Abstract
Objectives
To assess the impact of diabetes mellitus (DM) on the postoperative motor and somatosensory functional recoveries of degenerative cervical myelopathy (DCM) patients.
Methods
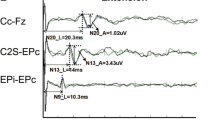
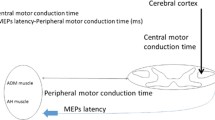
Motor and somatosensory evoked potentials (MEP and SSEPs) and modified Japanese Orthopedic Association (mJOA) scores were recorded in 27 diabetic (DCM-DM group) and 38 non-diabetic DCM patients (DCM group) before and 1 year after surgery. The central motor (CMCT) and somatosensory (CSCT) conduction time were recorded to evaluate the conductive functions of the spinal cord.
Results
The mJOA scores, CMCT and CSCT improved (t test, p < 0.05) in both of the DCM-DM and DCM groups 1 year after surgery. The mJOA recovery rate (RR) and CSCT recovery ratio were significantly worse (t test, p < 0.05) in the DCM-DM group compared to the DCM group. DM proved to be a significant independent risk factor for poor CSCT recovery (OR = 4.52, 95% CI 2.32–7.12) after adjusting for possible confounding factors. In DCM-DM group, CSCT recovery ratio was also correlated with preoperative HbA1 level (R = − 0.55, p = 0.003). Furthermore, DM duration longer than 10 years and insulin dependence were risk factors for lower mJOA, CMCT and CSCT recoveries among all DCM-DM patients (t test, p < 0.05).
Conclusions
DM may directly hinders spinal cord conduction recovery in DCM patients after surgery. Corticospinal tract impairments are similar between DCM and DCM-DM patients, but significantly worsened in chronic or insulin-dependent DM patients. The dorsal column is more sensitively affected in all DCM-DM patients. Deeper investigation into the mechanisms and neural regeneration strategies is needed.


Similar content being viewed by others
References
Badhiwala JH, Ahuja CS, Akbar MA, Witiw CD, Nassiri F, Furlan JC, Curt A, Wilson JR, Fehlings MG (2020) Degenerative cervical myelopathy—update and future directions. Nat Rev Neurol 16:108–124. https://doi.org/10.1038/s41582-019-0303-0
Kusin DJ, Ahn UM, Ahn NU (2016) The influence of diabetes on surgical outcomes in cervical myelopathy. Spine (Phila Pa 1976) 41:1436–1440. https://doi.org/10.1097/BRS.0000000000001560
Kawaguchi Y, Matsui H, Ishihara H, Gejo R, Yasuda T (2000) Surgical outcome of cervical expansive laminoplasty in patients with diabetes mellitus. Spine (Phila Pa 1976) 25:551–555. https://doi.org/10.1097/00007632-200003010-00004
Nori S, Nagoshi N, Yoshioka K, Nojiri K, Takahashi Y, Fukuda K, Ikegami T, Yoshida H, Iga T, Tsuji O, Suzuki S, Okada E, Yagi M, Nakamura M, Matsumoto M, Watanabe K, Ishii K, Yamane J (2021) Diabetes does not adversely affect neurological recovery and reduction of neck pain after posterior decompression surgery for cervical spondylotic myelopathy: results from a retrospective multicenter study of 675 patients. Spine (Phila Pa 1976) 46:433–439. https://doi.org/10.1097/BRS.0000000000003817
Machino M, Yukawa Y, Ito K, Inoue T, Kobayakawa A, Matsumoto T, Ouchida J, Tomita K, Kato F (2014) Impact of diabetes on the outcomes of cervical laminoplasty: a prospective cohort study of more than 500 patients with cervical spondylotic myelopathy. Spine (Phila Pa 1976) 39:220–227. https://doi.org/10.1097/BRS.0000000000000102
Arnold PM, Fehlings MG, Kopjar B, Yoon ST, Massicotte EM, Vaccaro AR, Brodke DS, Shaffrey CI, Smith JS, Woodard EJ, Banco RJ, Chapman JR, Janssen ME, Bono CM, Sasso RC, Dekutoski MB, Gokaslan ZL (2014) Mild diabetes is not a contraindication for surgical decompression in cervical spondylotic myelopathy: results of the AOSpine North America multicenter prospective study (CSM). Spine J 14:65–72. https://doi.org/10.1016/j.spinee.2013.06.016
Maertens de Noordhout A, Remacle JM, Pepin JL, Born JD, Delwaide PJ (1991) Magnetic stimulation of the motor cortex in cervical spondylosis. Neurology 41:75–80. https://doi.org/10.1212/wnl.41.1.75
