Abstract
Purpose
This single-institutional study was designed to determine whether S-1, an oral fluoropyrimidine, plus cisplatin with concurrent radiotherapy is feasible as an induction treatment for locally advanced non-small cell lung cancer (NSCLC).
Methods
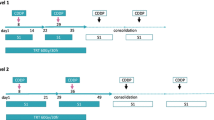
Eighteen patients were analyzed in this study from July 2005 to March 2008. The patients received 40 mg/m2 S-1 orally twice per day on days 1 through 14 and 22 through 35, and cisplatin (60 mg/m2) was injected intravenously on days 8 and 29. The patients also underwent radiotherapy, and received a total dose of 40 Gy in 20 fractions beginning on day 1. Surgical resection was performed from 3 to 6 weeks after completing the induction treatment.
Results
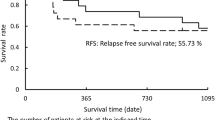
Nine (50%) of the 18 patients who received the induction treatment achieved a partial response. One patient refused to undergo surgery. The remaining 17 patients underwent a complete surgical resection. There were no deaths nor any major morbidities during the perioperative period. The recurrence-free survival and overall survival rate at 2 years for the patients who underwent resection were 63.3% and 88.2%, respectively.
Conclusion
Induction treatment using S-1 plus cisplatin and concurrent radiotherapy and surgical resection for selected patients with stage III NSCLC is a feasible and promising new treatment modality.
Similar content being viewed by others
References
Albain KS, Swann RS, Rusch VW, Turrisi AT 3rd, Shepherd FA, Smith C, et al. Radiotherapy plus chemotherapy with or without surgical resection for stage III non-small-cell lung cancer: a phase III randomised controlled trial. Lancet 2009;374:379–386.
van Meerbeeck JP, Kramer GWPM, Van Schil PEY, Legrand C, Smit EF, Schramel F, et al. Randomized controlled trial of resection versus radiotherapy after induction chemotherapy in stage IIIA-N2 non-small-cell lung cancer. J Natl Cancer Inst 2007;99:442–450.
Schaake-Koning C, van den Bogaert W, Dalesio O, Festen J, Hoogenhout J, van Houtte P, et al. Effects of concomitant cisplatin and radiotherapy on inoperable non-small-cell lung cancer. N Engl J Med 1992;326:524–530.
Segawa Y, Ueoka H, Kiura K, Kamei H, Tabata M, Sakae K, et al. A phase II study of cisplatin and 5-fluorouracil with concurrent hyperfractionated thoracic radiation for locally advanced nonsmall-cell lung cancer: a preliminary report from the Okayama Lung Cancer Study Group. Br J Cancer 2000;82:104–111.
Ichinose Y, Nakai Y, Kudoh S, Semba H, Yoshida S, Nukiwa T, et al. Uracil/tegafur plus cisplatin with concurrent radiotherapy for locally advanced non-small-cell lung cancer: a multi-institutional phase II trial. Clin Cancer Res 2004;10:4369–4373.
Ichinose Y, Fukuyama Y, Asoh H, Ushijima C, Okamoto T, Ikeda J, et al. Induction chemoradiotherapy and surgical resection for selected stage IIIB non-small-cell lung cancer. Ann Thorac Surg 2003;76:1810–1814.
Shirasaka T, Shimamato Y, Ohshimo H, Yamaguchi M, Kato T, Yonekura K, et al. Development of a novel form of an oral 5-fluorouracil derivative (S-1) directed to the potentiation of the tumour selective cytotoxicity of 5-fluorouracil by two biochemical modulators. Anticancer Drugs 1996;7:548–557.
Ohyanagi F, Yamamoto N, Horiike A, Harada H, Kozuka T, Murakami H, et al. Phase II trial of S-1 and cisplatin with concurrent radiotherapy for locally advanced non-small-cell lung cancer. Br J Cancer 2009;101:225–231.
The Japan Lung Cancer Society (editor). General rules for clinical and pathological record of lung cancer. 6th ed. Tokyo: Kanehara; 2003.
Travis WD CT, Corrin B, Shimosato Y, Brambilla E; Collaborators from 14 Countries. World Health Organization. International histological classification of tumors. Histological typing of lung and pleural tumors. 3rd ed. Berlin: Springer; 1999.
Therasse P, Arbuck SG, Eisenhauer EA, Wanders J, Kaplan RS, Rubinstein L, et al. New guidelines to evaluate the response to treatment in solid tumors. European Organization for Research and Treatment of Cancer, National Cancer Institute of the United States, National Cancer Institute of Canada. J Natl Cancer Inst 2000;92:205–216.
Kaplan E, Meier P. Nonparametric estimation from incomplete observation. J Am Stat Assoc 1958;53:457–481.
Thomas M, Rube C, Hoffknecht P, Macha HN, Freitag L, Linder A, et al. Effect of preoperative chemoradiation in addition to preoperative chemotherapy: a randomised trial in stage III nonsmall-cell lung cancer. Lancet Oncol 2008;9:636–648.
Allen AM, Mentzer SJ, Yeap BY, Soto R, Baldini EH, Rabin MS, et al. Pneumonectomy after chemoradiation: the Dana-Farber Cancer Institute/Brigham and Women’s Hospital experience. Cancer 2008;112:1106–1113.
Shrager JB. Mediastinoscopy: still the gold standard. Ann Thorac Surg 2010;89:S2084–S2089.
Detterbeck FC, Jantz MA, Wallace M, Vansteenkiste J, Silvestri GA. Invasive mediastinal staging of lung cancer: ACCP evidencebased clinical practice guidelines (2nd ed.). Chest 2007;132: 202S–220S.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Maruyama, R., Hirai, F., Ondo, K. et al. Concurrent chemoradiotherapy using cisplatin plus s-1, an oral fluoropyrimidine, followed by surgery for selected patients with stage III non-small cell lung cancer: A single-center feasibility study. Surg Today 41, 1492–1497 (2011). https://doi.org/10.1007/s00595-011-4509-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00595-011-4509-z