Abstract
Purpose
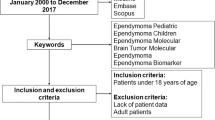
Although ependymomas occur in both the brain and the spine, the prognosis is quite varied by tumor location. Spinal ependymomas usually follow a relatively benign course with more favorable prognosis than that of the intracranial ependymomas. The aim of this study is to evaluate the genetic differences between spinal ependymomas and their intracranial counterparts using a meta-analysis.
Methods
We searched PubMed, Embase, Web of Science, and the Cochrane library. Comparative or single arm genetic studies that enrolled patients with both intracranial and spinal ependymoma were included. The frequency of genetic aberration was calculated in each group. We calculated the odds ratio (OR) with 95 % confidence intervals (CIs) for direct comparative studies and the logit event rate (LER) and 95 % CI for single arm studies.
Results
Twenty-five studies comprising of 380 spinal ependymomas and 964 intracranial ependymomas were compared to determine the association of the genetic differences of ependymomas at different locations. There were 25 comparable genetic aberrations between spinal and intracranial ependymomas. Among the genes, the NF2 mutation was significantly associated with the spinal ependymomas rather than with the intracranial ependymomas (spinal tumor: LER −0.750, 95 % CI −1.233 to −0.266, intracranial tumor: LER −3.080, 95 % CI −3.983 to −2.177). Intracranial ependymomas were found to be significantly associated with EPB41L3 deletion (OR 0.34; 95 % CI 0.14–0.80) and HIC1 methylation (OR 0.12; 95 % CI 0.02–0.68).
Conclusion
The genetic aberrations of spinal ependymomas are quite different from those of intracranial ependymomas. The difference in prognosis of ependymoma by location may be associated with genetic difference. A more detailed understanding of them may enable the development of targeted therapy and the estimation of prognosis.





Similar content being viewed by others
References
Wani K, Armstrong TS, Vera-Bolanos E, Raghunathan A, Ellison D, Gilbertson R, Vaillant B, Goldman S, Packer RJ, Fouladi M, Pollack I, Mikkelsen T, Prados M, Omuro A, Soffietti R, Ledoux A, Wilson C, Long LH, Gilbert MR, Aldape K, Collaborative Ependymoma R (2012) A prognostic gene expression signature in infratentorial ependymoma. Acta Neuropathol 123:727–738
Vera-Bolanos E, Aldape K, Yuan Y, Wu J, Wani K, Necesito-Reyes MJ, Colman H, Dhall G, Lieberman FS, Metellus P, Mikkelsen T, Omuro A, Partap S, Prados M, Robins HI, Soffietti R, Wu J, Gilbert MR, Armstrong TS, Foundation C (2015) Clinical course and progression-free survival of adult intracranial and spinal ependymoma patients. Neuro Oncol 17:440–447
Modena P, Lualdi E, Facchinetti F, Veltman J, Reid JF, Minardi S, Janssen I, Giangaspero F, Forni M, Finocchiaro G, Genitori L, Giordano F, Riccardi R, Schoenmakers E, Massimino M, Sozzi G (2006) Identification of tumor-specific molecular signatures in intracranial ependymoma and association with clinical characteristics. J Clin Oncol 24:5223–5233
Taylor MD, Poppleton H, Fuller C, Su XP, Liu YX, Jensen P, Magdaleno S, Dalton J, Calabrese C, Board J, MacDonald T, Rutka J, Guha A, Gajjar A, Curran T, Gilbertson RJ (2005) Radial glia cells are candidate stem cells of ependymoma. Cancer Cell 8:323–335
Puget S, Grill J, Valent A, Bieche I, Dantas-Barbosa C, Kauffmann A, Dessen P, Lacroix L, Geoerger B, Job B, Dirven C, Varlet P, Peyre M, Dirks PB, Sainte-Rose C, Vassal G (2009) Candidate genes on chromosome 9q33–34 involved in the progression of childhood ependymomas. J Clin Oncol 27:1884–1892