Yu Z, Lin K, Chen J, Chen KH, Guo W, Dai Y, Chen Y, Zou X, Peng X (2020) Magnetic resonance imaging and dynamic X-ray’s correlations with dynamic electrophysiological findings in cervical spondylotic myelopathy: a retrospective cohort study. BMC Neurol 20:367. https://doi.org/10.1186/s12883-020-01945-4
Di Lazzaro V, Restuccia D, Colosimo C, Tonali P (1992) The contribution of magnetic stimulation of the motor cortex to the diagnosis of cervical spondylotic myelopathy. Correlation of central motor conduction to distal and proximal upper limb muscles with clinical and MRI findings. Electroencephalogr Clin Neurophysiol 85:311–320. https://doi.org/10.1016/0168-5597(92)90107-m
Nakai S, Sonoo M, Shimizu T (2008) Somatosensory evoked potentials (SEPs) for the evaluation of cervical spondylotic myelopathy: utility of the onset-latency parameters. Clin Neurophysiol 119:2396–2404. https://doi.org/10.1016/j.clinph.2008.07.003
Nakanishi K, Tanaka N, Kamei N, Hiramatsu T, Ujigo S, Sumiyoshi N, Rikita T, Takazawa A, Ochi M (2015) Electrophysiological assessments of the motor pathway in diabetic patients with compressive cervical myelopathy. J Neurosurg Spine 23:707–714. https://doi.org/10.3171/2015.3.SPINE141060
Chistyakov AV, Soustiel JF, Hafner H, Kaplan B, Feinsod M (2004) The value of motor and somatosensory evoked potentials in evaluation of cervical myelopathy in the presence of peripheral neuropathy. Spine (Phila Pa 1976) 29:E239-247. https://doi.org/10.1097/01.brs.0000127191.12310.fb
Committee of the Japan Diabetes Society on the Diagnostic Criteria of Diabetes M, Seino Y, Nanjo K, Tajima N, Kadowaki T, Kashiwagi A, Araki E, Ito C, Inagaki N, Iwamoto Y, Kasuga M, Hanafusa T, Haneda M, Ueki K (2010) Report of the committee on the classification and diagnostic criteria of diabetes mellitus. J Diabetes Investig 1:212–228. https://doi.org/10.1111/j.2040-1124.2010.00074.x
American Diabetes A (2021) 6. Glycemic targets: standards of medical care in diabetes-2021. Diabetes Care 44:S73–S84. https://doi.org/10.2337/dc21-S006
Tetreault L, Kopjar B, Nouri A, Arnold P, Barbagallo G, Bartels R, Qiang Z, Singh A, Zileli M, Vaccaro A, Fehlings MG (2017) The modified Japanese Orthopaedic Association scale: establishing criteria for mild, moderate and severe impairment in patients with degenerative cervical myelopathy. Eur Spine J 26:78–84. https://doi.org/10.1007/s00586-016-4660-8
Hirabayashi K, Miyakawa J, Satomi K, Maruyama T, Wakano K (1981) Operative results and postoperative progression of ossification among patients with ossification of cervical posterior longitudinal ligament. Spine (Phila Pa 1976) 6:354–364. https://doi.org/10.1097/00007632-198107000-00005
Nuwer MR, Aminoff M, Desmedt J, Eisen AA, Goodin D, Matsuoka S, Mauguière F, Shibasaki H, Sutherling W, Vibert JF (1994) IFCN recommended standards for short latency somatosensory evoked potentials. Report of an IFCN committee. International Federation of Clinical Neurophysiology. Electroencephalogr Clin Neurophysiol 91:6–11. https://doi.org/10.1016/0013-4694(94)90012-4
Yu Z, Chen J, Cheng X, Xie D, Chen Y, Zou X, Peng X (2022) Patients with degenerative cervical myelopathy exhibit neurophysiological improvement upon extension and flexion: a retrospective cohort study with a minimum 1-year follow-up. BMC Neurol 22:110. https://doi.org/10.1186/s12883-022-02641-1
Zegers de Beyl D, Delberghe X, Herbaut AG, Brunko E (1988) The somatosensory central conduction time: physiological considerations and normative data. Electroencephalogr Clin Neurophysiol 71:17–26. https://doi.org/10.1016/0168-5597(88)90015-9
Nakanishi K, Tanaka N, Kamei N, Ohta R, Fujioka Y, Hiramatsu T, Ujigo S, Ochi M (2014) Electrophysiological evidence of functional improvement in the corticospinal tract after laminoplasty in patients with cervical compressive myelopathy: clinical article. J Neurosurg Spine 21:210–216. https://doi.org/10.3171/2014.4.SPINE13545
Yu Z, Pan W, Chen J, Peng X, Ling Z, Zou X (2022) Application of electrophysiological measures in degenerative cervical myelopathy. Front Cell Dev Biol 10:834668. https://doi.org/10.3389/fcell.2022.834668
