Abstract
Purpose
To investigate the effects of metyrosine, lacidipine, clonidine, and moxonidine on the renal damage in rats with unilateral ureteral ligation by examining the histological evidence of parenchymal damage and tubular dilatation, as well as biochemical changes indicating cell membrane damage and DNA oxidation.
Methods
Thirty-six albino Wistar rats were randomly divided into six equal groups: a healthy (intact) group, a unilateral ureteral ligation (control) group, and four drug treatment groups given metyrosine (50 mg/kg), lacidipine (2 mg/kg), clonidine (0.075 mg/kg), or moxonidine (0.2 mg/kg), respectively, for 10 days. The latter five groups underwent ligation of the left ureter. Ten days after the operation, we removed both kidneys from each rat in the control and drug treatment groups for renal pathological and biochemical [malondialdehyde (MDA), total glutathione, 8-hydroxy-2-deoxyguanine (8-OH-Gua)] examinations. Spectrophotometric assays were used to detect the malondialdehyde and total glutathione levels of the renal tissue. High-performance liquid chromatography was used to measure the 8-hydroxy-2-deoxyguanine levels.
Results
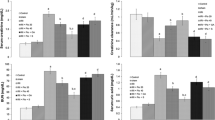
When the drug treatment groups were compared with the control group, the drug treatment groups’ total glutathione level was higher and their malondialdehyde level was lower than that of the control group (P < 0.05), especially in the clonidine group (P < 0.0001). The 8-hydroxy-2-deoxyguanine levels of the drug treatment groups, except the lacidipine group, were significantly lower than that of the control group (P < 0.0001). There was no significant difference between the contralateral kidneys of the treatment groups and control group, according to the biochemical results. As revealed via light microscopy, clonidine and moxonidine treatment significantly reduced the tubular and glomerular damage, as well as the tubular dilation. The interstitial inflammation of the kidneys in the lacidipine group was higher than that of the other treatment groups. However, the apoptotic cell count was at a high level in both the lacidipine and metyrosine groups. The increase in the collagen content was most pronounced in the lacidipine and metyrosine groups. An examination of the contralateral kidneys showed no marked pathological findings.
Conclusions
The use of a direct or indirect α2-adrenergic receptor agonist for the temporary treatment of unilateral ureteral obstruction-induced renal damage may be important for preventing renal structural injury. A more advanced study is necessary to determine the mechanisms underlying the protective effects of these drugs with regard to renal damage in ureteral obstruction.






Similar content being viewed by others
References
Moosavi SM, Ashtiyani SC, Hosseinkhani S, Shirazi M. Comparison of the effects of l-carnitine and alpha-tocopherol on acute ureteral obstruction-induced renal oxidative imbalance and altered energy metabolism in rats. Urol Res. 2010;38:187–94.
Chevalier RL, Peters CA. Congenital urinary tract obstruction: proceedings of the state-of-the-art strategic planning workshop, National Institutes of Health, Bethesda, Maryland, USA, 11–12 March 2002. Pediatr Nephrol. 2003;18:576–606.
Felsen D, Loo MH, Vaughan ED Jr. Effect of ureteral obstruction on renal hemodynamics. Semin Urol. 1987;5:160–6.
Fink RL, Caridis DT, Chmiel R, Ryan G. Renal impairment and its reversibility following variable periods of complete ureteric obstruction. Aust N Z J Surg. 1980;50:77–83.
Anderson D. Antioxidant defences against reactive oxygen species causing genetic and other damage. Mutat Res. 1996;350:103–8.
Naito Y, Yoshikawa T, Yoshida N, Kondo M. Role of oxygen radical and lipid peroxidation in indomethacin-induced gastric mucosal injury. Dig Dis Sci. 1998;43:30S–4S.
Bast A, Haenen GR, Doelman CJ. Oxidants and antioxidants: state of the art. Am J Med. 1991;91:2S–13S.
Grollman AP, Moriya M. Mutagenesis by 8-oxoguanine: an enemy within. Trends Genet. 1993;9:246–9.
Polat B, Suleyman H, Alp HH. Adaptation of rat gastric tissue against indomethacin toxicity. Chem Biol Interact. 2010;186:82–9.
