Purpose
Abstract
Total disc replacements, comprising all-metal articulations, are compromised by wear and particle production. Metallic wear debris and ions trigger a range of biological responses including inflammation, genotoxicity, cytotoxicity, hypersensitivity and pseudotumour formation, therefore we hypothesise that, due to proximity to the spinal cord, glial cells may be adversely affected.
Methods
Clinically relevant cobalt chrome (CoCr) and stainless steel (SS) wear particles were generated using a six-station pin-on-plate wear simulator. The effects of metallic particles (0.5–50 μm3 debris per cell) and metal ions on glial cell viability, cellular activity (glial fibrillary acidic protein (GFAP) expression) and DNA integrity were investigated in 2D and 3D culture using live/dead, immunocytochemistry and a comet assay, respectively.
Results
CoCr wear particles and ions caused significant reductions in glial cell viability in both 2D and 3D culture systems. Stainless steel particles did not affect glial cell viability or astrocyte activation. In contrast, ions released from SS caused significant reductions in glial cell viability, an effect that was especially noticeable when astrocytes were cultured in isolation without microglia. DNA damage was observed in both cell types and with both biomaterials tested. CoCr wear particles had a dose-dependent effect on astrocyte activation, measured through expression of GFAP.
Conclusions
The results from this study suggest that microglia influence the effects that metal particles have on astrocytes, that SS ions and particles play a role in the adverse effects observed and that SS is a less toxic biomaterial than CoCr alloy for use in spinal devices.
Graphic abstract
These slides can be retrieved under Electronic Supplementary Material.

Similar content being viewed by others
Introduction
Significant economic and patient issues associated with early failure of metal-on-metal (MOM) total hip and resurfacing replacements have been the focus of the public and scientific media recently [1]. These events have led to concerns over other MOM implant systems, including those used in spinal surgery, where a number of articulating total disc replacements (TDRs) are still in use. TDRs comprise structures that have a large number of interfaces, e.g. endplate/bone interface, endplate screws and bone as well as the articulating surfaces. All of these interacting surfaces may produce debris and/or ions with the potential to invoke adverse biological responses, which may lead to failure and ultimately device revision. Metallic TDRs generate wear, debris and/or corrosion products [2,3,4], therefore cytotoxic and immunological responses may become a significant problem in the future. Of particular concern is a recent case report describing metallosis and the formation of pseudotumours around a metal-on-metal TDR, similar to failing hip resurfacing arthroplasties [5]. There is evidence documenting the release of metallic particulates from other spinal devices and the inflammatory responses that ensue, including cellular infiltrates and cytokine release (predominantly TNF-α) [6,7,8,9,10,11,12]. Clinically there are reports that describe neurological symptoms, including radicular pain and paraparesis, in patients with metallosis associated with spinal devices [7, 13]. Inflammation would appear to be a key factor in the development of such symptoms; however, other reports also describe late operative site pain [8, 9].
