Abstract
Aims
The incidence of type 1 diabetes has increased over the last decades. The pathological pathway is not yet clear, even if genetic and environmental risk factors are known. An early diagnosis can avoid ketoacidosis and its complications. This work aims to discuss the determinants of both ketoacidosis at the onset and access by hospital emergency departments without a suspected diagnosis.
Methods
An observational bi-centric prospective study was conducted in Northern Italy, on a paediatric population including Italian and migrant patients at the diabetes onset. Seventy-four type 1 diabetes patients, both Italian and migrant, were included in the study. Anthropometric, socio-economic, behavioural, clinical data were collected, and microbiota analyses were performed using stool samples.
Results
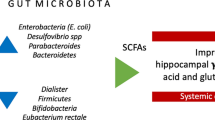
Regular physical activity is associated with lower ketoacidosis incidence at onset (OR 0.33 95% CI 0.12–0.95 p < 0.05), as is higher blood vitamin D level (OR 0.92 95% CI 0.85–0.99 p < 0.05). Moreover, a higher weaning age (OR 0.49 95% CI 0.27–0.89 p < 0.05), higher vitamin D level (OR 0.90 95% CI 0.83–0.98 p < 0.05) and a higher level of Akkermansia muciniphila (OR 0.46 95% CI 0.25–0.87 p < 0.05) are associated factors to lower frequency of type 1 diabetes onset without a suspected diagnosis. Diabetes migrant status is not a risk factor for severe type 1 diabetes onset; on the other hand, some protective factors are significantly more diffused among Italians, such as regular sport activity and non-critical vitamin D levels.
Conclusion
Behavioural and nutritional data, such as microbiota bio-indicators, seem to be useful to identify an at-risk population to prevent ketoacidosis and its severe complications.

Similar content being viewed by others
References
World Health Organization—Department for Management of Noncommunicable Diseases, Disability, Violence and Injury Prevention, Geneva, Switzerland (2019) Classification of diabetes mellitus 2019 https://www.who.int/health-topics/diabetes
National Center for Chronic Disease Prevention and Health Promotion (2017) National Diabetes Statistics Report, 2017. Estimates of diabetes and its burden in the United States.
Patterson CC, Harjutsalo V, Rosenbauer J et al (2019) Trends and cyclical variation in the incidence of childhood type 1 diabetes in 26 European centres in the 25 year period 1989–2013: a multicentre prospective registration study. Diabetologia 62:408–417. https://doi.org/10.1007/s00125-018-4763-3
Bruno G (2016) Il registro diabete Piemonte. Italian Health Policy Brief. Anno VI Speciale, pp. 1–8
Atkinson MA, Eisenbarth GS, Michels AW (2014) Type 1 diabetes. Lancet 383:69–82. https://doi.org/10.1016/S0140-6736(13)60591-7
Dedrick S, Sundaresh B, Huang Q et al (2020) The role of gut microbiota and environmental factors in type 1 diabetes pathogenesis. Front Endocrinol (Lausanne) 11:1–20. https://doi.org/10.3389/fendo.2020.00078
Knip M, Luopajärvi K, Härkönen T (2017) Early life origin of type 1 diabetes. Semin Immunopathol 39:653–667. https://doi.org/10.1007/s00281-017-0665-6
Park A, Zhao G (2018) Mining the Virome for Insights into Type 1 Diabetes. DNA Cell Biol 37:422–425. https://doi.org/10.1089/dna.2018.4185
Paschou SA, Papadopoulou-Marketou N, Chrousos GP, Kanaka-Gantenbein C (2018) On type 1 diabetes mellitus pathogenesis. Endocr Connect 7:R38–R46. https://doi.org/10.1530/EC-17-0347
Zipitis CS, Akobeng AK (2008) Vitamin D supplementation in early childhood and risk of type 1 diabetes: a systematic review and meta-analysis. Arch Dis Child 93:512–517
Feng R, Li Y, Li G et al (2015) Lower serum 25 (OH) D concentrations in type 1 diabetes: a meta-analysis. Diabetes Res Clin Pract 108:e71–e75. https://doi.org/10.1016/j.diabres.2014.12.008
Giri D, Pintus D, Burnside G et al (2017) Treating vitamin D deficiency in children with type 1 diabetes could improve their glycaemic control. BMC Res Notes. https://doi.org/10.1186/s13104-017-2794-3
Rewers M, Ludvigsson J (2016) Environmental risk factors for type 1 diabetes. Lancet 387:2340–2348
Bizzarri C, Pitocco D, Napoli N et al (2010) No protective effect of calcitriol on β-cell function in recent-onset type 1 diabetes: the IMDIAB XIII trial. Diabetes Care 33:1962–1963. https://doi.org/10.2337/dc10-0814
