Abstract
Aims
Fatty acid-binding protein 4 (FABP4) is mainly expressed in adipocytes and macrophages and is demonstrated to be elevated in diabetes patients. The aim of this study was to evaluate the possible role of FABP4 in the diagnosis of GDM and to investigate the relationship between FABP4 and overweight, insulin resistance and inflammatory marker TNF-α.
Methods
A total of 46 women with GDM and 55 age-matched pregnant women without GDM (non-GDM) were eligible for the study. Demographic and biochemical parameters and fasting venous blood samples of two groups were collected from all cases. Serum concentrations of FABP4 were determined using enzyme-linked immunosorbent assay (ELISA). The predictive value of Serum FABP4 level was evaluated using receiver operating characteristic curve (ROC curve) analysis.
Results
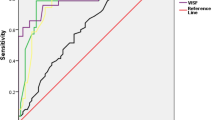
We found that the serum FABP4 levels were significantly higher in GDM compared to the non-GDM group. The area under the ROC curve assay yielded a satisfactory result of 0.94 (95 % confidence interval 0.90–0.98; p < 0.001). The best compromise between 86.96 % specificity and 89.09 % sensitivity was obtained with a cutoff value of 1.96 ng/mL for GDM diagnosis. Moreover, a significant positive correlation was observed between FABP4 and overweight, insulin resistance and TNF-α in pregnant women with GDM.
Conclusions
These results suggest that serum FABP4 may potentially serve as a novel biomarker for the prediction of GDM.



Similar content being viewed by others
Change history
18 July 2018
The title “Plasma fatty acid-binding protein 4 (FABP4) as a novel biomarker to predict gestational diabetes mellitus” should be replaced by “Serum fatty acid-binding protein 4 (FABP4) as a novel biomarker to predict gestational diabetes mellitus”.
References
Group HSCR, Metzger BE, Lowe LP et al (2008) Hyperglycemia and adverse pregnancy outcomes. N Engl J Med 358:1991–2002. doi:10.1056/NEJMoa0707943
Ben-Haroush A, Yogev Y, Hod M (2004) Epidemiology of gestational diabetes mellitus and its association with type 2 diabetes. Diabet Med 21:103–113
El-Chaar D, Finkelstein SA, Tu X et al (2013) The impact of increasing obesity class on obstetrical outcomes. J Obstet Gynaecol Can 35:224–233
Kim SY, Saraiva C, Curtis M, Wilson HG, Troyan J, Sharma AJ (2013) Fraction of gestational diabetes mellitus attributable to overweight and obesity by race/ethnicity, California, 2007–2009. Am J Public Health 103:e65–e72. doi:10.2105/AJPH.2013.301469
Boney CM, Verma A, Tucker R, Vohr BR (2005) Metabolic syndrome in childhood: association with birth weight, maternal obesity, and gestational diabetes mellitus. Pediatrics 115:e290–e296. doi:10.1542/peds.2004-1808
Crowther CA, Hiller JE, Moss JR (2005) Effect of treatment of gestational diabetes mellitus on pregnancy outcomes. N Engl J Med 352:2477–2486. doi:10.1056/NEJMoa042973
Yogev Y, Chen R, Hod M et al (2010) Hyperglycemia and adverse pregnancy outcome (HAPO) study: preeclampsia. Am J Obstet Gynecol 202(255):e251–e257. doi:10.1016/j.ajog.2010.01.024
Fang P, Shi M, Yu M, Guo L, Bo P, Zhang Z (2014) Endogenous peptides as risk markers to assess the development of insulin resistance. Peptides 51:9–14. doi:10.1016/j.peptides.2013.10.025
Hunt CR, Ro JH, Dobson DE, Min HY, Spiegelman BM (1986) Adipocyte P2 gene: developmental expression and homology of 5′-flanking sequences among fat cell-specific genes. Proc Natl Acad Sci USA 83:3786–3790
Spiegelman BM, Frank M, Green H (1983) Molecular cloning of mRNA from 3T3 adipocytes. Regulation of mRNA content for glycerophosphate dehydrogenase and other differentiation-dependent proteins during adipocyte development. J Biol Chem 258:10083–10089
Baxa CA, Sha RS, Buelt MK et al (1989) Human adipocyte lipid-binding protein: purification of the protein and cloning of its complementary DNA. Biochemistry 28:8683–8690
