Abstract
Purpose
In this meta-analysis, we aim to compare ketamine use versus a control group (saline solution) during induction of anesthesia in adolescent idiopathic scoliosis patients undergoing fusion surgery in terms of postoperative opioid consumption, pain control, and side effects.
Methods
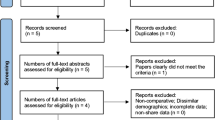
A PubMed search of studies published over the last 20 years using the descriptor/terms “ketamine AND scoliosis” was performed. Baseline characteristics of each article were obtained and efficacy measures analyzed (morphine equivalent treatment at 24, 48, and 72 h postoperatively, complications (vomiting/nausea and pruritus), length of hospital stay (days); and pain score (VAS)) (Review Manager 5.4 software package).
Results
Five randomized clinical trials were included. Morphine administration showed statistically significant differences at 24 and 48 h (MD − 0.15, 95% CI − 0.18 to − 0.12) and (MD − 0.26, 95% CI − 0.31 to − 0.21) between the ketamine and control (saline solution), respectively. No intergroup differences were found regarding nausea/vomiting and pruritus (OR 0.77, 95% CI 0.35 to 1.67) and (OR 0.71, 95% CI 0.31 to 1.62), respectively, same as for the pain score (MD − 0.75, 95% CI − 1.71 to 0.20).
Conclusions
The use intraoperative and postoperative continuous low doses of ketamine significantly reduces opioid use throughout the first 48 h in patients with AIS who undergo posterior spinal fusion.






Similar content being viewed by others
References
Wong GTC, Yuen VMY, Chow BFM et al (2007) Persistent pain inpatients following scoliosis surgery. Eur Spine J 16:1551–1556. https://doi.org/10.1007/s00586-007-0361-7
Gaukroger PB, Tomkins DP, van der Walt JH (1989) Patient-controlled analgesia in children. Anaesth Intensive Care 17:264–268. https://doi.org/10.1177/0310057X8901700304
Crawford MW, Hickey C, Zaarour C et al (2006) Development of acute opioid tolerance during infusion of remifentanil for pediatric scoliosis surgery. Anesth Analg 102:1662–1667. https://doi.org/10.1213/01.ane.0000216036.95705.c2
Wilder-Smith OHG, Arendt-Nielsen L (2006) Postoperative hyperalgesia. Anesthesiology 104:601–607. https://doi.org/10.1097/00000542-200603000-00028
Koppert W, Schmelz M (2007) The impact of opioid-induced hyperalgesia for postoperative pain. Best Pract Res Clin Anaesthesiol 21:65–83. https://doi.org/10.1016/j.bpa.2006.12.004 (PMID: 17489220)
Subramaniam K, Subramaniam B, Steinbrook RA (2004) Ketamine as adjuvant analgesic to opioids: a quantitative and qualitative systematic review. Anesth Analg 99:482–495. https://doi.org/10.1213/01.ANE.0000118109.12855.07
Kollender Y, Bickels J, Stocki D et al (2008) Subanesthetic ketamine spares postoperative morphine and controls pain better than standard morphine does alone in orthopedic-oncological patients. Eur J Cancer 44(7):954–962. https://doi.org/10.1016/j.ejca.2008.02.021
Becke K, Albrecht S, Schmitz B et al (2005) Intraoperative low-dose S-ketamine has no preventive effects on postoperative pain and morphine consumption after major urological surgery in children. Pediatr Anesth 15:484–490. https://doi.org/10.1111/j.1460-9592.2005.01476.x
Joseph C, Gaillat F, Duponq R, Lieven R, Baumstarck K, Thomas P, Penot-Ragon C, Kerbaul F (2012) Is there any benefit to adding intravenous ketamine to patient-controlled epidural analgesia after thoracic surgery? A randomized double-blind study. Eur J Cardiothorac Surg 42(4):e58-65. https://doi.org/10.1093/ejcts/ezs398
Liberati A, Altman DG, Tetzlaff J et al (2009) The PRISMA statement for reporting systematic reviews and meta-analyses of studies that evaluate health care interventions: explanation and elaboration. PLoS Med 6(7):e1000100. https://doi.org/10.1371/journal.pmed.1000100
Ricciardelli RM, Walters NM, Pomerantz M et al (2020) The efficacy of ketamine for postoperative pain control in adolescent patients undergoing spinal fusion surgery for idiopathic scoliosis. Spine Deform 8(3):433–440. https://doi.org/10.1007/s43390-020-00073-w
Perelló M, Artés D, Pascuets C et al (2017) Prolonged perioperative low-dose ketamine does not improve short and long-term outcomes after pediatric idiopathic scoliosis surgery. Spine 42(5):E304–E312. https://doi.org/10.1097/BRS.0000000000001772
Engelhardt T, Zaarour C, Naser B et al (2008) Intraoperative low-dose ketamine does not prevent a remifentanil-induced increase in morphine requirement after pediatric scoliosis surgery. Anesth Analg 107(4):1170–1175. https://doi.org/10.1213/ane.0b013e318183919e
Minoshima R, Kosugi S, Nishimura D et al (2015) Intra- and postoperative low-dose ketamine for adolescent idiopathic scoliosis surgery: a randomized controlled trial. Acta Anaesthesiol Scand 59(10):1260–1268. https://doi.org/10.1111/aas.12571
Pestieau SR, Finkel JC, Junqueira MM et al (2014) Prolonged perioperative infusion of low-dose ketamine does not alter opioid use after pediatric scoliosis surgery. Paediatr Anaesth 24(6):582–590. https://doi.org/10.1111/pan.12417
