Abstract
Background and purpose
We reported previously that hydrogen gas (H2) reduced hepatic ischemia and reperfusion injury (IRI) after prolonged cold storage (CS) of livers retrieved from heart-beating donors. The present study was designed to assess whether H2 reduced hepatic IRI during donation of a cardiac death (DCD) graft with subsequent CS.
Methods
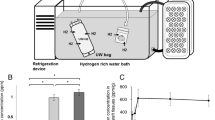
Rat livers were harvested after 30-min cardiac arrest and stored for 4 h in University of Wisconsin solution. The graft was reperfused with oxygenated buffer, with or without H2 (H2 or NT groups, respectively), at 37° for 90 min on isolated perfused rat liver apparatus.
Results
In the NT group, liver enzyme leakage, apoptosis, necrosis, energy depletion, redox status, impaired microcirculation, and bile production were indicative of severe IRI, whereas in the H2 group these impairments were significantly suppressed. The phosphorylation of cytoplasmic MKK4 and JNK were enhanced in the NT group and suppressed in the H2 group. NFkB-p65 and c-Fos in the nucleus were unexpectedly unchanged by IRI regardless of H2 treatment, indicating the absence of inflammation in this model.
Conclusion
H2 was observed to ameliorate IRI in the DCD liver by maintaining microcirculation, mitochondrial functions, and redox status, as well as suppressing the cytoplasmic MKK4–JNK-mediated cellular death pathway.





Similar content being viewed by others
Abbreviations
- ATP:
-
Adenosine triphosphate
- ALT:
-
Alanine aminotransferase
- AST:
-
Aspartate aminotransferase
- CPA:
-
Cardiopulmonary arrest
- DCD:
-
Donation after cardiac death
- DBD:
-
Donation after brain death
- ECD:
-
Expanded criteria donor
- GSSG:
-
Oxidized glutathione
- GSH:
-
Reduced glutathione
- HPFs:
-
High power fields
- H2 :
-
Hydrogen gas
- 4-HNE:
-
4-Hydroxy-2-nonenal
- IRI:
-
Ischemia and reperfusion injury
- IPRL:
-
Isolated perfused rat liver
- JNK:
-
c-jun N-terminal kinase
- KHB:
-
Krebs–Henseleit bicarbonate buffer
- LDH:
-
Lactate dehydrogenase
- LPO:
-
Lipid peroxidation
- MDA:
-
Malondialdehyde
- MKK4:
-
Mitogen-activated protein kinase 4
- NADPH:
-
Nicotinamide adenine dinucleotide phosphate
- NMP:
-
Normothermic machine perfusion
- OCR:
-
Oxygen consumption rate
- PNF:
-
Primary non-function
- PVP:
-
Portal vein pressure
- PVR:
-
Portal vein resistance
- ROS:
-
Reactive oxygen species
- SCD:
-
Standard criteria donor
- SDS-PAGE:
-
SDS polyacrylamide gel electrophoresis
- TUNEL:
-
Terminal deoxynucleotidyl transferase
References
Foley DP, Fernandez LA, Leverson G, Chin LT, Krieger N, Cooper JT, et al. Donation after cardiac death: the University of Wisconsin experience with liver transplantation. Ann Surg. 2005;242:724–31.
Karp SJ, Johnson S, Evenson A, Curry MP, Manning D, Malik R, et al. Minimising cold ischaemic time is essential in cardiac death donor-associated liver transplantation. HPB (Oxford). 2011;13:411–6.
Schlegel A, Kalisvaart M, Scalera I, Laing RW, Mergental H, Mirza DF, et al. The UK DCD risk score: a new proposal to define futility in donation-after-circulatory-death liver transplantation. J Hepatol. 2018;68:456–64.
Nasralla D, Coussios CC, Mergental H, Akhtar MZ, Butler AJ, Ceresa CDL, et al. A randomized trial of normothermic preservation in liver transplantation. Nature. 2018;557:50–6.
Tredger JM. Ischaemia–reperfusion injury of the liver: treatment in theory and in practice. Biofactors. 1998;8:161–4.
