Abstract
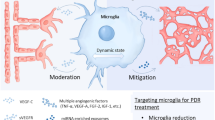
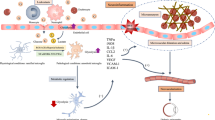
During last decades, the diagnosis of diabetes has been associated with several chronic complications such as diabetic retinopathy (DR). Recent studies of DR have revealed an inflammatory component, which precedes the detection of alterations in the visual function. During DR, the inflammatory process presents two opposite roles depending on the polarization of resident immune cells of the retina triggering proinflammatory (M1) or antiinflammatory (M2) actions. In an early stage of DR, the M2 response concurs with the M1 and is able to ameliorate inflammation and delay the progression of the disease. However, during the progression of DR, the M1 response is maintained whereas the M2 declines and, in this scenario, the classical proinflammatory signaling pathways are chronically activated leading to retinal neurodegeneration and the loss of visual function. The M1/M2 responses are closely related to the activation and polarization of microglial cells. This review aims to offer an overview of the recent insights into the role of microglial cells during inflammation in DR. We have focused on the possibility of modulating microglia polarization as a new therapeutic strategy in DR treatments.

Similar content being viewed by others
References
Bourne RR et al (2013) Causes of vision loss worldwide, 1990–2010: a systematic analysis. Lancet Glob Health 1(6):e339–e349
Romero-Aroca P et al (2016) Changes observed in diabetic retinopathy: eight-year follow-up of a Spanish population. Br J Ophthalmol 100(10):1366–1371
Cheung N, Mitchell P, Wong TY (2010) Diabetic retinopathy. Lancet 376(9735):124–136
Nentwich MM, Ulbig MW (2015) Diabetic retinopathy—ocular complications of diabetes mellitus. World J Diabetes 6(3):489–499
Oellers P, Mahmoud TH (2016) Surgery for proliferative diabetic retinopathy: new tips and tricks. J Ophthalmic Vis Res 11(1):93–99
Porta M, Bandello F (2002) Diabetic retinopathy A clinical update. Diabetologia 45(12):1617–1634
Aiello LP, D.E.R. Group (2014) Diabetic retinopathy and other ocular findings in the diabetes control and complications trial/epidemiology of diabetes interventions and complications study. Diabetes Care 37(1):17–23
Antonetti DA, Klein R, Gardner TW (2012) Diabetic retinopathy. N Engl J Med 366(13):1227–1239
van Dijk HW et al (2012) Early neurodegeneration in the retina of type 2 diabetic patients. Invest Ophthalmol Vis Sci 53(6):2715–2719
Simo R, Hernandez C, European R (2014) Consortium for the early treatment of diabetic, neurodegeneration in the diabetic eye: new insights and therapeutic perspectives. Trends Endocrinol Metab 25(1):23–33
Crosby-Nwaobi R et al (2015) cross talk between lipid metabolism and inflammatory markers in patients with diabetic retinopathy. J Diabetes Res 2015:191382
Kowluru RA, Mishra M (2015) Contribution of epigenetics in diabetic retinopathy. Sci China Life Sci 58(6):556–563
Gardner TW et al (2011) An integrated approach to diabetic retinopathy research. Arch Ophthalmol 129(2):230–235
Anand-Apte B et al (2010) Betacellulin induces increased retinal vascular permeability in mice. PLoS ONE 5(10):e13444
Powell ED, Field RA (1964) Diabetic retinopathy and rheumatoid arthritis. Lancet 2(7349):17–18
Brucklacher RM et al (2008) Whole genome assessment of the retinal response to diabetes reveals a progressive neurovascular inflammatory response. BMC Med Genomics 1:26
Joussen AM et al (2001) Leukocyte-mediated endothelial cell injury and death in the diabetic retina. Am J Pathol 158(1):147–152
Joussen AM et al (2004) A central role for inflammation in the pathogenesis of diabetic retinopathy. FASEB J 18(12):1450–1452
Silva PS, Sun JK, Aiello LP (2009) Role of steroids in the management of diabetic macular edema and proliferative diabetic retinopathy. Semin Ophthalmol 24(2):93–99
Vujosevic S et al (2016) Proteome analysis of retinal glia cells-related inflammatory cytokines in the aqueous humour of diabetic patients. Acta Ophthalmol 94(1):56–64
Kettenmann H et al (2011) Physiology of microglia. Physiol Rev 91(2):461–553
Davalos D et al (2005) ATP mediates rapid microglial response to local brain injury in vivo. Nat Neurosci 8(6):752–758
Lynch MA (2009) The multifaceted profile of activated microglia. Mol Neurobiol 40(2):139–156