Cashen K, Petersen T (2019) Diabetic ketoacidosis. Pediatr Rev 40:412–420. https://doi.org/10.1542/pir.2018-0231
Rabbone I, Maltoni G, Tinti D et al (2019) Diabetic ketoacidosis at the onset of disease during a national awareness campaign: a 2-year observational study in children aged 0–18 years. Arch Dis Child. https://doi.org/10.1136/archdischild-2019-316903
World Health Organization (2018) Template for Parental Consent-clinical studies 20, avenue Appia – CH-1211 Geneva 27 – Switzerland. http://www.who.int/ethics/review-committee
Franzosa EA, Morgan XC, Segata N et al (2014) Relating the metatranscriptome and metagenome of the human gut. PNAS 111:E2329–E2338. https://doi.org/10.1073/pnas.1319284111
IHMS Consortium (2015) IHMS-SOP 02 V2: Standard Operating Procedure for Fecal Samples Self ‐ Collection Laboratory Analysis Handled Within 4 To 24 Hour. http://www.microbiome-standards.org/index.php?id=289
Wolfsdorf JI, Glaser N, Agus M et al (2018) ISPAD clinical practice consensus guidelines 2018: diabetic ketoacidosis and the hyperglycemic hyperosmolar state. Pediatr Diabetes 19:155–177. https://doi.org/10.1111/pedi.12701
Istituto Superiore di Sanità (2017) Sovrappeso e obesità infantile, i dati del Sistema di Sorveglianza OKkio alla Salute. http://www.salute.gov.it/portale/news/p3_2_1_1_1.jsp?lingua=italiano&menu=notizie&p=dalministero&id=2929
Verduci E, Radaelli G, Stival G et al (2007) Dietary macronutrient intake during the first 10 years of life in a cohort of Italian children. J Pediatr Gastroenterol Nutr 45:90–95. https://doi.org/10.1097/MPG.0b013e318058ca4e
Meek RL, LeBoeuf RC, Saha SA et al (2013) Glomerular cell death and inflammation with high-protein diet and diabetes. Nephrol Dial Transplant 28:1711–1720. https://doi.org/10.1093/ndt/gfs579
Gunn ER, Albert BB, Hofman PL et al (2017) Pathways to reduce diabetic ketoacidosis with new onset type 1 diabetes: evidence from a regional pediatric diabetes center: Auckland, New Zealand, 2010 to 2014. Pediatr Diabetes 18:553–558. https://doi.org/10.1111/pedi.12456
Borgo F, Verduci E, Riva A et al (2017) Relative abundance in bacterial and fungal gut microbes in obese children: a case control study. Child Obes 13:78–84. https://doi.org/10.1089/chi.2015.0194
Szypowska A, Dżygało K, Wysocka-Mincewicz M et al (2017) High incidence of diabetic ketoacidosis at diagnosis of type 1 diabetes among polish children aged 10–12 and under 5 years of age: a multicenter study. Pediatr Diabetes 18:722–728. https://doi.org/10.1111/pedi.12446
Lee HJ, Yu HW, Jung HW et al (2017) Factors associated with the presence and severity of diabetic ketoacidosis at diagnosis of type 1 diabetes in korean children and adolescents. @bull J Korean Med Sci 32:303–309. https://doi.org/10.3346/jkms.2017.32.2.303
Cherubini V, Skrami E, Ferrito L et al (2019) Exercise has the guts: How physical activity may positively modulate gut microbiota in chronic and immune-based diseases. Dig Liver Dis 62:1–4. https://doi.org/10.1007/s00125-018-4763-3
Vecchio F, Lo BN, Stabilini A et al (2018) Abnormal neutrophil signature in the blood and pancreas of presymptomatic and symptomatic type 1 diabetes federica vecchio, …, the type 1 diabetes trialnet study group, manuela battaglia find the latest version : abnormal neutrophil signature in the bl. JCI Insight 3:1–17. https://doi.org/10.1172/JCI.INSIGHT.122146
Muyzer G, Waal ECDE, Uitierlinden AG (1993) Profiling of complex microbial populations by denaturing gradient gel electrophoresis analysis of polymerase chain reaction-amplified genes coding for 16S rRNA. Applaied Environ Microbiol 59:695–700
Vanhoutte T, Huys G, De Brandt E, Swings J (2004) Temporal stability analysis of the microbiota in human feces by denaturing gradient gel electrophoresis using universal and group-specific 16S rRNA gene primers. FEMS Microbiol Ecol 48:437–446. https://doi.org/10.1016/j.femsec.2004.03.001
Joossens M, Huys G, Van Steen K et al (2011) High-throughput method for comparative analysis of denaturing gradient gel electrophoresis profiles from human fecal samples reveals significant increases in two bifidobacterial species after inulin-type prebiotic intake. FEMS Microbiol Ecol 75:343–349. https://doi.org/10.1111/j.1574-6941.2010.01008.x
