Abstract
Purposes
Laparoscopic gastrectomy using ultrasonic devices occasionally causes postoperative pancreatic fistula. Robotic gastrectomy using monopolar scissors may reduce intraoperative injury to the pancreas. We evaluated the safety and feasibility of robotic gastrectomy.
Methods
A multicenter prospective study was conducted to evaluate the surgical outcomes of robotic gastrectomy. The primary endpoints were the incidence of intraoperative and postoperative complications and operative mortality.
Results
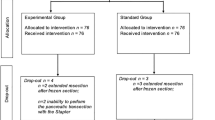
A total of 115 patients were enrolled. The clinical T stages were T1 in 68 patients and T2 or higher in 47 patients. The types of surgery included distal gastrectomy (n = 72), total gastrectomy (n = 39), and proximal gastrectomy (n = 4). Two patients developed intraoperative complications (1.7%), but no cases required conversion to open surgery. The amylase concentration in drainage fluid was higher in cases with pancreatic compression, especially in those with compression for longer than 20 min. Postoperative complications of Clavien–Dindo grade ≥ II occurred in 11 patients (9.6%). There was no mortality. A multivariate analysis indicated that a high body mass index and pancreatic compression by an assistant for longer than 20 min were independent risk factors for postoperative complications (P = 0.029 and P = 0.010).
Conclusions
Robotic gastrectomy using monopolar scissors is safe and feasible. Robotic dissection without compression of the pancreas may reduce postoperative complications.



Similar content being viewed by others
References
Kitano S, Iso Y, Moriyama M, Sugimachi K. Laparoscopy-assisted Billroth I gastrectomy. Surg Laparosc Endosc. 1994;4(2):146–8.
Obama K, Okabe H, Hosogi H, Tanaka E, Itami A, Sakai Y. Feasibility of laparoscopic gastrectomy with radical lymph node dissection for gastric cancer: from a viewpoint of pancreas-related complications. Surgery. 2010;149:15–21.
Kim W, Kim HH, Han SU, Kim MC, Hyung WJ, Ryu SW, et al. Decreased morbidity of laparoscopic distal gastrectomy compared with open distal gastrectomy for stage I gastric cancer: short-term outcomes from a multicenter randomized controlled trial (KLASS-01). Ann Surg. 2016;263(1):28–35.
Viñuela EF, Gonen M, Brennan MF, Coit DG, Strong VE. Laparoscopic versus open distal gastrectomy for gastric cancer: a meta-analysis of randomized controlled trials and high-quality nonrandomized studies. Ann Surg. 2012;255(3):446–56.
Association Japanese Gastric Cancer. Japanese gastric cancer treatment guidelines 2010 (ver.3). Gastric Cancer. 2011;14(2):112–23.
Hiki N, Honda M, Etoh T, Yoshida K, Kodera Y, Kakeji Y, et al. Higher incidence of pancreatic fistula in laparoscopic gastrectomy. Real-world evidence from a nationwide prospective cohort study. Gastric Cancer. 2018;21(1):162–70.
Kobayashi N, Shinohara H, Haruta S, Ohkura Y, Mizuno A, Ueno M, et al. Process of pancreas head as a risk factor for postoperative pancreatic fistula in laparoscopic gastric cancer surgery. World J Surg. 2016;40(9):2194–201.
Irino T, Hiki N, Ohashi M, Nunobe S, Sano T, Yamaguchi T. The hit and away technique: optimal usage of the ultrasonic scalpel in laparoscopic gastrectomy. Surg Endosc. 2015;30:245–50.
Migita K, Matsumoto S, Wakatsuki K, Ito M, Kunishige T, Nakade H, et al. The anatomical location of the pancreas is associated with the incidence of pancreatic fistula after laparoscopic gastrectomy. Surg Endosc. 2016;30(12):5481–9.