The effects of ions and metal debris remain largely unknown for the neural structures in the spine. Cells of the central nervous system (CNS) are protected by the meninges. Disruption of these anatomical features makes the spinal cord and adjacent neural structures particularly vulnerable to exposure to tribo-corrosive products. Recent research has shown that porcine dural cells and dural organ cultures exposed to cobalt chromium (CoCr) nanoparticles produced significantly elevated levels of the pro-inflammatory chemokine IL-8 [14, 15], which adversely affected the barrier function of the endothelial cell layer [16]. In addition, significant alterations to the structural integrity of the dura mater were observed together with elevated expression of matrix metalloproteinases (MMP-1, 3, 9, 13) and tissue inhibitor of metalloproteinase-1 indicating that tissue remodelling and collagen fibre loosening were prevalent in the presence of CoCr particles. In combination, these effects may enhance nanoparticle penetration into the dura mater and enable access to the cord tissue. Indeed, there is evidence that particles introduced epidurally have the ability to cross both the dural barrier in vivo [11] and the blood spinal cord barrier; the latter has been exploited for drug delivery [17]. In addition, it has been shown that rapid CSF fluid flow occurs from the subarachnoid space into the spinal cord canal via the peri-vascular spaces and these may allow nanoparticle access to these tissues [18, 19]. This has led to the hypothesis that the barrier functions protecting the spinal cord will be compromised by exposure to metal wear particles and/or ions and that subsequent adverse responses to these species by neural cell populations will result in neurotoxicity, leading to complications and subsequent failure of spinal implants. This is likely to be determined by material type, with some materials (e.g. titanium, PEEK) being less neurotoxic and some materials causing adverse effects. It should be noted that both titanium and PEEK are not or are no longer used as bearing materials due to poor tribological properties and inadequate wear resistance of titanium and a lack of evidence regarding the tribological performance of PEEK as a bearing surface compared to the more commonly used polymer, ultra high molecular weight polyethylene (UHMWPE). The present study investigated the response of primary rat glial cells to CoCr and stainless steel particles and ions from implant biomaterials in 2D and 3D cell culture systems over time. Particulate and ion effects on neural cell viability, DNA integrity and glial fibrillary acidic protein (GFAP) expression in astrocytes were the focus of the study.
Materials and methods
Wear particle generation and characterisation
Clinically relevant cobalt–chromium and stainless steel wear particles were generated using an in-house-manufactured pin-on-plate tribometer and distilled water as the lubricant as described by Germain et al. [20]. Both the pin and plate were made from either medical-grade wrought cobalt–chromium alloy (according to ASTM F1537) with a high carbon content or medical-grade stainless steel (316L). Debris was generated using smooth (Ra 0.01–0.02 µm) counterfaces with a multidirectional motion (rotation ± 30°, stroke length 28 mm, frequency 1 Hz) and a contact stress of 11 MPa over 80 h. For particle characterisation, 1 ml aliquot of each lubricant was sequentially filtered through 5-, 1-, 0.1- and 0.015-µm-sized polycarbonate filters (Whatman) and filters examined using high-resolution scanning electron microscopy (SEM), at magnifications up to × 150K, combined with energy-dispersive X-ray (EDX) analysis to determine particle composition. Particle size and area distributions were generated using Image Pro Plus 6.0 (Media Cybernetics, USA). Stock solutions (1 mg ml−1) of each debris type were generated from sterile, endotoxin-free particles [20]. Particles were dispersed by sonication prior to use.
Neural cell isolation and culture
All experiments were performed according to the UK Animals (Scientific Procedures) Act (1986) and approved by the appropriate institutional ethical committee. Primary astrocyte cultures were prepared from the cortices of postnatal 2-day-old Wistar rat pups as described by East et al. [21]. The tissue was chopped and cells isolated by incubation with 250 μg ml−1 trypsin. Digests were repeatedly triturated using a 1-ml pipette to break down any larger clumps of tissue. The resultant cells were resuspended in Dulbecco’s modified Eagle’s medium (DMEM) supplemented with penicillin/streptomycin (100 U ml−1 and 100 μg ml−1, respectively) and with 10% (v/v) foetal calf serum (FCS). This suspension was dispensed into tissue culture flasks that had been coated with poly-d-lysine (50 µg ml−1) and placed in a humidified incubator at 37 °C with 5% CO2/95% air. Cells were expanded in culture for 10–14 days to reach confluence. After this time, cell types were separated by shaking at 150 rpm for 4 h to remove microglia and less adherent cell types from the resulting astrocytes. Both cell types were passaged into fresh media before being exposed to wear particles and/or ions.