Korshunov A, Witt H, Hielscher T, Benner A, Remke M, Ryzhova M, Milde T, Bender S, Wittmann A, Schottler A, Kulozik AE, Witt O, von Deimling A, Lichter P, Pfister S (2010) Molecular staging of intracranial ependymoma in children and adults. J Clin Oncol 28:3182–3190
Pajtler KW, Witt H, Sill M, Jones DTW, Hovestadt V, Kratochwil F, Wani K, Tatevossian R, Punchihewa C, Johann P, Reimand J, Warnatz HJ, Ryzhova M, Mack S, Ramaswamy V, Capper D, Schweizer L, Sieber L, Wittmann A, Huang Z, van Sluis P, Volckmann R, Koster J, Versteeg R, Fults D, Toledano H, Avigad S, Hoffman LM, Donson AM, Foreman N, Hewer E, Zitterbart K, Gilbert M, Armstrong TS, Gupta N, Allen JC, Karajannis MA, Zagzag D, Hasselblatt M, Kulozik AE, Witt O, Collins VP, von Hoff K, Rutkowski S, Pietsch T, Bader G, Yaspo ML, von Deimling A, Lichter P, Taylor MD, Gilbertson R, Ellison DW, Aldape K, Korshunov A, Kool M, Pfister SM (2015) Molecular classification of ependymal tumors across all CNS compartments, histopathological grades, and age groups. Cancer Cell 27:728–743
Huang B, Starostik P, Schraut H, Krauss J, Sorensen N, Roggendorf W (2003) Human ependymomas reveal frequent deletions on chromosomes 6 and 9. Acta Neuropathol 106:357–362
Scheil S, Bruderlein S, Eicker M, Herms J, Herold-Mende C, Steiner HH, Barth TFE, Moller P (2001) Low frequency of chromosomal imbalances in anaplastic ependymomas as detected by comparative genomic hybridization. Brain Pathol 11:133–143
Ward S, Harding B, Wilkins P, Harkness W, Hayward R, Darling JL, Thomas DGT, Warr T (2001) Gain of 1q and loss of 22 are the most common changes detected by comparative genomic hybridisation in paediatric ependymoma. Genes Chromosomes Cancer 32:59–66
Zheng PP, Pang JC, Hui AB, Ng HK (2000) Comparative genomic hybridization detects losses of chromosomes 22 and 16 as the most common recurrent genetic alterations in primary ependymomas. Cancer Genet Cytogenet 122:18–25
Witt H, Mack SC, Ryzhova M, Bender S, Sill M, Isserlin R, Benner A, Hielscher T, Milde T, Remke M, Jones DT, Northcott PA, Garzia L, Bertrand KC, Wittmann A, Yao Y, Roberts SS, Massimi L, Van Meter T, Weiss WA, Gupta N, Grajkowska W, Lach B, Cho YJ, von Deimling A, Kulozik AE, Witt O, Bader GD, Hawkins CE, Tabori U, Guha A, Rutka JT, Lichter P, Korshunov A, Taylor MD, Pfister SM (2011) Delineation of two clinically and molecularly distinct subgroups of posterior fossa ependymoma. Cancer Cell 20:143–157
Johnson RA, Wright KD, Poppleton H, Mohankumar KM, Finkelstein D, Pounds SB, Rand V, Leary SES, White E, Eden C, Hogg T, Northcott P, Mack S, Neale G, Wang YD, Coyle B, Atkinson J, DeWire M, Kranenburg TA, Gillespie Y, Allen JC, Merchant T, Boop FA, Sanford RA, Gajjar A, Ellison DW, Taylor MD, Grundy RG, Gilbertson RJ (2010) Cross-species genomics matches driver mutations and cell compartments to model ependymoma. Nature 466:632–636
Ebert C, von Haken M, Meyer-Puttlitz B, Wiestler OD, Reifenberger G, Pietsch T, von Deimling A (1999) Molecular genetic analysis of ependymal tumors. NF2 mutations and chromosome 22q loss occur preferentially in intramedullary spinal ependymomas. Am J Pathol 155:627–632
Singh PK, Gutmann DH, Fuller CE, Newsham IF, Perry A (2002) Differential involvement of protein 4.1 family members DAL-1 and NF2 in intracranial and intraspinal ependymomas. Mod Pathol 15:526–531
Zadnik PL, Gokaslan ZL, Burger PC, Bettegowda C (2013) Spinal cord tumours: advances in genetics and their implications for treatment. Nat Rev Neurol 9:257–266
Kilday JP, Rahman R, Dyer S, Ridley L, Lowe J, Coyle B, Grundy R (2009) Pediatric ependymoma: biological perspectives. Mol Cancer Res 7:765–786
Andreiuolo F, Ferreira C, Puget S, Grill J (2013) Current and evolving knowledge of prognostic factors for pediatric ependymomas. Future Oncol 9:183–191