Mizuguchi Y, Miyajima A, Kosaka T, Asano T, Hayakawa M. Atorvastatin ameliorates renal tissue damage in unilateral ureteral obstruction. J Urol. 2004;172:2456–9.
Moriyama T, Kawada N, Nagatoya K, Takeji M, Horio M, Ando A, et al. Fluvastatin suppresses oxidative stress and fibrosis in the interstitium of mouse kidneys with unilateral ureteral obstruction. Kidney Int. 2001;59:2095–103.
Albayrak A, Polat B, Cadirci E, Hacimuftuoglu A, Halici Z, Gulapoglu M, et al. Gastric anti-ulcerative and anti-inflammatory activity of metyrosine in rats. Pharmacol Rep. 2010;62:113–9.
Halici Z, Borekci B, Ozdemir Y, Cadirci E, Suleyman H. Protective effects of amlodipine and lacidipine on ovariectomy-induced bone loss in rats. Eur J Pharmacol. 2008;579:241–5.
Poyhonen-Alho MK, Manhem K, Katzman P, Kibarskis A, Antikainen RL, Erkkola RU, et al. Central sympatholytic therapy has anti-inflammatory properties in hypertensive postmenopausal women. J Hypertens. 2008;26:2445–9.
Robert B, Petit JY, Grimaud N, Juge M, Welin L. Effect of clonidine on experimental brain edema in the rat. Agents Actions. 1992;37:268–72.
Suleyman H, Halici Z, Hacimuftuoglu A, Gocer F. Role of adrenal gland hormones in antiinflammatory effect of calcium channel blockers. Pharmacol Rep. 2006;58:692–9.
Nasrallah HA, Donnelly EF, Bigelow LB, Rivera-Calimlim L, Rogol A, Potkin S, et al. Inhibition of dopamine synthesis in chronic schizophrenia. Clinical ineffectiveness of metyrosine. Arch Gen Psychiatry. 1977;34:649–55.
De Paoli P, Cerbai E, Koidl B, Kirchengast M, Sartiani L, Mugelli A. Selectivity of different calcium antagonists on T- and L-type calcium currents in guinea-pig ventricular myocytes. Pharmacol Res. 2002;46:491–7.
Chachua T, Bilanishvili I, Khizanishvili N, Nanobashvili Z. Noradrenergic modulation of seizure activity. Georgian Med News. 2010:34–9.
Nasr MA, El-Gowilly SM, El-Mas MM. Comparable renovascular protective effects of moxonidine and simvastatin in rats exposed to cigarette smoke. Vascul Pharmacol. 2010;53:53–60.
Salman S, Kumbasar S, Yilmaz M, Kumtepe Y, Borekci B, Bakan E, et al. Investigation of the effects of the chronic administration of some antihypertensive drugs on enzymatic and non-enzymatic oxidant/antioxidant parameters in rat ovarian tissue. Gynecol Endocrinol. 2011;27:895–9.
Kang DH, Joly AH, Oh SW, Hugo C, Kerjaschki D, Gordon KL, et al. Impaired angiogenesis in the remnant kidney model: I. Potential role of vascular endothelial growth factor and thrombospondin-1. J Am Soc Nephrol. 2001;12:1434–47.
Sedlak J, Lindsay RH. Estimation of total, protein-bound, and nonprotein sulfhydryl groups in tissue with Ellman’s reagent. Anal Biochem. 1968;25:192–205.
Ohkawa H, Ohishi N, Yagi K. Assay for lipid peroxides in animal tissues by thiobarbituric acid reaction. Anal Biochem. 1979;95:351–8.
Bradford MM. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem. 1976;72:248–54.
Kaur H, Halliwell B. Measurement of oxidized and methylated DNA bases by HPLC with electrochemical detection. Biochem J. 1996;318(Pt 1):21–3.
Floyd RA, Watson JJ, Wong PK, Altmiller DH, Rickard RC. Hydroxyl free radical adduct of deoxyguanosine: sensitive detection and mechanisms of formation. Free Radic Res Commun. 1986;1:163–72.