Culture of cells with particles and ions
Primary rat astrocytes and microglia in isolation and in co-culture were cultured with CoCr or stainless steel wear particles or ions (0.05–50 µm3 per cell) for up to 5 days in both 2D and 3D culture systems. In the 2D system, cells were seeded at 1 × 104 cells per well (n = 6) in supplemented DMEM medium in poly-D lysine-coated 96-well plates. Particles were added, and cell viability was assessed after 1, 3 and 5 days using the ATP-Lite assay (PerkinElmer, Windsor, UK). DNA damage was also assessed using the alkaline Comet assay (Trevigen, Maryland, USA) at 24-h, 48-h and 5-day time points [22]. After staining with SYBR Gold (Thermo Fisher Scientific, UK), slides were imaged using a Zeiss fluorescent microscope and the Olive tail moment was used to calculate DNA damage over time using Comet IV Lite software (Perceptive Instruments, UK). Data were expressed as mean comet tail moment ± 95% confidence limits (n = 6).
For 3D systems, cells were seeded at 1 × 106 ml−1 into collagen gels (First Link, UK; 2 mg ml−1) along with the appropriate volume of particles suspended in DMEM. A volume of 100 µl of collagen gel containing cells and particles was seeded into 96-well plates. Particles were dosed at 0.5, 5, 50 µm3 per cell (n = 6), and cell viability assessed using a live/dead cytotoxicity assay for mammalian cells (Thermo Fisher Scientific, UK) after 48 h and 5 days in culture. The expression of glial fibrillary acidic protein (GFAP), a marker of astrocyte activation and reactive gliosis, was assessed at the same time points using a polyclonal primary antibody (Agilent Technologies UK Ltd) and an anti-rabbit DyLight® 549 (Vector Laboratories, UK) secondary antibody. Hoescht 33258 was used to stain cell nuclei. Cells were imaged at six locations within each gel (n = 6) using a LSM510 confocal laser scanning microscope (Carl Zeiss Ltd) leading to 36 images per test. The number of live and dead cells were calculated, and immunopositive cells were scored (images were negative (GFAP expression not detectable) or positive (GFAP upregulated to detectable level)) using ImageJ software (NIH, USA) compared to the negative control images.
For the culture of primary glial cells with CoCr or SS ions, the appropriate mass of particles (equivalent to 0.5, 5, 50 µm3 particles per cell) was incubated with supplemented DMEM for 24 h at 37 °C in 5% (v/v) CO2 in air with agitation. The particle suspensions were centrifuged at 160,000 × g, and the supernatants containing ions were added to wells containing cells suspended in collagen gel. The same assays as described above were performed to assess the effect of metal ions on cell viability and the expression of GFAP.
Statistical analysis
Data were presented as mean ± 95% confidence limits of the mean and statistical analysis performed using a two-way ANOVA. Significant differences between means were determined using Tukey’s post hoc analysis. A p value of p < 0.05 was indicative of statistical significance.
Results
Characterisation of cobalt chromium and stainless steel particles
Particles were isolated through sequential filtration through 5-, 1-, 0.1- and 0.015-μm filters, which were coated with platinum and imaged using high-resolution FEGSEM. Cobalt chromium particles were viewed as agglomerates on all filters (Fig. 1a–d). At higher magnifications (> × 30K) on the 0.1- and 0.015-μm filters, particles were round to oval and in the nanoscale. The majority of particles produced were between 10 and 120 nm, and the mode was 30–39 nm (Fig. 2). Particles were identified as comprising cobalt, chromium, molybdenum and oxygen as the major elements (Online Resources 1). Stainless steel particles were observed as irregular granular particles on the larger pore size filters and round nanoscale particles on the smaller pore size filters (Fig. 3a–d). The particles ranged in size from 10 nm to larger than 1 mm, and the mode was between 30 and 39 nm (Fig. 4) similar to CoCr. The elemental composition of the stainless steel particles was comprised largely of iron and nickel plus carbon and oxygen (Online Resource 2).