Salter MW, Beggs S (2014) Sublime microglia: expanding roles for the guardians of the CNS. Cell 158(1):15–24
Arroba AI et al (2016) Modulation of microglia polarization dynamics during diabetic retinopathy in db/db mice. Biochim Biophys Acta 1862(9):1663–1674
Su F et al (2015) Fluoxetine and S-citalopram inhibit M1 activation and promote M2 activation of microglia in vitro. Neuroscience 294:60–68
Lampron A, Elali A, Rivest S (2013) Innate immunity in the CNS: redefining the relationship between the CNS and Its environment. Neuron 78(2):214–232
Martinez FO, Helming L, Gordon S (2009) Alternative activation of macrophages: an immunologic functional perspective. Annu Rev Immunol 27:451–483
Ajami B et al (2011) Infiltrating monocytes trigger EAE progression, but do not contribute to the resident microglia pool. Nat Neurosci 14(9):1142–1149
Higuchi M et al (2011) Alzheimer’s disease and microglia. Rinsho Shinkeigaku 51(11):1031
Ouchi Y et al (2005) Microglial activation and dopamine terminal loss in early Parkinson’s disease. Ann Neurol 57(2):168–175
Bosco A et al (2015) Neurodegeneration severity can be predicted from early microglia alterations monitored in vivo in a mouse model of chronic glaucoma. Dis Model Mech 8(5):443–455
Vujosevic S et al (2013) Hyperreflective intraretinal spots in diabetics without and with nonproliferative diabetic retinopathy: an in vivo study using spectral domain OCT. J Diabetes Res 2013:491835
Robinson R et al (2012) Update on animal models of diabetic retinopathy: from molecular approaches to mice and higher mammals. Dis Model Mech 5(4):444–456
Bogdanov P et al (2014) The db/db mouse: a useful model for the study of diabetic retinal neurodegeneration. PLoS ONE 9(5):e97302
Holopigian K et al (1992) A comparison of photopic and scotopic electroretinographic changes in early diabetic retinopathy. Invest Ophthalmol Vis Sci 33(10):2773–2780
Shirao Y, Kawasaki K (1998) Electrical responses from diabetic retina. Prog Retin Eye Res 17(1):59–76
Kinoshita J et al (2015) Sildenafil-induced reversible impairment of rod and cone phototransduction in monkeys. Invest Ophthalmol Vis Sci 56(1):664–673
Kline RP, Ripps H, Dowling JE (1978) Generation of b-wave currents in the skate retina. Proc Natl Acad Sci USA 75(11):5727–5731
Klemp K et al (2005) The multifocal ERG in diabetic patients without retinopathy during euglycemic clamping. Invest Ophthalmol Vis Sci 46(7):2620–2626
Kahn SE et al (2006) Obesity is a major determinant of the association of C-reactive protein levels and the metabolic syndrome in type 2 diabetes. Diabetes 55(8):2357–2364
Kim CS et al (2006) Circulating levels of MCP-1 and IL-8 are elevated in human obese subjects and associated with obesity-related parameters. Int J Obes (Lond) 30(9):1347–1355
Bruewer M et al (2003) Proinflammatory cytokines disrupt epithelial barrier function by apoptosis-independent mechanisms. J Immunol 171(11):6164–6172
Brun P et al (2007) Increased intestinal permeability in obese mice: new evidence in the pathogenesis of nonalcoholic steatohepatitis. Am J Physiol Gastrointest Liver Physiol 292(2):G518–G525
Wada T et al (2013) Cilostazol ameliorates systemic insulin resistance in diabetic db/db mice by suppressing chronic inflammation in adipose tissue via modulation of both adipocyte and macrophage functions. Eur J Pharmacol 707(1–3):120–129
Hotamisligil GS, Erbay E (2008) Nutrient sensing and inflammation in metabolic diseases. Nat Rev Immunol 8(12):923–934
Xu Y et al (2015) Decreased expression levels of Nurr1 are associated with chronic inflammation in patients with type 2 diabetes. Mol Med Rep 12(4):5487–5493
Dong N et al (2014) Retinal neuronal MCP-1 induced by AGEs stimulates TNF-alpha expression in rat microglia via p38, ERK, and NF-kappaB pathways. Mol Vis 20:616–628
Legacy J et al (2013) Granulocyte macrophage colony-stimulating factor promotes regeneration of retinal ganglion cells in vitro through a mammalian target of rapamycin-dependent mechanism. J Neurosci Res 91(6):771–779
Sappington RM, Chan M, Calkins DJ (2006) Interleukin-6 protects retinal ganglion cells from pressure-induced death. Invest Ophthalmol Vis Sci 47(7):2932–2942
Li Q et al (2002) Early retinal damage in experimental diabetes: electroretinographical and morphological observations. Exp Eye Res 74(5):615–625