Parker M, Mohankumar KM, Punchihewa C, Weinlich R, Dalton JD, Li YJ, Lee R, Tatevossian RG, Phoenix TN, Thiruvenkatam R, White E, Tang B, Orisme W, Gupta K, Rusch M, Chen X, Li YX, Nagahawhatte P, Hedlund E, Finkelstein D, Wu G, Shurtleff S, Easton J, Boggs K, Yergeau D, Vadodaria B, Mulder HL, Becksford J, Gupta P, Huether R, Ma J, Song GC, Gajjar A, Merchant T, Boop F, Smith AA, Ding L, Lu C, Ochoa K, Zhao D, Fulton RS, Fulton LL, Mardis ER, Wilson RK, Downing JR, Green DR, Zhang JH, Ellison DW, Gilbertson RJ (2014) C11orf95-RELA fusions drive oncogenic NF-kappa B signalling in ependymoma. Nature 506:451–455
Rajaram V, Leuthardt EC, Singh PK, Ojemann JG, Brat DJ, Prayson RA, Perry A (2003) 9p21 and 13q14 dosages in ependymomas. A clinicopathologic study of 101 cases. Mod Pathol 17:9–14
Bettegowda C, Agrawal N, Jiao YC, Wang YX, Wood LD, Rodriguez FJ, Hruban RH, Gallia GL, Binder ZA, Riggins CJ, Salmasi V, Riggins GJ, Reitman ZJ, Rasheed A, Keir S, Shinjo S, Marie S, McLendon R, Jallo G, Vogelstein B, Bigner D, Yan H, Kinzler KW, Papadopoulos N (2013) Exomic sequencing of four rare central nervous system tumor types. Oncotarget 4:572–583
Barton VN, Donson AM, Kleinschmidt-DeMasters BK, Birks DK, Handler MH, Foreman NK (2010) Unique molecular characteristics of pediatric myxopapillary ependymoma. Brain Pathol 20:560–570
Stephen JH, Sievert AJ, Madsen PJ, Judkins AR, Resnick AC, Storm PB, Rushing EJ, Santi M (2012) Spinal cord ependymomas and myxopapillary ependymomas in the first 2 decades of life: a clinicopathological and immunohistochemical characterization of 19 cases. J Neurosurg Pediatrics 9:646–653
Karakoula K, Jacques TS, Phipps KP, Harkness W, Thompson D, Harding BN, Darling JL, Warr TJ (2014) Epigenetic genome-wide analysis identifies BEX1 as a candidate tumour suppressor gene in paediatric intracranial ependymoma. Cancer Lett 346:34–44
Rajaram V, Gutmann DH, Prasad SK, Mansur DB, Perry A (2005) Alterations of protein 4.1 family members in ependymomas: a study of 84 cases. Mod Pathol 18:991–997
Alonso ME, Bello MJ, Gonzalez-Gomez P, Arjona D, de Campos JM, Gutierrez M, Rey JA (2004) Aberrant CpG island methylation of multiple genes in ependymal tumors. J Neurooncol 67:159–165
Andreiuolo F, Puget S, Peyre M, Dantas-Barbosa C, Boddaert N, Philippe C, Mauguen A, Grill J, Varlet P (2010) Neuronal differentiation distinguishes supratentorial and infratentorial childhood ependymomas. Neuro Oncol 12:1126–1134
Athanasiou A, Perunovic B, Quilty RD, Gorgoulis VG, Kittas C, Love S (2003) Expression of mos in ependymal gliomas. Am J Clin Pathol 120:699–705
Bortolotto S, Chiado-Piat L, Cavalla P, Bosone I, Mauro A, Schiffer D (2001) CDKN2A/p16 in ependymomas. J Neurooncol 54:9–13
Gilbertson RJ, Bentley L, Hernan R, Junttila TT, Frank AJ, Haapasalo H, Connelly M, Wetmore C, Curran T, Elenius K, Ellison DW (2002) ERBB receptor signaling promotes ependymoma cell proliferation and represents a potential novel therapeutic target for this disease. Clin Cancer Res 8:3054–3064
Hamilton DW, Lusher ME, Lindsey JC, Ellison DW, Clifford SC (2005) Epigenetic inactivation of the RASSF1A tumour suppressor gene in ependymoma. Cancer Lett 227:75–81
Kraus JA, de Millas W, Sorensen N, Herbold C, Schichor C, Tonn JC, Wiestler OD, von Deimling A, Pietsch T (2001) Indications for a tumor suppressor gene at 22q11 involved in the pathogenesis of ependymal tumors and distinct from hSNF5/INI1. Acta Neuropathol 102:69–74
Lamszus K, Lachenmayer L, Heinemann U, Kluwe L, Finckh U, Hoppner W, Stavrou D, Fillbrandt R, Westphal M (2001) Molecular genetic alterations on chromosomes 11 and 22 in ependymomas. Int J Cancer 91:803–808
Magrassi L, Marziliano N, Inzani F, Cassini P, Chiaranda I, Skrap M, Pizzolito S, Arienta C, Arbustini E (2010) EDG3 and SHC3 on chromosome 9q22 are co-amplified in human ependymomas. Cancer Lett 290:36–42