Bennett GJ (2000) Update on the neurophysiology of pain transmission and modulation: focus on the NMDAreceptor. J Pain Symptom Manage 19:2–6. https://doi.org/10.1016/s0885-3924(99)00120-7
Joly V, Richebe P, Guignard B et al (2005) Remifentanilinduced postoperative hyperalgesia and its prevention with small-dose ketamine. Anesthesiology 103:147–155. https://doi.org/10.1097/00000542-200507000-00022
Dallimore D, Anderson B (2008) Ketamine anesthesia in children—exploring infusion regimens. Paediatr Anaesth 18:708–714. https://doi.org/10.1111/j.1460-9592.2008.02665.x
Engelhardt T, Zaarour C, Crawford MW (2011) Plasma cyclic guanosine 3′,5′-monophosphate levels: a marker of glutamate-nitric oxideguanyl cyclase activity? J Opioid Manag 7:462–466. https://doi.org/10.5055/jom.2011.0087
Lavand’homme P, De Kock M, Waterloos H (2005) Intraoperative epidural analgesia combined with ketamine provides effective preventive analgesia in patients undergoing major digestive surgery. Anesthesiology 103(4):813–820. https://doi.org/10.1097/00000542-200510000-00020
Adriaenssens G, Vermeyen KM, Hoffmann VL et al (1999) Postoperative analgesia with i.v. patient-controlled morphine: effect of adding ketamine. Br J Anaesth 83(3):393–396. https://doi.org/10.1093/bja/83.3.393
Laskowski K, Stirling A, McKay W et al (2011) A systematic review of intravenous ketamine for postoperative analgesia. Can J Anesth 58:911–923. https://doi.org/10.1007/s12630-011-9560-0
Carstensen M, Møller A (2010) Adding ketamine to morphine for intravenous patient-controlled analgesia for acute postoperative pain: a qualitative review of randomized trials. Br J Anaesth 104:401–406. https://doi.org/10.1093/bja/aeq041
Sveticic G, Farzanegan F (2008) Is the combination of morphine with ketamine better than morphine alone for postoperative intravenous patient-controlled analgesia? Anesth Analg 106:287–293. https://doi.org/10.1213/01.ane.0000289637.11065.8f
Dahmani S, Michelet D, Abback P-S et al (2011) Ketamine for perioperative pain management in children: a meta-analysis of published studies. Paediatr Anaesth 21:636–652. https://doi.org/10.1111/j.1460-9592.2011.03566.x
Himmelseher S, Durieux ME (2005) Ketamine for perioperative pain management. Anesthesiology 102:211–220. https://doi.org/10.1097/00000542-200501000-00030
Min TJ, Kim WY, Jeong WJ et al (2012) Effect of ketamine on intravenous patient-controlled analgesia using hydromorphone and ketorolac after the Nuss surgery in pediatric patients. Korean J Anesthesiol 62(2):142–147. https://doi.org/10.4097/kjae.2012.62.2.142
Abrishamkar S, Eshraghi N, Feizi A et al (2012) Analgesic effects of ketamine infusion on postoperative pain after fusion and instrumentation of the lumbar spine: a prospective randomized clinical trial. Med Arch 66(2):107–110. https://doi.org/10.5455/medarh.2012.66.107-110
Batra YK, Shamsah M, Al-Khasti MJ et al (2007) Intraoperative small-dose ketamine does not reduce pain or analgesic consumption during perioperative opioid analgesia in children after tonsillectomy. Int J Clin Pharmacol Ther 45:155–160. https://doi.org/10.5414/cpp45155
Burstal R, Danjoux G, Hayes C et al (2001) PCA ketamine and morphine after abdominal hysterectomy. Anaesth Intensive Care 29(3):246–251. https://doi.org/10.1177/0310057X0102900304
Murdoch CJ, Crooks BA, Miller CD (2002) Effect of the addition of ketamine to morphine in patient-controlled analgesia. Anaesthesia 57(5):484–488. https://doi.org/10.1046/j.0003-2409.2001.02409.x
Reeves M, Lindholm DE, Myles PS et al (2001) Adding ketaminem to morphine for patient-controlled analgesia after major abdominal surgery: a double-blinded, randomized controlled trial. Anesth Analg 93(1):116–120. https://doi.org/10.1097/00000539-200107000-00025
Kissin I, Lee SS, Arthur GR et al (1996) Time course characteristics of acute tolerance development to continuously infused alfentanil in rats. Anesth Analg 83:600–605. https://doi.org/10.1097/00000539-199609000-00029
Englelhardt T, Lowe PR, Galley HF et al (2006) Inhibition of neuronal nitric oxide synthase reduces the propofol requirements in wild-type and nNOS knockout mice. Eur J Anaesthesiol 23:197–201. https://doi.org/10.1017/S0265021505002188
Aubrun F, Salvi N, Coriat P et al (2005) Sex- and age-related differences in morphine requirements for postoperative pain relief. Anesthesiology 103:156–160. https://doi.org/10.1097/00000542-200507000-00023
Jabbour HJ, Naccache NM, Jawish RJ et al (2014) Ketamine and magnesium association reduces morphine consumption after scoliosis surgery: prospective randomised double-blind study. Acta Anaesthesiol Scand 58(5):572–579. https://doi.org/10.1111/aas.12304
Funding
No funding was received.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors have no relevant financial or non-financial interests to disclose.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Mariscal, G., Morales, J., Pérez, S. et al. Meta-analysis of the efficacy of ketamine in postoperative pain control in adolescent idiopathic scoliosis patients undergoing spinal fusion. Eur Spine J 31, 3492–3499 (2022). https://doi.org/10.1007/s00586-022-07422-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00586-022-07422-5