Engerman RL, Kern TS (1995) Retinopathy in animal models of diabetes. Diabetes Metab Rev 11(2):109–120
Obrosova IG et al (2003) Aldose reductase inhibitor fidarestat prevents retinal oxidative stress and vascular endothelial growth factor overexpression in streptozotocin-diabetic rats. Diabetes 52(3):864–871
Kato N et al (2003) Long-term treatment with fidarestat suppresses the development of diabetic retinopathy in STZ-induced diabetic rats. J Diabetes Complications 17(6):374–379
Hernandez C et al (2016) Topical administration of GLP-1 receptor agonists prevents retinal neurodegeneration in experimental diabetes. Diabetes 65(1):172–187
Park KW et al (2007) Interleukin-10 endogenously expressed in microglia prevents lipopolysaccharide-induced neurodegeneration in the rat cerebral cortex in vivo. Exp Mol Med 39(6):812–819
Du L et al (2016) Role of microglia in neurological disorders and their potentials as a therapeutic target. Mol Neurobiol. doi:10.1007/s12035-016-0245-0
Jiao H et al (2015) Spatiotemporal cadence of macrophage polarisation in a model of light-induced retinal degeneration. PLoS ONE 10(12):e0143952
Arroba AI et al (2011) Microglia-mediated IGF-I neuroprotection in the rd10 mouse model of retinitis pigmentosa. Invest Ophthalmol Vis Sci 52(12):9124–9130
Block ML, Zecca L, Hong JS (2007) Microglia-mediated neurotoxicity: uncovering the molecular mechanisms. Nat Rev Neurosci 8(1):57–69
O’Koren EG, Mathew R, Saban DR (2016) Fate mapping reveals that microglia and recruited monocyte-derived macrophages are definitively distinguishable by phenotype in the retina. Sci Rep 6:20636
Wang Y et al (2012) Autoantibody against transient receptor potential M1 cation channels of retinal ON bipolar cells in paraneoplastic vitelliform retinopathy. BMC Ophthalmol 12:56
Cai Y et al (2016) Procyanidins alleviates morphine tolerance by inhibiting activation of NLRP3 inflammasome in microglia. J Neuroinflammation 13(1):53
Mittal K, Katare DP (2016) Shared links between type 2 diabetes mellitus and Alzheimer’s disease: a review. Diabetes Metab Syndr 10(2 Suppl 1):S144–S149
Verdile G et al (2015) Inflammation and oxidative stress: the molecular connectivity between insulin resistance, obesity, and Alzheimer’s disease. Mediators Inflamm 2015:105828
Lim JK et al (2016) The eye as a biomarker for Alzheimer’s disease. Front Neurosci 10:536
Reed BT, Behar-Cohen F, Krantic S (2016) Seeing early signs of Alzheimer’s disease through the lens of the eye. Curr Alzheimer Res 14(1):6–17
Serini S, Calviello G (2016) Reduction of oxidative/nitrosative stress in brain and its involvement in the neuroprotective effect of n-3 PUFA in Alzheimer’s disease. Curr Alzheimer Res 13(2):123–134
Roche SL et al (2016) Progesterone attenuates microglial-driven retinal degeneration and stimulates protective fractalkine-CX3CR1 signaling. PLoS ONE 11(11):e0165197
Acknowledgements
The research of A.M.V. laboratory has been supported by the Spanish Ministry of Economy and Competitivity (Grant SAF2015-65267-R MINECO/FEDER cofounded by the Fondos Europeos de Desarrollo Regional de la Unión Europea), Instituto de Salud Carlos III (Grant INFLAMES-PIE14/00045 and CIBERdem), and European Union (FP7 HEALTH.2011.2.4.3.1, EUROCONDOR Grant Agreement Number 278040). We acknowledge all EUROCONDOR partners for support and helpful discussions.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
None.
Ethical approval
All applicable international, national, and/or institutional guidelines for the care and use of animals were followed.
Human and animal rights disclosure
All animal procedures were performed according to the Ethical standards of the institutional research committee and with the 1964 Declaration of Helsinki and its later amendments or comparable ethical standards.
Informed consent
Informed consent was obtained from all participants included in this study.
Additional information
Managed By Massimo Porta.
Rights and permissions
About this article
Cite this article
Arroba, A.I., Valverde, Á.M. Modulation of microglia in the retina: new insights into diabetic retinopathy. Acta Diabetol 54, 527–533 (2017). https://doi.org/10.1007/s00592-017-0984-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00592-017-0984-z