Ikeda T, Yanaga K, Kishikawa K, Kakizoe S, Shimada M, Sugimachi K. Ischemic injury in liver transplantation: difference in injury sites between warm and cold ischemia in rats. Hepatology. 1992;16:454–61.
Monbaliu D, Crabbe T, Roskams T, Fevery J, Verwaest C, Pirenne J. Livers from non-heart-beating donors tolerate short periods of warm ischemia. Transplantation. 2005;79:1226–30.
Ohsawa I, Ishikawa M, Takahashi K, Watanabe M, Nishimaki K, Yamagata K, et al. Hydrogen acts as a therapeutic antioxidant by selectively reducing cytotoxic oxygen radicals. Nat Med. 2007;13:688–94.
Kawamura T, Wakabayashi N, Shigemura N, Huang CS, Masutani K, Tanaka Y, et al. Hydrogen gas reduces hyperoxic lung injury via the Nrf2 pathway in vivo. Am J Physiol Lung Cell Mol Physiol. 2013;304:L646–L656.
Sun Q, Kang Z, Cai J, Liu W, Liu Y, Zhang JH, et al. Hydrogen-rich saline protects myocardium against ischemia/reperfusion injury in rats. Exp Biol Med (Maywood). 2009;234:1212–9.
Zheng X, Mao Y, Cai J, Li Y, Liu W, Sun P, et al. Hydrogen-rich saline protects against intestinal ischemia/reperfusion injury in rats. Free Radic Res. 2009;43:478–84.
Abe T, Li XK, Yazawa K, Hatayama N, Xie L, Sato B, et al. Hydrogen-rich University of Wisconsin solution attenuates renal cold ischemia–reperfusion injury. Transplantation. 2012;94:14–21.
Fukuda K, Asoh S, Ishikawa M, Yamamoto Y, Ohsawa I, Ohta S. Inhalation of hydrogen gas suppresses hepatic injury caused by ischemia/reperfusion through reducing oxidative stress. Biochem Biophys Res Commun. 2007;361:670–4.
Liu Q, Shen WF, Sun HY, Fan DF, Nakao A, Cai JM, et al. Hydrogen-rich saline protects against liver injury in rats with obstructive jaundice. Liver Int. 2010;30:958–68.
Uehara T, Bennett B, Sakata ST, Satoh Y, Bilter GK, Westwick JK, et al. JNK mediates hepatic ischemia reperfusion injury. J Hepatol. 2005;42:850–9.
King LA, Toledo AH, Rivera-Chavez FA, Toledo-Pereyra LH. Role of p38 and JNK in liver ischemia and reperfusion. J Hepatobiliary Pancreat Surg. 2009;16:763–70.
Kamata H, Honda S, Maeda S, Chang L, Hirata H, Karin M. Reactive oxygen species promote TNFalpha-induced death and sustained JNK activation by inhibiting MAP kinase phosphatases. Cell. 2005;120:649–61.
Shimada S, Wakayama K, Fukai M, Shimamura T, Ishikawa T, Fukumori D, et al. Hydrogen gas ameliorates hepatic reperfusion injury after prolonged cold preservation in isolated perfused rat liver. Artif Organs. 2016;40(12):1128–1136. https://doi.org/10.1111/aor.12710.
Fukai M, Hayashi T, Yokota R, Shimamura T, Suzuki T, Taniguchi M, et al. Lipid peroxidation during ischemia depends on ischemia time in warm ischemia and reperfusion of rat liver. Free Radic Biol Med. 2005;38:1372–81.
Bessems M, t Hart NA, Tolba R, Doorschodt BM, Leuvenink HG, Ploeg RJ, et al. The isolated perfused rat liver: standardization of a time-honoured model. Lab Anim. 2006;40:236–46.
Matsuno N, Watanabe R, Kimura M, Iwata S, Fujiyama M, Kono S, et al. Beneficial effects of hydrogen gas on porcine liver reperfusion injury with use of total vascular exclusion and active venous bypass. Transplant Proc. 2014;46:1104–6.
Nassar A, Liu Q, Farias K, Buccini L, Baldwin W, Bennett A, et al. Impact of temperature on porcine liver machine perfusion from donors after cardiac death. Artif Organs. 2016;40:999–1008.