Japanese Gastric Cancer Association, Japanese classification of gastric carcinoma: 3rd English edn., Gastric Cancer, vol 14, no 2; 2011. pp. 101–12.
Okabe H, Obama K, Tsunoda S, Tanaka E, Sakai Y. Advantage of completely laparoscopic gastrectomy with linear stapled reconstruction: a long-term follow-up study. Ann Surg. 2014;259(1):109–16.
Clavien PA, Barkun J, de Oliveira ML, Vauthey JN, Dindo D, Schulick RD, et al. The Clavien–Dindo classification of surgical complications: five-year experience. Ann Surg. 2009;250(2):187–96.
Suda K, Man-I M, Ishida Y, Kawamura Y, Satoh S, Uyama I. Potential advantages of robotic radical gastrectomy for gastric adenocarcinoma in comparison with conventional laparoscopic approach: a single institutional retrospective comparative cohort study. Surg Endosc. 2015;29(3):673–85.
Okabe H, Tsunoda S, Obama K, Tanaka E, Hisamori S, Shinohara H, et al. Feasibility of laparoscopic radical gastrectomy for gastric cancer of clinical stage II or higher: early outcomes in a phase II study (KUGC04). Ann Surg Oncol. 2016;23(Suppl 4):516–23.
Tsujiura M, Hiki N, Ohashi M, Nunobe S, Kumagai K, Ida S, et al. “Pancreas-compressionless gastrectomy”: a novel laparoscopic approach for suprapancreatic lymph node dissection. Ann Surg Oncol. 2017;24(11):3331–7.
Hikage M, Tokunaga M, Makuuchi R, Irino T, Tanizawa Y, Bando E, et al. Comparison of surgical outcomes between robotic and laparoscopic distal gastrectomy for cT1 gastric cancer. World J Surg. 2017;42:1803–10.
Etoh T, Honda M, Kumamaru H, Miyata H, Yoshida K, Kodera Y, et al. Morbidity and mortality from a propensity score-matched, prospective cohort study of laparoscopic versus open total gastrectomy for gastric cancer: data from a nationwide web-based database. Surg Endosc. 2018;32(6):2766–73.
Sano T, Sasako M, Katai H, Maruyama K. Amylase concentration of drainage fluid after total gastrectomy. Br J Surg. 1997;84(9):1310–2.
Fujita T, Ohta M, Ozaki Y, Takahashi Y, Miyazaki S, Harada T, et al. Collateral thermal damage to the pancreas by ultrasonic instruments during lymph node dissection in laparoscopic gastrectomy. Asian J Endosc Surg. 2015;8(3):281–8.
Kim HI, Han SU, Yang HK, Kim YW, Lee HJ, Ryu KW, et al. Multicenter prospective comparative study of robotic versus laparoscopic gastrectomy for gastric adenocarcinoma. Ann Surg. 2016;263(1):103–9.
Funding
This work was supported by operating support grants from Kyoto University.
Author information
Authors and Affiliations
Contributions
OH contributed to the conception, data acquisition, analysis, and writing the manuscript. OK contributed to the conception, data acquisition, analysis, interpretation of data, and revision. MK contributed to the data acquisition. TS, TE, HS, and SY contributed to the interpretation of data and revision.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflicts of interest in association with the present study.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Supplemental Fig.
1. A scatter diagram of the amylase concentrations in drainage fluid and the compression time of the pancreas. Spearman’s rank correlation coefficient (rs) was 0.373 indicating a weak correlation between the two values. Five plots with amylase concentrations of > 5000 were not shown in this figure but were included in the calculation of the correlation coefficient (TIFF 2947 kb)
Rights and permissions
About this article
Cite this article
Okabe, H., Obama, K., Tsunoda, S. et al. Feasibility of robotic radical gastrectomy using a monopolar device for gastric cancer. Surg Today 49, 820–827 (2019). https://doi.org/10.1007/s00595-019-01802-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00595-019-01802-z