Abstract
Purpose
The treatment outcomes for inflammatory bowel disease (IBD) have been improving, but the development of better therapies is needed. Stem cell therapy is promising, but little is known about the differences in adipose-derived stem cells (ADSCs) between IBD patients and healthy individuals.
Methods
ADSCs were isolated from subcutaneous adipose tissue (SAT) in IBD (Crohn’s disease, 3; ulcerative colitis, 2) and non-IBD (colorectal cancer, 5; breast cancer, 1) patients. We also analyzed the effects of tumor necrosis factor (TNF)-α on murine ADSCs.
Results
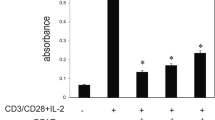
The numbers of stromal vascular fraction (SVF) cells per gram of SAT were 7.72 ± 3.03 × 105 in IBD and 8.51 ± 8.80 × 105 in non-IBD patients (p > 0.05). The proportions of ADSCs in SVF cells were 4.98 ± 2.61% in IBD and 1.02 ± 0.67% in non-IBD patients. The numbers of ADSCs per gram of SAT were 4.16 ± 2.96 × 104 in IBD and 0.88 ± 1.04 × 104 in non-IBD patients. The proportions and numbers of ADSCs were significantly higher in IBD patients than in non-IBD patients. TNF-α significantly facilitated the proliferation and motility of murine ADSCs.
Conclusion
These results showed the potential advantage of freshly isolated autologous ADSCs in IBD patients.



Similar content being viewed by others
References
Yui S, Nakamura T, Sato T, Nemoto Y, Mizutani T, Zheng X, et al. Functional engraftment of colon epithelium expanded in vitro from a single adult Lgr5(+) stem cell. Nat Med. 2012;18:618–23.
Rubin JP, Marra KG. Adipose stem cell therapy for soft tissue reconstruction. Lancet. 2013;382:1077–9.
Fisher SA, Brunskill SJ, Doree C, Mathur A, Taggart DP, Martin-Rendon E. Stem cell therapy for chronic ischaemic heart disease and congestive heart failure. Cochrane Database Syst Rev. 2016;12:CD007888.
El-Badawy A, El-Badri N. Clinical efficacy of stem cell therapy for diabetes mellitus: a meta-analysis. PLoS One. 2016;11:e0151938.
Dentelli P, Barale C, Togliatto G, Trombetta A, Olgasi C, Gili M, et al. A diabetic milieu promotes OCT4 and NANOG production in human visceral-derived adipose stem cells. Diabetologia. 2013;56:173–84.
Iwen KA, Priewe A-C, Minnefeld M, Rose C, Siemers F, Rohwedel J, et al. Gluteal and abdominal subcutaneous adipose tissue depots as stroma cell source: gluteal cells display increased adipogenic and osteogenic differentiation potentials. Exp Dermatol. 2014;23:395–400.
Ohmura Y, Tanemura M, Kawaguchi N, Machida T, Tanida T, Deguchi T, et al. Combined transplantation of pancreatic islets and adipose tissue-derived stem cells enhances the survival and insulin function of islet grafts in diabetic mice. Transplantation. 2010;90:1366–73.
Zhao QJ, Yu YB, Zuo XL, Dong YY, Li YQ. Milk fat globule-epidermal growth factor 8 is decreased in intestinal epithelium of ulcerative colitis patients and thereby causes increased apoptosis and impaired wound healing. Mol Med. 2012;18:497–506.
Lenoir N. Europe confronts the embryonic stem cell research challenge. Science. 2000;287:1425–7.
Takahashi K, Yamanaka S. Induction of pluripotent stem cells from mouse embryonic and adult fibroblast cultures by defined factors. Cell. 2006;126:663–76.
Ohgushi H, Caplan AI. Stem cell technology and bioceramics: from cell to gene engineering. J Biomed Mater Res. 1999;48:913–27.
Al-Saqi SH, Saliem M, Asikainen S, Quezada HC, Ekblad A, Hovatta O, et al. Defined serum-free media for in vitro expansion of adipose-derived mesenchymal stem cells. Cytotherapy. 2014;16:915–26.
Anderson P, Souza-Moreira L, Morell M, Caro M, O’Valle F, Gonzalez-Rey E, et al. Adipose-derived mesenchymal stromal cells induce immunomodulatory macrophages which protect from experimental colitis and sepsis. Gut. 2013;62:1131–41.