Amri EZ, Bertrand B, Ailhaud G, Grimaldi P (1991) Regulation of adipose cell differentiation I. Fatty acids are inducers of the aP2 gene expression. J Lipid Res 32:1449–1456
Melki SA, Abumrad NA (1993) Expression of the adipocyte fatty acid-binding protein in streptozotocin-diabetes: effects of insulin deficiency and supplementation. J Lipid Res 34:1527–1534
Xu A, Wang Y, Xu JY et al (2006) Adipocyte fatty acid-binding protein is a plasma biomarker closely associated with obesity and metabolic syndrome. Clin Chem 52:405–413. doi:10.1373/clinchem.2005.062463
Xu A, Tso AW, Cheung BM et al (2007) Circulating adipocyte-fatty acid binding protein levels predict the development of the metabolic syndrome: a 5-year prospective study. Circulation 115:1537–1543. doi:10.1161/CIRCULATIONAHA.106.647503
Tso AW, Xu A, Sham PC et al (2007) Serum adipocyte fatty acid binding protein as a new biomarker predicting the development of type 2 diabetes: a 10-year prospective study in a Chinese cohort. Diabetes Care 30:2667–2672. doi:10.2337/dc07-0413
Ota H, Furuhashi M, Ishimura S et al (2012) Elevation of fatty acid-binding protein 4 is predisposed by family history of hypertension and contributes to blood pressure elevation. Am J Hypertens 25:1124–1130. doi:10.1038/ajh.2012.88
Baessler A, Lamounier-Zepter V, Fenk S et al (2014) Adipocyte fatty acid-binding protein levels are associated with left ventricular diastolic dysfunction in morbidly obese subjects. Nutr Diabetes 4:e106. doi:10.1038/nutd.2014.3
Fuseya T, Furuhashi M, Yuda S et al (2014) Elevation of circulating fatty acid-binding protein 4 is independently associated with left ventricular diastolic dysfunction in a general population. Cardiovasc Diabetol 13:126. doi:10.1186/s12933-014-0126-7
Reiser H, Klingenberg R, Hof D et al (2015) Circulating FABP4 is a prognostic biomarker in patients with acute coronary syndrome but not in asymptomatic individuals. Arterioscler Thromb Vasc Biol 35:1872–1879. doi:10.1161/ATVBAHA.115.305365
Parra S, Cabre A, Marimon F et al (2014) Circulating FABP4 is a marker of metabolic and cardiovascular risk in SLE patients. Lupus 23:245–254. doi:10.1177/0961203313517405
Djousse L, Gaziano JM (2012) Plasma levels of FABP4, but not FABP3, are associated with increased risk of diabetes. Lipids 47:757–762. doi:10.1007/s11745-012-3689-7
Li YY, Xiao R, Li CP, Huangfu J, Mao JF (2015) Increased plasma levels of FABP4 and PTEN is associated with more severe insulin resistance in women with gestational diabetes mellitus. Med Sci Monit 21:426–431. doi:10.12659/MSM.892431
International Association of D, Pregnancy Study Groups Consensus P, Metzger BE et al (2010) International association of diabetes and pregnancy study groups recommendations on the diagnosis and classification of hyperglycemia in pregnancy. Diabetes Care 33:676–682. doi:10.2337/dc09-1848
Hartling L, Dryden DM, Guthrie A et al. (2012) Screening and diagnosing gestational diabetes mellitus. Evid Rep Technol Assess (Full Rep) 12:1–327
Agarwal MM (2015) Gestational diabetes mellitus: an update on the current international diagnostic criteria. World J Diabetes 6:782–791. doi:10.4239/wjd.v6.i6.782
Kohler M, Ziegler AG, Beyerlein A (2015) Development of a simple tool to predict the risk of postpartum diabetes in women with gestational diabetes mellitus. Acta Diabetol. doi:10.1007/s00592-015-0814-0
Zhao C, Wang F, Wang P, Ding H, Huang X, Shi Z (2015) Early second-trimester plasma protein profiling using multiplexed isobaric tandem mass tag (TMT) labeling predicts gestational diabetes mellitus. Acta Diabetol 52:1103–1112. doi:10.1007/s00592-015-0796-y
He X, de Seymour JV, Sulek K et al (2016) Maternal hair metabolome analysis identifies a potential marker of lipid peroxidation in gestational diabetes mellitus. Acta Diabetol 53:119–122. doi:10.1007/s00592-015-0737-9
