Abstract
Aims
As an emerging interventional technique to treat resistant hypertension, renal denervation (RDN) has also attracted considerable attention due to its potential beneficial effects on glucose and lipid metabolism. Given that inconsistent results were documented among studies, we aimed to perform a systematic review and meta-analysis to elaborate on this issue.
Methods
The PubMed, EMBASE, Web of Science (SCI) and ClinicalTrials.gov databases were comprehensively searched from their inception date to June 18, 2020, for relevant clinical studies evaluating the efficacy of RDN on glucose and lipid levels. The outcomes of interest were changes in fasting glucose, insulin, C-peptide, hemoglobin A1C (HbA1C), homeostatic model assessment-insulin resistance (HOMA-IR), cholesterol and triglyceride (TG) levels before versus after RDN and also RDN versus the control group. The mean differences (MDs) of the outcomes measured before versus after RDN and RDN versus the control group were pooled by a randomized effects model. Heterogeneity was quantified with Chi-square (χ2) and inconsistency index (I2). Assessment of publication bias was performed by the funnel plot and Egger’s test.
Results
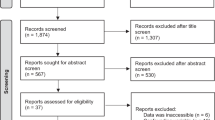
A total of 1600 studies were initially identified. Nineteen of the identified studies (six randomized controlled studies, one non-randomized controlled studies and 12 observational cohort studies) involving 2245 subjects were included in the final analysis. No significant change was observed after RDN in fasting glucose (weighted mean difference [WMD] − 0.19 mmol/L; 95% CI − 0.37, 0.00 mmol/L), insulin (standardized mean difference [SMD] − 0.01; 95% CI − 0.41, 0.39), C-peptide (SMD − 0.05; 95% CI − 0.30, 0.21), HbA1C (SMD − 0.05; 95% CI − 0.17, 0.07), HOMA-IR (SMD − 0.29; 95% CI − 0.72, 0.14), total cholesterol (TC) (WMD − 0.11 mmol/L; 95% CI − 0.37, 0.15 mmol/L), and low-density lipoprotein cholesterol (LDL-C) levels (WMD − 0.18 mmol/L; 95% CI − 0.59, 0.24 mmol/L) during follow-up. Changes in fasting glucose, insulin, HbA1C and TC levels in RDN groups were not significantly different from those in the control group. High-density lipoprotein cholesterol (HDL-C) and TG were slightly improved after RDN (WMD 0.07 mmol/L, 95% CI 0.01, 0.14 mmol/L; WMD − 0.26 mmol/l, 95% CI − 0.51, − 0.01 mmol/L, respectively). The funnel plot and Egger’s test demonstrated the absence of potential publication bias.
Conclusions
Catheter-based RDN appeared to have no impact on glucose metabolism. There was a statistically significant but clinically negligible improvement in HDL-C and TG levels based on the current evidence. Future research with more rigorous designs is warranted to draw definitive conclusions.
Registration details
The protocol of this meta-analysis was registered on PROSPERO (CRD42020192805). (https://www.crd.york.ac.uk/PROSPERO/display_record.php?RecordID=192805)






Similar content being viewed by others
References
DiBona GF (2005) Physiology in perspective. The wisdom of the body neural control of the kidney. Am J Physiol Regul Integr Comp Physiol 289(3):R633–R641
Hayek SS, Abdou MH, Demoss BD, Legaspi JM, Veledar E, Deka A et al (2013) Prevalence of resistant hypertension and eligibility for catheter-based renal denervation in hypertensive outpatients. Am J Hypertens 26(12):1452–1458
Carey RM, Calhoun DA, Bakris GL, Brook RD, Daugherty SL, Dennison-Himmelfarb CR et al (2018) Resistant hypertension: detection, evaluation, and management: a scientific statement from the American heart association. Hypertension 72(5):e53–e90
Williams B, Mancia G, Spiering W, Agabiti Rosei E, Azizi M, Burnier M et al (2018) Practice guidelines for the management of arterial hypertension of the European Society of Hypertension and the European Society of Cardiology: ESH/ESC task force for the management of arterial hypertension. J Hypertens 36(12):2284–2309
Krum H, Schlaich M, Whitbourn R, Sobotka PA, Sadowski J, Bartus K et al (2009) Catheter-based renal sympathetic denervation for resistant hypertension: a multicentre safety and proof-of-principle cohort study. The Lancet 373(9671):1275–1281