Asami S, Hirano T, Yamaguchi R, Tomioka Y, Itoh H, Kasai H. Increase of a type of oxidative DNA damage, 8-hydroxyguanine, and its repair activity in human leukocytes by cigarette smoking. Cancer Res. 1996;56:2546–9.
Del Soldato P. Gastric lesion-preventing or -potentiating activity of clonidine in rats. Jpn J Pharmacol. 1986;41:257–9.
Suleyman H, Halici Z, Cadirci E, Hacimuftuoglu A, Keles S, Gocer F. Indirect role of alpha2-adrenoreceptors in anti-ulcer effect mechanism of nimesulide in rats. Naunyn Schmiedebergs Arch Pharmacol. 2007;375:189–98.
Suleyman H, Dursun H, Bilici M, Cadirci E, Halici Z, Gulaboglu M, et al. Relation of adrenergic receptors, which have roles in gastroprotective and anti-inflammatory effect of adrenal gland hormones, with cyclooxygenase enzyme levels in rats. J Physiol Pharmacol. 2009;60:129–34.
Suleyman H, Demircan B, Karagoz Y. Anti-inflammatory and side effects of cyclooxygenase inhibitors. Pharmacol Rep. 2007;59:247–58.
Suleyman H, Halici Z, Cadirci E, Hacimuftuoglu A, Bilen H. Indirect role of beta2-adrenergic receptors in the mechanism of anti-inflammatory action of NSAIDS. J Physiol Pharmacol. 2008;59:661–72.
Suleyman H, Cadirci E, Albayrak A, Halici Z. Nimesulide is a selective COX-2 inhibitory, atypical non-steroidal anti-inflammatory drug. Curr Med Chem. 2008;15:278–83.
Ghanayem BI, Boor PJ, Ahmed AE. Acrylonitrile-induced gastric mucosal necrosis: role of gastric glutathione. J Pharmacol Exp Ther. 1985;232:570–7.
Stein HJ, Hinder RA, Oosthuizen MM. Gastric mucosal injury caused by hemorrhagic shock and reperfusion: protective role of the antioxidant glutathione. Surgery 1990;108:467–73 (discussion 473–4).
Lomaestro BM, Malone M. Glutathione in health and disease: pharmacotherapeutic issues. Ann Pharmacother. 1995;29:1263–73.
Blaustein A, Deneke SM, Stolz RI, Baxter D, Healey N, Fanburg BL. Myocardial glutathione depletion impairs recovery after short periods of ischemia. Circulation. 1989;80:1449–57.
Freeman BA, Crapo JD. Biology of disease: free radicals and tissue injury. Lab Invest. 1982;47:412–26.
Marnett LJ. Lipid peroxidation-DNA damage by malondialdehyde. Mutat Res. 1999;424:83–95.
Milligan JR, Ward JF. Yield of single-strand breaks due to attack on DNA by scavenger-derived radicals. Radiat Res. 1994;137:295–9.
Cadet J, Douki T, Gasparutto D, Ravenna JL. Oxidative damage to DNA: formation, measurement and biochemical features. Mutat Res. 2003;531:5–23.
Collins AR, Dusinska M, Gedik CM, Stetina R. Oxidative damage to DNA: do we have a reliable biomarker? Environ Health Perspect. 1996;104(Suppl 3):465–9.
Borekci B, Kumtepe Y, Karaca M, Halici Z, Cadirci E, Albayrak F, et al. Role of alpha-2 adrenergic receptors in anti-ulcer effect mechanism of estrogen and luteinising hormone on rats. Gynecol Endocrinol. 2009;25:264–8.
Kumtepe Y, Borekci B, Karaca M, Salman S, Alp HH, Suleyman H. Effect of acute and chronic administration of progesterone, estrogen, FSH and LH on oxidant and antioxidant parameters in rat gastric tissue. Chem Biol Interact. 2009;182:1–6.
Conflict of interest
None of the authors has any conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Yigiter, M., Yildiz, A., Polat, B. et al. The protective effects of metyrosine, lacidipine, clonidine, and moxonidine on kidney damage induced by unilateral ureteral obstruction in rats. Surg Today 42, 1051–1060 (2012). https://doi.org/10.1007/s00595-011-0074-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00595-011-0074-8