High-resolution FEGSEM images of cobalt chromium particles generated by pin-on-plate simulation in water lubricant. a Aggregate particles isolated on a 5-µm filter, × 120 magnification; b aggregates of cobalt chromium particles isolated on a 1-µm filter, × 400 magnification; c aggregates of cobalt chromium particles isolated on a 0.1-µm filter, × 30,000 magnification; d high-magnification image of cobalt chromium aggregates isolated on a 0.1-µm filter, × 60,000 magnification. Arrows indicate large irregular micron-sized particles. ** indicates agglomerate of submicron-sized particles
High-resolution FEGSEM images of stainless steel particles generated by pin-on-plate simulation in water lubricant. a Aggregate particles isolated on a 5-µm filter, × 120 magnification; b aggregates of stainless steel particles isolated on a 1-µm filter, × 120 magnification; c aggregates of stainless steel particles isolated on a 1-µm filter, × 400 magnification; d high-magnification image of stainless steel aggregates isolated on a 0.1-µm filter, × 60,000 magnification
The effect of CoCr and stainless steel particles on primary astrocytes and microglia in 2D culture
The effect of increasing volumes of cobalt chrome particles on the viability of primary astrocytes and microglia in 2D co-culture was assessed using the ATP-Lite™ assay over time. After 24 h, there were no significant effects on cell viability; however, after 3 and 5 days in culture a statistically significant reduction in viability was observed for all particle doses (Fig. 5a; p < 0.05, ANOVA). With stainless steel particles, there was an initial adverse effect on viability observed after 24 h at concentrations of between 0.5 and 50 µm3 per cell and at day 5 at the highest particle dose only (Fig. 5b; p < 0.05, ANOVA).
The effect of cobalt chrome and stainless steel particles on the viability of primary astrocytes and microglia in co-culture in 2D monolayer culture over 5 days. Cell viability measured using ATP-Lite assay after exposure to increasing concentrations of a CoCr and b stainless steel particles; *significant difference between means (two-way ANOVA plus Tukey, p < 0.05) when compared to the cell only negative control
Only the highest concentration of CoCr particles (50 µm3 per cell) had a significant effect on DNA integrity in the first 24 h of culture (Fig. 6a; p < 0.05 ANOVA plus Tukey). After 48 h, all concentrations of CoCr particles adversely affected DNA integrity; however, after 5 days only the 50 µm3 particle concentration caused a significant increase in DNA damage (Fig. 6a). When astrocytes were cultured in isolation in 2D monolayer culture with CoCr particles, all particle concentrations caused significant DNA damage from 24 h onwards (Fig. 6b; p < 0.05 ANOVA and Tukey). Stainless steel particles compromised DNA integrity from 24 h, an effect that was dose dependent (Fig. 6c). When primary astrocytes were cultured in isolation with stainless steel particles, significant effects on DNA integrity were observed at 2 and 5 days with the 5 and 50 µm3 particle per cell concentrations (Fig. 6d; p < 0.05 ANOVA and Tukey).
The effect of increasing cobalt chrome and stainless steel particle volumes on the integrity of primary glial cell DNA over 5 days co-cultured in 2D culture, mean tail moment ± 95% confidence intervals, hydrogen peroxide was used as a positive control. a Effect of CoCr particles on astrocyte and microglial DNA in co-culture, b effect of CoCr particles on astrocyte DNA cultured in isolation, c effect of stainless steel particles on astrocyte and microglial DNA in co-culture, d effect of stainless steel particles on astrocyte DNA cultured in isolation. *Indicates a significant increase in the level of DNA damage expressed as a value for tail moment (p < 0.05) when compared with cell only negative control using two-way ANOVA. \(\bullet\) Indicates a significant decrease in the level of DNA damage expressed as a value for tail moment (p < 0.05) when compared with cell only negative control using two-way ANOVA
The effect of CoCr and stainless steel particles on primary astrocytes and microglia in 3D culture
A live/dead assay was used to determine the effect of wear particles on the viability of primary astrocytes and microglia in co-culture and primary astrocytes in isolation in a 3D collagen hydrogel cell culture system. Gels were imaged using fluorescence microscopy, and the percentage of living cells calculated. After 2 days in culture, 50 µm3 of CoCr particles adversely affected cell viability (Fig. 7a; p < 0.05 ANOVA plus Tukey). After 5 days in culture, both the 5 and 50 µm3 per cell of CoCr particles significantly reduced cell viability (Fig. 7a; p < 0.05, ANOVA and Tukey’s post hoc test). Significant effects on viability were observed when the primary astrocytes were cultured with only the 50 μm3 CoCr particle concentration after 2 and 5 days in culture (Fig. 7b; p < 0.05 ANOVA and Tukey’s post hoc test).