Symplicity HTNI, Esler MD, Krum H, Sobotka PA, Schlaich MP, Schmieder RE et al (2010) Renal sympathetic denervation in patients with treatment-resistant hypertension (the symplicity HTN-2 trial): a randomised controlled trial. Lancet 376(9756):1903–1909
Bhatt DL, Kandzari DE, O’Neill WW, D’Agostino R, Flack JM, Katzen BT et al (2014) A controlled trial of renal denervation for resistant hypertension. N Engl J Med 370(15):1393–1401
Kandzari DE, Bhatt DL, Brar S, Devireddy CM, Esler M, Fahy M et al (2015) Predictors of blood pressure response in the SYMPLICITY HTN-3 trial. Eur Heart J 36(4):219–227
Böhm M, Kario K, Kandzari DE, Mahfoud F, Weber MA, Schmieder RE et al (2020) Efficacy of catheter-based renal denervation in the absence of antihypertensive medications (SPYRAL HTN-OFF MED Pivotal): a multicentre, randomised, sham-controlled trial. The Lancet 395(10234):1444–1451
Kandzari DE, Böhm M, Mahfoud F, Townsend RR, Weber MA, Pocock S et al (2018) Effect of renal denervation on blood pressure in the presence of antihypertensive drugs: 6-month efficacy and safety results from the SPYRAL HTN-ON MED proof-of-concept randomised trial. The Lancet 391(10137):2346–2355
Mahfoud F, Schlaich M, Kindermann I, Ukena C, Cremers B, Brandt MC et al (2011) Effect of renal sympathetic denervation on glucose metabolism in patients with resistant hypertension a pilot study. Circulation 123(18):1940–1946
Pourmoghaddas M, Khosravi A, Akhbari M, Akbari M, Purbehi M, Ziaei F et al (2016) One year follow-up effect of renal sympathetic denervation in patients with resistant hypertension. Arya Atherosclerosis 12(2):109–113
D. Moher, A. Liberati, J. Tetzlaff, D.G. Altman, P. Group (2009) Preferred reporting items for systematic reviews and meta-analyses: the PRISMA statement. Ann Intern Med 151(4):264–269
Stang A (2010) Critical evaluation of the Newcastle-Ottawa scale for the assessment of the quality of nonrandomized studies in meta-analyses. Eur J Epidemiol 25(9):603–605
Verloop WL, Spiering W, Vink EE, Beeftink MMA, Blankestijn PJ, Doevendans PA et al (2015) Denervation of the renal arteries in metabolic syndrome the DREAMS-study. Hypertension 65(4):751–757
Falkovskaya AY, Mordovin VF, Pekarsky SY, Bayev AY, Semke GV, Ripp TM et al (2015) Dynamics of glycemic control after renal denervation in patients with resistant hypertension and type 2 diabetes mellitus. Byulleten Sibirskoy Meditsiny 14(5):83–90
Matous D, Jiravsky O, Nykl I, Branny M (2015) Effect of renal denervation on glucose metabolism after a 12 month follow-up. Biomedical Papers-Olomouc 159(2):246–250
Eikelis N, Hering D, Marusic P, Duval J, Hammond LJ, Walton AS et al (2017) The effect of renal denervation on plasma adipokine profile in patients with treatment resistant hypertension. Front Physiol 8:369
Tsioufis C, Dimitriadis K, Kasiakogias A, Kalos T, Liatakis I, Koutra E et al (2017) Effects of multielectrode renal denervation on elevated sympathetic nerve activity and insulin resistance in metabolic syndrome. J Hypertens 35(5):1100–1108
Kampmann U, Mathiassen ON, Christensen KL, Buus NH, Bjerre M, Vase H et al (2017) Effects of renal denervation on insulin sensitivity and inflammatory markers in nondiabetic patients with treatment-resistant hypertension. J Diabetes Res 2017:1–9
Witkowski A, Prejbisz A, Florczak E, Kadziela J, Sliwinski P, Bielen P et al (2011) Effects of renal sympathetic denervation on blood pressure, sleep apnea course, and glycemic control in patients with resistant hypertension and sleep apnea. Hypertension 58(4):559–565
Daniels F, De Freitas S, Smyth A, Garvey J, Judge C, Gilmartin JJ et al (2017) Effects of renal sympathetic denervation on blood pressure, sleep apnoea severity and metabolic indices: a prospective cohort study. Sleep Med 30:180–184
Aripov M, Mussayev A, Alimbayev S, Goncharov A, Zhusupova G, Pya Y (2017) Individualised renal artery denervation improves blood pressure control in Kazakhstani patients with resistant hypertension. Kardiologia Polska 75(2):101–107
Miroslawska AK, Gjessing PF, Solbu MD, Fuskevåg OM, Jenssen TG, Steigen TK (2016) Renal denervation for resistant hypertension fails to improve insulin resistance as assessed by hyperinsulinemic-euglycemic step clamp. Diabetes 65(8):2164–2168
