Abstract
Aims
Early studies have identified type 1 diabetes mellitus (T1DM) as a disease that is caused by the autoimmune destruction of the insulin-producing pancreatic β-cells. Genetics, environment and the immune pathogenesis of T1DM are three major pillars of T1DM research. We try to understand the changes in the gene expression profile during the pathogenesis of T1DM.
Methods
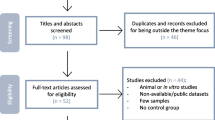
We performed a systematic search in the Gene Expression Omnibus (GEO) database for microarray studies of T1DM with samples taken at or before the T1DM onset.
Results
The results of an integrated analysis of different GEO datasets and a comparison of the gene expression level in T1DM samples taken at the time of appearance of the islet autoantibodies, 1 year before T1DM onset, and at the time of T1DM onset showed that CD274, which encodes PD-L1, was up-regulated in the newly onset T1DM samples. CD274 had a stable expression level in the control samples but showed a gradual up-regulation from the appearance of autoantibodies to the onset of T1DM.
Conclusions
These results indicate that CD274 up-regulation in T1DM is correlated with disease pathogenesis. PD-L1 might play a protective role in preventing the pancreatic islets from autoimmune destruction, which may help researchers find strategies for preventing the destruction process of pancreas β-cells in T1DM.





Similar content being viewed by others
References
Bottazzo GF, Florin-Christensen A, Doniach D (1974) Islet-cell antibodies in diabetes mellitus with autoimmune polyendocrine deficiencies. Lancet 2:1279–1283
Atkinson MA, Eisenbarth GS (2001) Type 1 diabetes: new perspectives on disease pathogenesis and treatment. Lancet 358:221–229. doi:10.1016/S0140-6736(01)05415-0
Redondo MJ, Fain PR, Eisenbarth GS (2001) Genetics of type 1A diabetes. Recent Progr Horm Res 56:69–89
Wen L et al (2008) Innate immunity and intestinal microbiota in the development of type 1 diabetes. Nature 455:1109–1113. doi:10.1038/nature07336
Knip M, Virtanen SM, Akerblom HK (2010) Infant feeding and the risk of type 1 diabetes. Am J Clin Nutr 91:1506S–1513S. doi:10.3945/ajcn.2010.28701C
Hyoty H, Taylor KW (2002) The role of viruses in human diabetes. Diabetologia 45:1353–1361. doi:10.1007/s00125-002-0852-3
Baekkeskov S et al (1990) Identification of the 64 K autoantigen in insulin-dependent diabetes as the GABA-synthesizing enzyme glutamic acid decarboxylase. Nature 347:151–156. doi:10.1038/347151a0
Kaufman DL et al (1992) Autoimmunity to two forms of glutamate decarboxylase in insulin-dependent diabetes mellitus. J Clin Invest 89:283–292. doi:10.1172/JCI115573
Palmer JP et al (1983) Insulin antibodies in insulin-dependent diabetics before insulin treatment. Science 222:1337–1339
Passini N et al (1995) The 37/40-kilodalton autoantigen in insulin-dependent diabetes mellitus is the putative tyrosine phosphatase IA-2. Proc Natl Acad Sci USA 92:9412–9416
Wenzlau JM et al (2007) The cation efflux transporter ZnT8 (Slc30A8) is a major autoantigen in human type 1 diabetes. Proc Natl Acad Sci USA 104:17040–17045. doi:10.1073/pnas.0705894104
Pesenacker AM et al (2016) A regulatory T-Cell gene signature is a specific and sensitive biomarker to identify children with new-onset type 1 diabetes. Diabetes 65:1031–1039. doi:10.2337/db15-0572
Barrett T et al (2013) NCBI GEO: archive for functional genomics data sets–update. Nucleic Acids Res 41:D991–D995. doi:10.1093/nar/gks1193
Kallionpaa H et al (2014) Innate immune activity is detected prior to seroconversion in children with HLA-conferred type 1 diabetes susceptibility. Diabetes 63:2402–2414. doi:10.2337/db13-1775
Kaizer EC et al (2007) Gene expression in peripheral blood mononuclear cells from children with diabetes. J Clin Endocrinol Metab 92:3705–3711. doi:10.1210/jc.2007-0979
Dennis G Jr et al (2003) DAVID: database for annotation, visualization, and integrated discovery. Genome Biol 4:P3
Szklarczyk D et al (2015) STRING v10: protein-protein interaction networks, integrated over the tree of life. Nucleic Acids Res 43:D447–D452. doi:10.1093/nar/gku1003
Barthson J et al (2011) Cytokines tumor necrosis factor-alpha and interferon-gamma induce pancreatic beta-cell apoptosis through STAT1-mediated Bim protein activation. J Biol Chem 286:39632–39643. doi:10.1074/jbc.M111.253591
Santin I et al (2011) PTPN2, a candidate gene for type 1 diabetes, modulates pancreatic beta-cell apoptosis via regulation of the BH3-only protein Bim. Diabetes 60:3279–3288. doi:10.2337/db11-0758
Arafat HA et al (2007) Osteopontin protects the islets and beta-cells from interleukin-1 beta-mediated cytotoxicity through negative feedback regulation of nitric oxide. Endocrinology 148:575–584. doi:10.1210/en.2006-0970