The effect of increasing volumes (0.5–50 μm3 per cell) of CoCr and stainless steel particles on the viability of primary glial cells cultured together and in isolation in 3D in culture over 5 days. Cell viability measured using a live/dead assay, mean percentage of living cells ± 95% confidence intervals. DMSO was used as a positive control. a Effect of CoCr particles on astrocyte and microglial viability in co-culture, b effect of CoCr particles on astrocyte viability cultured in isolation, c effect of stainless steel particles on astrocyte and microglial viability in co-culture, d effect of stainless steel particles on astrocyte viability cultured in isolation. *Indicates a significant reduction in viability (p < 0.05) when compared with cell only negative control using two-way ANOVA and Tukey’s post hoc test
No adverse effects on cell viability were observed when primary astrocytes and microglia were cultured together or in isolation with any concentration of stainless steel debris at any time point (Fig. 7c, d).
The effect of ions released from CoCr and stainless steel wear debris on the viability of primary astrocytes and microglia in 3D culture
There were no adverse effects on cell viability of CoCr ions at any concentration at the 48-h time point (Fig. 8a). However, a significant reduction in viability at the 5 µm3 per cell concentration of cobalt chrome ions was observed after 5 days in culture (Fig. 8a; p < 0.05 ANOVA and Tukey’s post hoc test). When primary astrocytes were cultured in isolation with CoCr ions, all doses affected cell viability significantly (Fig. 8b; p < 0.05, ANOVA and Tukey’s post hoc test) after 2 days. After 5 days, the ions released from 5 µm3 and 50 µm3 CoCr particles significantly affected cell viability (Fig. 8b). No adverse effects were observed when primary astrocytes and microglia were cultured in 3D co-culture with ions released from stainless steel particles at concentrations of 0.5 and 5 µm3 per cell (Fig. 8c). However, after 5 days in culture, ions from the highest concentration of stainless steel debris (50 µm3 per cell) adversely affected cell viability (Fig. 8c; p < 0.05 ANOVA and post hoc Tukey’s test). When astrocytes were cultured in isolation with ions from stainless steel particles, after 2 days only the 5 µm3 per cell concentration had a significant effect on cell viability; however, by day 5 all concentrations of ions adversely affected cell viability (Fig. 8d).
The effect of increasing concentrations of ions from CoCr and stainless steel wear particles (0.5–50 μm3) on the viability of primary glial cells in 3D co-culture and in isolation over 5 days. Cell viability measured using a live/dead assay, mean percentage of living cells ± 95% confidence intervals. DMSO was used as the positive control. a Effect of CoCr ions on astrocyte and microglial viability in co-culture, b effect of CoCr ions on astrocyte vibility cultured in isolation, c effect of stainless steel ions on astrocyte and microglial viabilty in co-culture, d effect of stainless steel ions on astrocyte viability cultured in isolation. *Indicates a significant reduction in viability (p < 0.05) when compared with cell only negative control using two-way ANOVA and post hoc Tukey’s test
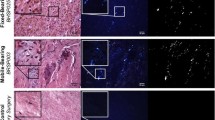
The effect of CoCr and stainless steel wear particles on the level of expression of glial fibrillary acidic protein (GFAP) in primary astrocytes in the presence and absence of microglia
When primary astrocytes were co-cultured with microglia in 3D culture with CoCr particles (0.5–50 μm3 per cell), the highest concentration of particles (50 μm3 per cell) resulted in increased expression of GFAP at both the 2- and 5-day time point (Table 1). However, lower concentrations of CoCr wear particles only caused increased expression of GFAP after 5 days. Stainless steel particles failed to increase GFAP expression to detectable levels in astrocytes at any particle concentration at any time point. When primary astrocytes were cultured in isolation in 3D culture with CoCr wear particles, the 5 and 0.5 μm3 per cell concentrations of particles increased the expression of GFAP after 5 days. Again, stainless steel particles did not cause increased expression of GFAP at any concentration or time point (Table 2).