Machino M, Yukawa Y, Ito K, Inoue T, Kobayakawa A, Matsumoto T, Ouchida J, Tomita K, Kato F (2014) Risk factors for poor outcome of cervical laminoplasty for cervical spondylotic myelopathy in patients with diabetes. J Bone Joint Surg Am 96:2049–2055. https://doi.org/10.2106/jbjs.N.00064
Kim HJ, Moon SH, Kim HS, Moon ES, Chun HJ, Jung M, Lee HM (2008) Diabetes and smoking as prognostic factors after cervical laminoplasty. J Bone Joint Surg Br 90:1468–1472. https://doi.org/10.1302/0301-620x.90b11.20632
Dokai T, Nagashima H, Nanjo Y, Tanida A, Teshima R (2012) Surgical outcomes and prognostic factors of cervical spondylotic myelopathy in diabetic patients. Arch Orthop Trauma Surg 132:577–582. https://doi.org/10.1007/s00402-011-1449-4
Tanishima S, Mihara T, Tanida A, Takeda C, Murata M, Takahashi T, Yamane K, Morishita T, Morio Y, Ishii H, Fukata S, Nanjo Y, Hamamoto Y, Dokai T, Nagashima H (2019) Influence of diabetes mellitus on surgical outcomes in patients with cervical myelopathy: a prospective, multicenter study. Asian Spine J 13:468–477. https://doi.org/10.31616/asj.2018.0082
Takahashi J, Hirabayashi H, Hashidate H, Ogihara N, Yamazaki I, Kamimura M, Ebara S, Kato H (2008) Assessment of cervical myelopathy using transcranial magnetic stimulation and prediction of prognosis after laminoplasty. Spine (Phila Pa 1976) 33:E15-20. https://doi.org/10.1097/BRS.0b013e31815e5dae
Biessels GJ, Cristino NA, Rutten GJ, Hamers FP, Erkelens DW, Gispen WH (1999) Neurophysiological changes in the central and peripheral nervous system of streptozotocin-diabetic rats. Course of development and effects of insulin treatment. Brain 122(Pt 4):757–768. https://doi.org/10.1093/brain/122.4.757
Suzuki C, Ozaki I, Tanosaki M, Suda T, Baba M, Matsunaga M (2000) Peripheral and central conduction abnormalities in diabetes mellitus. Neurology 54:1932–1937. https://doi.org/10.1212/wnl.54.10.1932
Krnjevic K, Morris ME (1976) Input-output relation of transmission through cuneate nucleus. J Physiol 257:791–815. https://doi.org/10.1113/jphysiol.1976.sp011398
Cameron NE, Eaton SE, Cotter MA, Tesfaye S (2001) Vascular factors and metabolic interactions in the pathogenesis of diabetic neuropathy. Diabetologia 44:1973–1988. https://doi.org/10.1007/s001250100001
Olsson Y, Save-Soderbergh J, Sourander P, Angervall L (1968) A patho-anatomical study of the central and peripheral nervous system in diabetes of early onset and long duration. Pathol Eur 3:62–79
Reske-Nielsen E, Lundbaek K (1968) Pathological changes in the central and peripheral nervous system of young long-term diabetics. II. The spinal cord and peripheral nerves. Diabetologia 4:34–43. https://doi.org/10.1007/BF01241031
Janahi NM, Santos D, Blyth C, Bakhiet M, Ellis M (2015) Diabetic peripheral neuropathy, is it an autoimmune disease? Immunol Lett 168:73–79. https://doi.org/10.1016/j.imlet.2015.09.009
Slager UT (1978) Diabetic myelopathy. Arch Pathol Lab Med 102:467–469
Funding
Natural Science Foundation of Guangdong Province (2022A1515012557), Guangzhou Science and Technology Program Key Projects (202103000053) and National Natural Science Foundation of China (82102636).
Author information
Authors and Affiliations
Contributions
ZY, CC and TY analyzed the data, drafted and revised the manuscript. YY revised the manuscript critically for important intellectual content. YC and XZ made substantial contributions to the conception and design of the study and provided final approval of the version to be published. SZ and SZ were involved in the acquisition of data. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors have no conflict of interest to declare.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Yu, Z., Chen, C., Yu, T. et al. Electrophysiological evidence of diabetes’ impacts on central conduction recoveries in degenerative cervical myelopathy after surgery. Eur Spine J 32, 2101–2109 (2023). https://doi.org/10.1007/s00586-023-07605-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00586-023-07605-8