Hopper I, Gronda E, Hoppe UC, Rundqvist B, Marwick TH, Shetty S et al (2017) Sympathetic response and outcomes following renal denervation in patients with chronic heart failure: 12-month outcomes from the symplicity HF feasibility study. J Cardiac Fail 23(9):702–707
Bohm M, Mahfoud F, Ukena C, Hoppe UC, Narkiewicz K, Negoita M et al (2015) First report of the Global SYMPLICITY Registry on the effect of renal artery denervation in patients with uncontrolled hypertension. Hypertension 65(4):766–774
Rosa J, Widimsky P, Tousek P, Petrak O, Curila K, Waldauf P et al (2015) Randomized comparison of renal denervation versus intensified pharmacotherapy including spironolactone in true-resistant hypertension: six-month results from the Prague-15 study. Hypertension 65(2):407–413
Warchol-Celinska E, Prejbisz A, Kadziela J, Florczak E, Januszewicz M, Michalowska I et al (2018) Renal denervation in resistant hypertension and obstructive sleep apnea: randomized proof-of-concept phase II trial. Hypertension 72(2):381–390
Weber MA, Kirtane AJ, Weir MR, Radhakrishnan J, Das T, Berk M et al (2020) The REDUCE HTN: REINFORCE: randomized, sham-controlled trial of bipolar radiofrequency renal denervation for the treatment of hypertension. JACC Cardiovasc Interv 13(4):461–470
Donazzan L, Mahfoud F, Ewen S, Ukena C, Cremers B, Kirsch CM et al (2016) Effects of catheter-based renal denervation on cardiac sympathetic activity and innervation in patients with resistant hypertension. Clin Res Cardiol 105(4):364–371
Cohen-Mazor M, Mathur P, Stanley JR, Mendelsohn FO, Lee H, Baird R et al (2014) Evaluation of renal nerve morphological changes and norepinephrine levels following treatment with novel bipolar radiofrequency delivery systems in a porcine model. J Hypertens 32(8):1678–1691 (discussion 1691-2)
Hering D, Lambert EA, Marusic P, Walton AS, Krum H, Lambert GW et al (2013) Substantial reduction in single sympathetic nerve firing after renal denervation in patients with resistant hypertension. Hypertension 61(2):457–464
Pan T, Guo JH, Ling L, Qian Y, Dong YH, Yin HQ et al (2018) Effects of multi-electrode renal denervation on insulin sensitivity and glucose metabolism in a canine model of type 2 diabetes mellitus. J Vasc Interv Radiol 29(5):731-738e2
Chen W, Chang Y, He L, Jian X, Li L, Gao L et al (2016) Effect of renal sympathetic denervation on hepatic glucose metabolism and blood pressure in a rat model of insulin resistance. J Hypertens 34(12):2465–2474
Guilherme A, Henriques F, Bedard AH, Czech MP (2019) Molecular pathways linking adipose innervation to insulin action in obesity and diabetes mellitus. Nat Rev Endocrinol 15(4):207–225
Underwood PC, Adler GK (2012) The renin angiotensin aldosterone system and insulin resistance in humans. Curr Hypertens Rep 15(1):59–70
Jamerson KA, Julius S, Gudbrandsson T, Andersson O, Brant DO (1993) Reflex sympathetic activation induces acute insulin resistance in the human forearm. Hypertension 21(5):618–623
Acknowledgement
The authors acknowledge Professor Chen for her promotion of renal denervation and related researches in our hospital.
Author information
Authors and Affiliations
Contributions
ZPZ and XPC were involved in the study design. ZPZ, SX, and KL were involved in the data collection and analysis. ZPZ was involved in the manuscript writing. KL and XPC were involved in the manuscript revision. All authors approved the final version of the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Human and animal rights
This article does not contain any studies with human participants or animals performed by any of the authors.
Ethical approval
For this type of study, no ethics approval was required.
Informed consent
For this type of study, informed consent was not required.
Additional information
Managed by Massimo Porta.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Zhang, Z., Liu, K., Xiao, S. et al. Effects of catheter-based renal denervation on glycemic control and lipid levels: a systematic review and meta-analysis. Acta Diabetol 58, 603–614 (2021). https://doi.org/10.1007/s00592-020-01659-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00592-020-01659-6