Discussion
Characterisation of wear particles
The particles of CoCr generated in this study were similar in terms of size and morphology to previous studies [20, 22], and comparable to those generated in metal-on-metal total disc replacement devices [2]. The stainless steel particles were similar in morphology to the CoCr particles; however, their size range was larger, which is in line with the different material properties of the two metals, particularly with respect to chemistry, hardness and microstructure, e.g. grain size [23].
Effects of wear particles on cell viability and DNA integrity in 2D culture
When CoCr wear particles were cultured with primary rat astrocytes and microglial cells in 2D culture, cell viability was adversely affected by all doses of particles. This is in line with previous studies that exposed monocytes and fibroblasts to CoCr wear particles [20]. When cells were exposed to stainless steel particles in 2D culture, initially an adverse effect was observed; however, this effect was only consistent at the highest particle concentration at the 5-day time point. Other studies have observed variable cellular toxicity associated with stainless steel particles, with some cell types, e.g. monocytes adversely affected [24] and others not affected, e.g. osteoblasts [25]. The different effects are postulated to be due to different sensitivities of cell lines and different compositions of the two metals, with CoCr having higher chromium content than stainless steel (27–30% compared to 16–18%) and the absence of cobalt in stainless steel [26].
When glial cells were exposed to CoCr and SS particles and DNA integrity analysed, again there were differences between the two biomaterials. High doses of CoCr particles significantly affected DNA integrity of glial cells. In isolation, astrocytes were more sensitive to CoCr particles. These results suggested that when microglial cells were present, these cells influenced the effects that the particles had on the astrocyte cells, often with the microglial cells experiencing adverse effects in the form of DNA damage, whilst appearing to protect the astrocytes from these effects in the co-culture. After culture with SS particles, adverse effects on glial cell DNA integrity were observed after 24 h with all concentrations of particles. In contrast, astrocytes in isolation were not affected by stainless steel particles.
Effects of wear particles and ions on cell viability and cell reactivity in 3D culture
When primary astrocytes and microglia were cultured with increasing particle volumes of CoCr, a dose-dependent effect on cell viability was observed. When primary astrocytes were cultured in isolation with increasing particle volumes of CoCr, only the highest particle dose significantly reduced cell viability. Interestingly, the removal of microglia from the culture environment appeared to reduce the sensitivity of primary astrocytes to CoCr wear particles, suggesting the resident macrophage cell type, the microglia, influences the effects that the particles have on astrocytes. When cells were exposed to CoCr ions, the results suggested that astrocytes in isolation were more sensitive than when glial cells were co-cultured. We postulate that this may be due to either active uptake (phagocytosis) of large volumes of particles into microglia, followed by the release of cytokines or signalling factors that trigger subsequent cell death in the astrocytes. When astrocytes were cultured in isolation with particles, uptake may occur via pinocytosis and hence be much slower, resulting in lower levels of toxicity [15, 16].
In contrast, there were no adverse effects on cell viability when glial cells were cultured with increasing particle volumes of stainless steel at any particle dose, or at any time point. These results echo the results in the 2D culture system, indicating that differences in elemental composition between the two biomaterials may be responsible for the different effects on cell viability. In contrast, adverse effects on cell viability were observed when glial cells were exposed to ions from stainless steel particles, particularly when astrocytes were cultured in isolation, indicating that in the absence of microglia, astrocytes were more sensitive to ions from implant biomaterials.