Carnevale ME, Balaban CL, Guibert EE, Bottai H, Rodriguez JV. Hypothermic machine perfusion versus cold storage in the rescuing of livers from non-heart-beating donor rats. Artif Organs. 2013;37:985–91.
Imai H, Nakagawa Y. Biological significance of phospholipid hydroperoxide glutathione peroxidase (PHGPx, GPx4) in mammalian cells. Free Radic Biol Med. 2003;34:145–69.
Kobayashi H, Nonami T, Kurokawa T, Kitahara S, Harada A, Nakao A, et al. Changes in the glutathione redox system during ischemia and reperfusion in rat liver. Scand J Gastroenterol. 1992;27:711–6.
Schild L, Reinheckel T, Wiswedel I, Augustin W. Short-term impairment of energy production in isolated rat liver mitochondria by hypoxia/reoxygenation: involvement of oxidative protein modification. Biochem J. 1997;328(Pt 1):205–10.
Bradham CA, Stachlewitz RF, Gao W, Qian T, Jayadev S, Jenkins G, et al. Reperfusion after liver transplantation in rats differentially activates the mitogen-activated protein kinases. Hepatology. 1997;25:1128–35.
Chambers JW, LoGrasso PV. Mitochondrial c-Jun N-terminal kinase (JNK) signaling initiates physiological changes resulting in amplification of reactive oxygen species generation. J Biol Chem. 2011;286:16052–62.
Chambers JW, Pachori A, Howard S, Iqbal S, LoGrasso PV. Inhibition of JNK mitochondrial localization and signaling is protective against ischemia/reperfusion injury in rats. J Biol Chem. 2013;288:4000–11.
Uehara T, Xi Peng X, Bennett B, Satoh Y, Friedman G, Currin R, et al. c-Jun N-terminal kinase mediates hepatic injury after rat liver transplantation. Transplantation. 2004;78:324–32.
Hanawa N, Shinohara M, Saberi B, Gaarde WA, Han D, Kaplowitz N. Role of JNK translocation to mitochondria leading to inhibition of mitochondria bioenergetics in acetaminophen-induced liver injury. J Biol Chem. 2008;283:13565–77.
Zheng H, Yu YS. Chronic hydrogen-rich saline treatment attenuates vascular dysfunction in spontaneous hypertensive rats. Biochem Pharmacol. 2012;83:1269–77.
Haam S, Lee S, Paik HC, Park MS, Song JH, Lim BJ, et al. The effects of hydrogen gas inhalation during ex vivo lung perfusion on donor lungs obtained after cardiac death. Eur J Cardiothorac Surg. 2015;48:542–7.
Shimada S, Fukai M, Wakayama K, Ishikawa T, Kobayashi N, Kimura T, et al. Hydrogen sulfide augments survival signals in warm ischemia and reperfusion of the mouse liver. Surg Today. 2015;45:892–903.
Nakamitsu A, Hiyama E, Imamura Y, Matsuura Y, Yokoyama T. Kupffer cell function in ischemic and nonischemic livers after hepatic partial ischemia/reperfusion. Surg Today. 2001;31:140–8.
Zheng Y, Zhu D. Molecular hydrogen therapy ameliorates organ damage induced by sepsis. Oxidative Med Cell Longev. 2016;2016:5806057.
Zwacka RM, Zhang Y, Zhou W, Halldorson J, Engelhardt JF. Ischemia/reperfusion injury in the liver of BALB/c mice activates AP-1 and nuclear factor kappaB independently of IkappaB degradation. Hepatology. 1998;28:1022–30.
Acknowledgements
We thank Mr. Masatoshi Horigome for the animal care, Ms. Sayaka Miyoshi for technical assistance, and the staff of the Department of Gastroenterological Surgery I, Hokkaido University Graduate School of Medicine, for their kind cooperation.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
We have no conflicts of interest to declare.
Rights and permissions
About this article
Cite this article
Ishikawa, T., Shimada, S., Fukai, M. et al. Post-reperfusion hydrogen gas treatment ameliorates ischemia reperfusion injury in rat livers from donors after cardiac death: a preliminary study. Surg Today 48, 1081–1088 (2018). https://doi.org/10.1007/s00595-018-1693-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00595-018-1693-0