Kase S et al (2007) Increased osteopontin levels in the vitreous of patients with diabetic retinopathy. Ophthalmic Res 39:143–147. doi:10.1159/000102936
Ashkar S et al (2000) Eta-1 (osteopontin): an early component of type-1 (cell-mediated) immunity. Science 287:860–864
Lampe MA, Patarca R, Iregui MV, Cantor H (1991) Polyclonal B cell activation by the Eta-1 cytokine and the development of systemic autoimmune disease. J Immunol 147:2902–2906
Marciano R et al (2009) Association of alleles at polymorphic sites in the Osteopontin encoding gene in young type 1 diabetic patients. Clin Immunol 131:84–91. doi:10.1016/j.clim.2008.11.004
Welzen-Coppens JM, van Helden-Meeuwsen CG, Leenen PJ, Drexhage HA, Versnel MA (2012) Reduced numbers of dendritic cells with a tolerogenic phenotype in the prediabetic pancreas of NOD mice. J Leukoc Biol 92:1207–1213. doi:10.1189/jlb.0312168
Plato A, Willment JA, Brown GD (2013) C-type lectin-like receptors of the dectin-1 cluster: ligands and signaling pathways. Int Rev Immunol 32:134–156. doi:10.3109/08830185.2013.777065
Francisco LM, Sage PT, Sharpe AH (2010) The PD-1 pathway in tolerance and autoimmunity. Immunol Rev 236:219–242. doi:10.1111/j.1600-065X.2010.00923.x
Zhang X et al (2004) Structural and functional analysis of the costimulatory receptor programmed death-1. Immunity 20:337–347
Sharpe AH, Wherry EJ, Ahmed R, Freeman GJ (2007) The function of programmed cell death 1 and its ligands in regulating autoimmunity and infection. Nat Immunol 8:239–245. doi:10.1038/ni1443
Sharpe AH, Abbas AK (2006) T-cell costimulation–biology, therapeutic potential, and challenges. N Engl J Med 355:973–975. doi:10.1056/NEJMp068087
Bullens DM et al (2001) Effects of co-stimulation by CD58 on human T cell cytokine production: a selective cytokine pattern with induction of high IL-10 production. Int Immunol 13:181–191
Bluestone JA, Herold K, Eisenbarth G (2010) Genetics, pathogenesis and clinical interventions in type 1 diabetes. Nature 464:1293–1300
Keir ME et al (2006) Tissue expression of PD-L1 mediates peripheral T cell tolerance. J Exp Med 203:883–895. doi:10.1084/jem.20051776
Ansari MJ et al (2003) The programmed death-1 (PD-1) pathway regulates autoimmune diabetes in nonobese diabetic (NOD) mice. J Exp Med 198:63–69. doi:10.1084/jem.20022125
Fiorina P et al (2011) Targeting the CXCR4-CXCL12 axis mobilizes autologous hematopoietic stem cells and prolongs islet allograft survival via programmed death ligand 1. J Immunol 186:121–131. doi:10.4049/jimmunol.1000799
Okamoto M et al (2016) Fulminant type 1 diabetes mellitus with anti-programmed cell death-1 therapy. J Diab Invest. doi:10.1111/jdi.12531
Martin-Liberal J et al (2015) Anti-programmed cell death-1 therapy and insulin-dependent diabetes: a case report. Cancer Immunol Immunother CII 64:765–767. doi:10.1007/s00262-015-1689-1
Mellati M et al (2015) Anti-PD-1 and Anti-PDL-1 monoclonal antibodies causing type 1 Diabetes. Diab care 38:e137–e138. doi:10.2337/dc15-0889
Guleria I et al (2007) Mechanisms of PDL1-mediated regulation of autoimmune diabetes. Clin Immunol 125:16–25. doi:10.1016/j.clim.2007.05.013
Gianchecchi E, Delfino DV, Fierabracci A (2013) Recent insights into the role of the PD-1/PD-L1 pathway in immunological tolerance and autoimmunity. Autoimmun Rev 12:1091–1100. doi:10.1016/j.autrev.2013.05.003
Acknowledgements
This study was supported by grants from the National Natural Science Foundation of China (81471041, to Ji Hu), the National Natural Science Foundation of China (8160030570, to Chen Fang) and the National Natural Science Foundation of China (81502865, to Yun Huang).
Author information
Authors and Affiliations
Contributions
Author Contributions
C. F wrote the manuscript, Y. F. P and Y. H. searched the library and reviewed all of the articles. H. H. Z. and X. H. C. extracted the data, H. M. G. and S. C. L. prepared the tables and X. Y. J. and J. H. prepared figures. All authors reviewed the manuscript.
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical standard
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Statement of human and animal rights
This article does not contain any studies with human or animal subjects performed by the any of the authors.
Informed consent
Informed consent was obtained from all patients for being included in the study.
Additional information
Managed by Massimo Federici.
Chen Fang and Yun Huang are co-first authors.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Fang, C., Huang, Y., Pei, Y. et al. Genome-wide gene expression profiling reveals that CD274 is up-regulated new-onset type 1 diabetes mellitus. Acta Diabetol 54, 757–767 (2017). https://doi.org/10.1007/s00592-017-1005-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00592-017-1005-y