Upregulation of the expression of glial fibrillary acidic protein (GFAP) to a variety of stimulants including injury [21] and biomaterial particles [27] has been widely reported in the literature. CoCr particles caused increased expression of GFAP at low particle concentrations by the 5-day time point; however, often the higher concentrations of particles did not. We postulate that these higher particle concentrations caused significant reductions in cell viability, and with a large proportion of the cells dead or dying, this masked the detection of GFAP. Stainless steel particles had no effect on the GFAP expression in glial cells at any dose or any time point.
In summary, CoCr particles adversely affected glial cell viability and triggered increased GFAP expression, indicating that astrocytes developed a reactive phenotype and were subjected to significant DNA damage. Cobalt chromium ions also had adverse effects on cell viability. In contrast, stainless steel particles rarely affected cell viability and did not elicit increased expression of GFAP. However, SS particles caused significant DNA damage and glial cells appeared to be more sensitive to stainless steel ions compared to CoCr ions, where the former were found to adversely affect cell viability at low concentrations and early time points. The results indicated that DNA damage was the result of interaction with ionic species rather than the particles themselves.
The particles used in the present study were generated using a simple configuration pin-on-plate wear simulator using water as a lubricant. In a previous study [2], the biocompatibility of CoCr wear particles generated from CoCr alloy on CCr alloy TDRs in serum and water was compared with particles generated in water using the same pin-on-plate set-up used in the present study. In L929 fibroblast cells, Pasko [2] found that there was no difference in the viability of L929 cells exposed to the different types of particles, with higher volumes of particles (5 and 50 µm3 of particles per cell) causing adverse effects on cell viability in a similar way that CoCr particles affected neural cells in the present study. The use of particles from joint simulators using serum lubricants causes issues with microbial contamination, endotoxin contamination and degraded proteins, which all affect the cellular responses, making it difficult to determine the effects of the particles alone. Simple configuration pin-on-plate simulators provide a quick, cost-effective way to generate large volumes of debris for cell culture easily avoiding these issues.
This study has revealed for the first time that clinically relevant wear particles from metallic biomaterials have numerous adverse effects on the cells of the spinal cord, and caution should be exercised when using these materials in spinal devices and instrumentation.
References
BBC News (2012) http://www.bbc.co.uk/news/health-17337993. Accessed 15 Apr 2014
Pasko K (2016) Ceramic coatings for cervical total disc replacement. Ph.D. thesis, University of Leeds
Pare PE, Chan FW, Powell ML et al (2007) Wear characteristics of the A-MAV anterior motion replacement using a spine wear simulator. Wear 263:1055–1059
Kurtz SM, Ciccarelli L, Siskey R et al (2012) Comparison of in vivo and simulator retrieved metal-on-metal cervical disc replacements. Int J Spine Surg 6:145–156
Guyer RD, Shellock J, MacLennan B et al (2013) Early failure of metal-on-metal artificial disc prostheses associated with lymphocytic reaction: diagnosis and treatment experience in four cases. Spine 36:E492–E497
Mody DR, Esses SI, Heggeness MH (1994) A histologic study of soft tissue reactions to spinal implants. Spine 19:1153–1156
Takahashi S, Delecrin J, Passuti N (2001) Intraspinal metallosis causing delayed neurologic symptoms aster spinal instrumentation surgery. Spine 26:1495–1499
Gaine WJ, Andrew SM, Chadwick P et al (2001) Late operative site pain with isola posterior instrumentation requiring implant removal. Spine 26:583–587