De Goffau MC, Luopajärvi K, Knip M et al (2013) Fecal microbiota composition differs between children with beta-cell autoimmunity and those without. Diabetes 62:1238–1244. https://doi.org/10.2337/db12-0526
Davis-Richardson AG, Ardissone AN, Dias R et al (2014) Bacteroides dorei dominates gut microbiome prior to autoimmunity in Finnish children at high risk for type 1 diabetes. Front Microbiol 5:1–11. https://doi.org/10.3389/fmicb.2014.00678
Brown CT, Davis-Richardson AG, Giongo A et al (2011) Gut microbiome metagenomics analysis suggests a functional model for the development of autoimmunity for type 1 diabetes. PLoS ONE 6:1–9. https://doi.org/10.1371/journal.pone.0025792
Ottman N, Reunanen J, Meijerink M et al (2017) Pili-like proteins of Akkermansia muciniphila modulate host immune responses and gut barrier function. PLoS ONE 12:1–18. https://doi.org/10.1371/journal.pone.0173004
Cani PD (2018) Severe obesity and gut microbiota: does bariatric surgery really reset the system? Gut gutjnl-2018-316815. https://doi.org/10.1136/gutjnl-2018-316815
Knight R, Vrbanac A, Taylor BC et al (2018) Best practices for analysing microbiomes. Nat Rev Microbiol. https://doi.org/10.1038/s41579-018-0029-9
Webster NS, Negri AP (2006) Site-specific variation in Antarctic marine biofilms established on artificial surfaces. Environ Microbiol 8:1177–1190. https://doi.org/10.1111/j.1462-2920.2006.01007.x
O’Sullivan LA, Webster G, Fry JC et al (2008) Modified linker-PCR primers facilitate complete sequencing of DGGE DNA fragments. J Microbiol Methods 75:579–581. https://doi.org/10.1016/j.mimet.2008.08.006
Dridi B, Henry M, El Khéchine A et al (2009) High prevalence of Methanobrevibacter smithii and Methanosphaera stadtmanae detected in the human gut using an improved DNA detection protocol. PLoS ONE 4:e7063. https://doi.org/10.1371/journal.pone.0007063
Guo X, Xia X, Tang R et al (2008) Development of a real-time PCR method for Firmicutes and Bacteroidetes in faeces and its application to quantify intestinal population of obese and lean pigs. Lett Appl Microbiol 47:367–373. https://doi.org/10.1111/j.1472-765X.2008.02408.x
Murri M, Leiva I, Gomez-zumaquero JM, et al (2013) Gut microbiota in children with type 1 diabetes differs from that in healthy children: a case-control study. BMC Med 11:46. http://www.biomedcentral.com/1741-7015/11/46
Nakayama T, Oishi K (2013) Influence of coffee (Coffea arabica) and galacto-oligosaccharide consumption on intestinal microbiota and the host responses. FEMS microbial lett 343(2):161–168
Matsuki T, Watanabe K, Fujimoto J et al (2004) Quantitative pcr with 16s primers for analysis of human intestinal bifidobacteria. Appl Environ Microbiol 70:167–173. https://doi.org/10.1128/AEM.70.1.167
Dao MC, Everard A, Aron-wisnewsky J et al (2016) Akkermansia muciniphila and improved metabolic health during a dietary intervention in obesity : relationship with gut microbiome richness and ecology. Gut microbiota 65:426–436. https://doi.org/10.1136/gutjnl-2014-308778
Johnston C, Ufnar JA, Griffith JF et al (2010) A real-time qPCR assay for the detection of the nifH gene of Methanobrevibacter smithii, a potential indicator of sewage pollution. J Appl Microbiol 109:1946–1956. https://doi.org/10.1111/j.1365-2672.2010.04824.x
Acknowledgements
The authors wish to thank the Italian Ministry of Health for funding (RF-2011-02350617), Dr. Barbara Di Stefano (Sanitary Direction AOU Novara), as well as Mrs. Rim Maatoug, Mrs. Shpresa Xheka and Mrs. Daniela Elena Zelinschi, cultural intermediaries at the Novara Hospital, for the translation of the questionnaire for migrant people. Finally, the authors especially acknowledge the participating children and their families.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
This article belongs to the topical collection Gut Microbiome and Metabolic Disorders, managed by Massimo Federici.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Rabbone, I., Traversi, D., Scaioli, G. et al. Microbiota, epidemiological and nutritional factors related to ketoacidosis at the onset of type 1 diabetes. Acta Diabetol 57, 1337–1349 (2020). https://doi.org/10.1007/s00592-020-01555-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00592-020-01555-z