yoshida Y, Tanaka H, et al. Prophylactic antibiotics given within 24 hours of surgery, compared with antibiotics given for 72 hours perioperatively, increased the rate of methicillin-resistant Staphylococcus aureus isolated from surgical site infections. J Infect Chemother. 2008;14:44–50.
Takahata M, Sugiura Y, Ameyama S, Yotsuji A, Mitsuyama J, Sumiyama Y, et al. Influence of various antimicrobial agents on the intestinal flora in an intestinal MRSA-carrying rat model. J Infect Chemother. 2004;10:338–42.
Ansaloni L, Pisano M, Coccolini F, Peitzmann AB, Fingerhut A, Catena F, et al. WSES guidelines on acute calculous cholecystitis. World J Emerg Surg. 2016;11:25.
Davido B, Batista R, Michelon H, Lepainteur M, Bouchand F, Lepeule R, et al. Is faecal microbiota transplantation an option to eradicate highly drug-resistant enteric bacteria carriage? J Hosp Infect. 2017;95:433–7.
Slimings C, Riley TV. Antibiotics and hospital-acquired Clostridium difficile infection: update of systematic review and meta-analysis. J Antimicrob Chemother. 2014;69:881–91.
Sullivan A, Edlund C, Nord CE. Effect of antimicrobial agents on the ecological balance of human microflora. Lancet Infect Dis. 2001;1:101–14.
Owens RC Jr, Donskey CJ, Gaynes RP, Loo VG, Muto CA. Antimicrobial-associated risk factors for Clostridium difficile infection. Clin Infect Dis. 2008;15(46 Suppl 1):S19–31.
Gerding DN, Johnson S, Peterson LR, Mulligan ME, Silva J Jr. Clostridium difficile-associated diarrhea and colitis. Infect Control Hosp Epidemiol. 1995;16(8):459–77.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
Shinya Kusachi and his co-authors have no conflicts of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Kusachi, S., Watanabe, M., Asai, K. et al. Successful perioperative infection control measures after gastroenterological surgery reduced the number of cases of methicillin-resistant Staphylococcus aureus or Clostridioides (Clostridium) difficile infection to almost zero over a 30-year period: a single-department experience. Surg Today 50, 258–266 (2020). https://doi.org/10.1007/s00595-019-01899-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00595-019-01899-2