Bhavadharini B, Mahalakshmi MM, Maheswari K et al (2016) Use of capillary blood glucose for screening for gestational diabetes mellitus in resource-constrained settings. Acta Diabetol 53:91–97. doi:10.1007/s00592-015-0761-9
Megia A, Gil-Lluis P, Naf S et al (2015) Cord blood FGF21 in gestational diabetes and its relationship with postnatal growth. Acta Diabetol 52:693–700. doi:10.1007/s00592-014-0705-9
Cabre A, Lazaro I, Girona J et al (2008) Plasma fatty acid binding protein 4 is associated with atherogenic dyslipidemia in diabetes. J Lipid Res 49:1746–1751. doi:10.1194/jlr.M800102-JLR200
Rhee EJ, Lee WY, Park CY et al (2009) The association of serum adipocyte fatty acid-binding protein with coronary artery disease in Korean adults. Eur J Endocrinol 160:165–172. doi:10.1530/EJE-08-0665
Biron-Shental T, Schaiff WT, Ratajczak CK, Bildirici I, Nelson DM, Sadovsky Y (2007) Hypoxia regulates the expression of fatty acid-binding proteins in primary term human trophoblasts. Am J Obstet Gynecol 197(516):e511–e516. doi:10.1016/j.ajog.2007.03.066
Makkar A, Mishima T, Chang G, Scifres C, Sadovsky Y (2014) Fatty acid binding protein-4 is expressed in the mouse placental labyrinth, yet is dispensable for placental triglyceride accumulation and fetal growth. Placenta 35:802–807. doi:10.1016/j.placenta.2014.07.008
Nourani MR, Owada Y, Kitanaka N et al (2005) Occurrence of immunoreactivity for adipocyte-type fatty acid binding protein in degenerating granulosa cells in atretic antral follicles of mouse ovary. J Mol Histol 36:491–497. doi:10.1007/s10735-006-9024-y
Fasshauer M, Seeger J, Waldeyer T et al (2008) Serum levels of the adipokine adipocyte fatty acid-binding protein are increased in preeclampsia. Am J Hypertens 21:582–586. doi:10.1038/ajh.2008.23
Shangguan X, Liu F, Wang H, He J, Dong M (2009) Alterations in serum adipocyte fatty acid binding protein and retinol binding protein-4 in normal pregnancy and preeclampsia. Clin Chim Acta 407:58–61. doi:10.1016/j.cca.2009.06.031
Scifres CM, Catov JM, Simhan H (2012) Maternal serum fatty acid binding protein 4 (FABP4) and the development of preeclampsia. J Clin Endocrinol Metab 97:E349–E356. doi:10.1210/jc.2011-2276
Di Cianni G, Lencioni C, Volpe L et al (2007) C-reactive protein and metabolic syndrome in women with previous gestational diabetes. Diabetes Metab Res Rev 23:135–140. doi:10.1002/dmrr.661
Heitritter SM, Solomon CG, Mitchell GF, Skali-Ounis N, Seely EW (2005) Subclinical inflammation and vascular dysfunction in women with previous gestational diabetes mellitus. J Clin Endocrinol Metab 90:3983–3988. doi:10.1210/jc.2004-2494
Winzer C, Wagner O, Festa A et al (2004) Plasma adiponectin, insulin sensitivity, and subclinical inflammation in women with prior gestational diabetes mellitus. Diabetes Care 27:1721–1727
Fakhrzadeh H, Alatab S, Sharifi F et al (2012) Carotid intima media thickness, brachial flow mediated dilation and previous history of gestational diabetes mellitus. J Obstet Gynaecol Res 38:1057–1063. doi:10.1111/j.1447-0756.2011.01829.x
Baeten JM, Bukusi EA, Lambe M (2001) Pregnancy complications and outcomes among overweight and obese nulliparous women. Am J Public Health 91:436–440
Sebire NJ, Jolly M, Harris JP et al (2001) Maternal obesity and pregnancy outcome: a study of 287,213 pregnancies in London. Int J Obes Relat Metab Disord 25:1175–1182. doi:10.1038/sj.ijo.0801670
Kim SY, Sappenfield W, Sharma AJ et al (2013) Racial/ethnic differences in the prevalence of gestational diabetes mellitus and maternal overweight and obesity, by nativity, Florida, 2004-2007. Obesity (Silver Spring) 21:E33–E40. doi:10.1002/oby.20025
Kim C, Newton KM, Knopp RH (2002) Gestational diabetes and the incidence of type 2 diabetes: a systematic review. Diabetes Care 25:1862–1868