Senaran H, Atilla P, Kaymaz F et al (2004) Ultrastructural analysis of metallic debris and tissue reaction around spinal implants in patients with late operative site pain. Spine 29:1618–1623
Cunningham BW, Orbegoso CM, Dmitriev AE et al (2002) The effect of titanium particulate on development and maintenance of a posterolateral spine arthrodesis. Spine 27:1971–1981
Cunningham BW, Hallab NJ, Hu N et al (2013) Epidural application of spinal instrumentation particulate wear debris: a comprehensive evaluation of neurotoxicity using an in vivo animal model. J Neurosurg Spine 19:336–350
Wang JC, Yu WD, Sandhu HS et al (2002) Metal debris from titanium spinal implants. Spine 24:899–903
Yoshihara H (2013) Rods in spinal surgery: a review of the literature. Spine J 13:1350–1358
Papageorgiou I, Marsh L, Tipper JL et al (2014) Interaction of micron and nano-sized particles with cells of the dura mater. J Biomed Mater Res Part B 102B:1496–1505
Behl B, Papageorgiou I, Brown C et al (2014) Biological effects of cobalt chromium particles and ions on dural fibroblasts and dural epithelial cells. Biomaterials 34:3547–3558
Papageorgiou I, Abberton T, Fuller M et al (2014) Biological effects of clinically relevant CoCr nanoparticles in the dura mater: an organ culture study. Nanomaterials 4:485–504
Chang B-S, Brown PR, Sieber A et al (2004) Evaluation of the biological response of wear debris. Spine J 4:S239–S244
Stoodley MA, Jones NR, Brown CJ (1996) Evidence for rapid fluid flow from the subarachnoid space into the spinal cord central canal in the rat. Brain Res 707:155–164
Stoodley MA, Brown SA, Brown CJ et al (1997) Arterial pulsation-dependent perivascular cerebrospinal fluid flow into the central canal in the sheep spinal cord. J Neurosurg 86:686–693
Germain M, Hatton A, Williams S et al (2003) Comparison of the cytotoxicity of clinically relevant cobalt–chromium and alumina ceramic wear particles in vitro. Biomaterials 24:469–479
East E, Golding JP, Phillips JB (2009) A versatile 3D culture model facilitates monitoring of astrocytes undergoing reactive gliosis. J Tissue Eng Regen Med 3:634–646
Lal S, Caseley EA, Hall RM et al (2018) Biological impact of silicon nitride for orthopaedic applications: role of particle size, surface composition and donor variation. Sci Rep 8:9109. https://doi.org/10.1038/s41598-018-27494-y
Chou YK, Evans CJ (1997) Tool wear mechanism in continuous cutting of hardened tool steels. Wear 212:59–65
Bailey LO, Lipiatt S, Biancanello FS et al (2005) The quantification of cellular viability and inflammatory response to stainless steel alloys. Biomaterials 26:5296–5302
Li M, Yin T, Wang Y et al (2014) Study of biocompatibility of medical grade high nitrogen nickel free austenitic stainless steel in vitro. Mater Sci Eng C 43:641–648
Kanaji A, Orhue V, Caicedo MS et al (2014) Cytotoxic effect of cobalt and nickel ions on osteocytes in vitro. J Orthop Surg Res 9:1–8
DeGuzman RC, VandeVord PJ (2007) Variations in astrocyte and fibroblast response due to biomaterial particulates in vitro. J Biomed Mater Res Part A 85:14–24
Acknowledgements
The research was funded by the Engineering and Physical Sciences Research Council, UK, through a Ph.D. scholarship to HL through the Centre for Doctoral Training Medical and Biological Engineering, School of Mechanical Engineering, University of Leeds, UK.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Online Resources 1.
High-resolution scanning electron micrograph and corresponding trace produced by energy-dispersive X-ray analysis (EDX) of cobalt chromium particles (PPTX 717 kb)
Online Resources 2.
High-resolution scanning electron micrograph and corresponding trace produced by energy-dispersive X-ray analysis (EDX) of stainless steel particles (PPTX 64 kb)
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
About this article
Cite this article
Lee, H., Phillips, J.B., Hall, R.M. et al. Neural cell responses to wear debris from metal-on-metal total disc replacements. Eur Spine J 29, 2701–2712 (2020). https://doi.org/10.1007/s00586-019-06177-w
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00586-019-06177-w