Kilroy GE, Foster SJ, Wu X, Ruiz J, Sherwood S, Heifetz A, et al. Cytokine profile of human adipose-derived stem cells: expression of angiogenic, hematopoietic, and pro-inflammatory factors. J Cell Physiol. 2007;212:702–9.
Garcia-Olmo D, Garcia-Arranz M, Garcia LG, Cuellar ES, Blanco IF, Prianes LA, et al. Autologous stem cell transplantation for treatment of rectovaginal fistula in perianal Crohn’s disease: a new cell-based therapy. Int J Colorectal Dis. 2003;18:451–4.
Lee WY, Park KJ, Cho YB, Yoon SN, Song KH, Kim DS, et al. Autologous adipose tissue-derived stem cells treatment demonstrated favorable and sustainable therapeutic effect for Crohn’s fistula. Stem Cells. 2013;31:2575–81.
Mizushima T, Takahashi H, Takeyama H, Naito A, Haraguchi N, Uemura M, et al. A clinical trial of autologous adipose-derived regenerative cell transplantation for a postoperative enterocutaneous fistula. Surg Today. 2016;46:835–42.
Panes J, Garcia-Olmo D, Assche GV, Colombel JF, Reinisch W, Baumgart DC. Expanded allogeneic adipose-derived mesenchymal stem cells (Cx601) for complex perianal fistulas in Crohn’s disease: a phase 3 randomised, double-blind controlled trial. Lancet. 2016;388:1281–90.
Ciccocioppo R, Bernardo ME, Sgarella A, Maccario R, Avanzini MA, Ubezio C, et al. Autologous bone marrow-derived mesenchymal stromal cells in the treatment of fistulising Crohn’s disease. Gut. 2011;60:788–98.
Garcia-Bosch O, Ricart E, Panes J. Review article: stem cell therapies for inflammatory bowel disease—efficacy and safety. Aliment Pharmacol Ther. 2010;32:939–52.
Duijvestein M, Vos ACW, Roelofs H, Wildenberg ME, Wendrich BB, Verspaget HW, et al. Autologous bone marrow-derived mesenchymal stromal cell treatment for refractory luminal Crohn’s disease: results of a phase I study. Gut. 2010;59:1662–9.
Forbes GM, Sturm MJ, Leong RW, Sparrow MP, Segarajasingam D, Cummins AG, et al. A phase 2 study of allogeneic mesenchymal stromal cells for luminal Crohn’s disease refractory to biologic therapy. Clin Gastroenterol Hepatol. 2014;12:64–71.
Izadpanah R, Trygg C, Patel B, Kriedt C, Dufour J, Gimble JM, et al. Biologic properties of mesenchymal stem cells derived from bone marrow and adipose tissue. J Cell Biochem. 2006;99:1285–97.
Baek SJ, Kang SK, Ra JC. In vitro migration capacity of human adipose tissue-derived mesenchymal stem cells reflects their expression of receptors for chemokines and growth factors. Exp Mol Med. 2011;43:596–603.
Suga H, Eto H, Shigeura T, Inoue K, Aoi N, Kato H, et al. IFATS collection: fibroblast growth factor-2-induced hepatocyte growth factor secretion by adipose-derived stromal cells inhibits postinjury fibrogenesis through a c-Jun N-terminal kinase-dependent mechanism. Stem Cells. 2009;27:238–49.
Pallagi-Kunstár É, Farkas K, Szepes Z, Nagy F, Szűcs M, Kui R, et al. Utility of serum TNF-α, infliximab trough level, and antibody titers in inflammatory bowel disease. World J Gastroenterol. 2014;20:5031–5.
Bertin B, Desreumaux P, Dubuquoy L. Obesity, visceral fat and Crohn’s disease. Curr Opin Clin Nutr Metab Care. 2010;13:574–80.
Acknowledgements
This work was supported by a Grant-in-Aid for Scientific Research.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflicts of interests.
Rights and permissions
About this article
Cite this article
Mizushima, T., Fukata, T., Takeyama, H. et al. The features of adipose-derived stem cells in patients with inflammatory bowel diseases. Surg Today 48, 352–358 (2018). https://doi.org/10.1007/s00595-017-1591-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00595-017-1591-x