Aye IL, Lager S, Ramirez VI et al (2014) Increasing maternal body mass index is associated with systemic inflammation in the mother and the activation of distinct placental inflammatory pathways. Biol Reprod 90:129. doi:10.1095/biolreprod.113.116186
Kleiblova P, Dostalova I, Bartlova M et al (2010) Expression of adipokines and estrogen receptors in adipose tissue and placenta of patients with gestational diabetes mellitus. Mol Cell Endocrinol 314:150–156. doi:10.1016/j.mce.2009.08.002
Hauguel-de Mouzon S, Guerre-Millo M (2006) The placenta cytokine network and inflammatory signals. Placenta 27:794–798. doi:10.1016/j.placenta.2005.08.009
Jansson N, Rosario FJ, Gaccioli F et al (2013) Activation of placental mTOR signaling and amino acid transporters in obese women giving birth to large babies. J Clin Endocrinol Metab 98:105–113. doi:10.1210/jc.2012-2667
Jansson T, Ekstrand Y, Bjorn C, Wennergren M, Powell TL (2002) Alterations in the activity of placental amino acid transporters in pregnancies complicated by diabetes. Diabetes 51:2214–2219
Jones HN, Jansson T, Powell TL (2009) IL-6 stimulates system A amino acid transporter activity in trophoblast cells through STAT3 and increased expression of SNAT2. Am J Physiol Cell Physiol 297:C1228–C1235. doi:10.1152/ajpcell.00195.2009
Jones HN, Woollett LA, Barbour N, Prasad PD, Powell TL, Jansson T (2009) High-fat diet before and during pregnancy causes marked up-regulation of placental nutrient transport and fetal overgrowth in C57/BL6 mice. FASEB J 23:271–278. doi:10.1096/fj.08-116889
Pantham P, Aye IL, Powell TL (2015) Inflammation in maternal obesity and gestational diabetes mellitus. Placenta 36:709–715. doi:10.1016/j.placenta.2015.04.006
Furuhashi M, Hiramitsu S, Mita T et al (2015) Reduction of serum FABP4 level by sitagliptin, a DPP-4 inhibitor, in patients with type 2 diabetes mellitus. J Lipid Res 56:2372–2380. doi:10.1194/jlr.M059469
Hotamisligil GS, Arner P, Caro JF, Atkinson RL, Spiegelman BM (1995) Increased adipose tissue expression of tumor necrosis factor-alpha in human obesity and insulin resistance. J Clin Invest 95:2409–2415. doi:10.1172/JCI117936
Dandona P, Weinstock R, Thusu K, Abdel-Rahman E, Aljada A, Wadden T (1998) Tumor necrosis factor-alpha in sera of obese patients: fall with weight loss. J Clin Endocrinol Metab 83:2907–2910. doi:10.1210/jcem.83.8.5026
Ahima RS, Flier JS (2000) Adipose tissue as an endocrine organ. Trends Endocrinol Metab 11:327–332
Cao H, Sekiya M, Ertunc ME et al (2013) Adipocyte lipid chaperone AP2 is a secreted adipokine regulating hepatic glucose production. Cell Metab 17:768–778. doi:10.1016/j.cmet.2013.04.012
Phillips CM, Perry IJ (2013) Does inflammation determine metabolic health status in obese and nonobese adults? J Clin Endocrinol Metab 98:E1610–E1619. doi:10.1210/jc.2013-2038
Furuhashi M, Ishimura S, Ota H, Miura T (2011) Lipid chaperones and metabolic inflammation. Int J Inflam 2011:642612. doi:10.4061/2011/642612
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical standard
All procedures performed in this study involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Human and animal rights
All procedures followed were in accordance with the ethical standards of the responsible committee on human experimentation (institutional and national) and with the Declaration of Helsinki 1975, as revised in 2008 (5).
Informed consent
Informed consent was obtained from all patients for being included in the study.
Additional information
Managed by Massimo Federici.
Rights and permissions
About this article
Cite this article
Ning, H., Tao, H., Weng, Z. et al. Plasma fatty acid-binding protein 4 (FABP4) as a novel biomarker to predict gestational diabetes mellitus. Acta Diabetol 53, 891–898 (2016). https://doi.org/10.1007/s00592-016-0867-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00592-016-0867-8