Mendrzyk F, Korshunov A, Benner A, Toedt G, Pfister S, Radlwimmer B, Lichter P (2006) Identification of gains on 1q and epidermal growth factor receptor overexpression as independent prognostic markers in intracranial ependymoma. Clin Cancer Res 12:2070–2079
Michalowski MB, de Fraipont F, Michelland S, Entz-Werle N, Grill J, Pasquier B, Favrot MC, Plantaz D (2006) Methylation of RASSFIA and TRAIL pathway-related genes is frequent in childhood intracranial ependymornas and benign choroid plexus papilloma. Cancer Genet Cytogenet 166:74–81
Modena P, Buttarelli FR, Miceli R, Piccinin E, Baldi C, Antonelli M, Morra I, Lauriola L, Di Rocco C, Garre ML, Sardi I, Genitori L, Maestro R, Gandola L, Facchinetti F, Collini P, Sozzi G, Giangaspero F, Massimino M (2012) Predictors of outcome in an AIEOP series of childhood ependymomas: a multifactorial analysis. Neuro Oncol 14:1346–1356
Rousseau E, Ruchoux MM, Scaravilli F, Chapon F, Vinchon M, De Smet C, Godfraind C, Vikkula M (2003) CDKN2A, CDKN2B and p14(ARF) are frequently and differentially methylated in ependymal tumours. Neuropathol Appl Neurobiol 29:574–583
Waha A, Koch A, Hartmann W, Mack H, Schramm J, Sorensen N, Berthold F, Wiestler OD, Pietsch T, Waha A (2004) Analysis of HIC-1 methylation and transcription in human ependymomas. Int J Cancer 110:542–549
Gupta RK, Sharma MC, Suri V, Kakkar A, Singh M, Sarkar C (2014) Study of chromosome 9q gain, Notch pathway regulators and Tenascin-C in ependymomas. J Neurooncol 116:267–274
Suzuki SO, Iwaki T (2000) Amplification and overexpression of mdm2 gene in ependymomas. Mod Pathol 13:548–553
Ostrom QT, Gittleman H, Fulop J, Liu M, Blanda R, Kromer C, Wolinsky Y, Kruchko C, Barnholtz-Sloan JS (2015) CBTRUS statistical report: primary brain and central nervous system tumors diagnosed in the United States in 2008–2012. Neuro Oncol 17 Suppl 4:iv1–iv62
Rubio MP, Correa KM, Ramesh V, MacCollin MM, Jacoby LB, Von Deimling A, Gusella JF, Louis DN (1994) Analysis of the neurofibromatosis 2 gene in human ependymomas and astrocytomas. Cancer Res 54:45–47
Garcia C, Gutmann DH (2014) Nf2/Merlin controls spinal cord neural progenitor function in a Rac1/ErbB2-dependent manner. PLoS One 9:e97320
Lee CH, Chung CK, Ohn JH, Kim CH (2016) The similarities and differences between intracranial and spinal ependymomas: a review from a genetic research perspective. J Korean Neurosurg Soc 59:83–90
Yang I, Nagasawa DT, Kim W, Spasic M, Trang A, Lu DC, Martin NA (2012) Chromosomal anomalies and prognostic markers for intracranial and spinal ependymomas. J Clin Neurosci 19:779–785
Yao Y, Mack SC, Taylor MD (2011) Molecular genetics of ependymoma. Chin J Cancer 30:669–681
Hanemann CO (2008) Magic but treatable? Tumours due to loss of merlin. Brain 131:606–615
Fouladi M, Stewart CF, Blaney SM, Onar-Thomas A, Schaiquevich P, Packer RJ, Gajjar A, Kun LE, Boyett JM, Gilbertson RJ (2010) Phase I trial of lapatinib in children with refractory CNS malignancies: a Pediatric Brain Tumor Consortium study. J Clin Oncol 28:4221–4227
DeWire M, Fouladi M, Turner DC, Wetmore C, Hawkins C, Jacobs C, Yuan Y, Liu D, Goldman S, Fisher P, Rytting M, Bouffet E, Khakoo Y, Hwang EI, Foreman N, Stewart CF, Gilbert MR, Gilbertson R, Gajjar A (2015) An open-label, two-stage, phase II study of bevacizumab and lapatinib in children with recurrent or refractory ependymoma: a collaborative ependymoma research network study (CERN). J Neurooncol 123:85–91
Daly AK, Day CP (2001) Candidate gene case-control association studies: advantages and potential pitfalls. Br J Clin Pharmacol 52:489–499
Thornton A, Lee P (2000) Publication bias in meta-analysis: its causes and consequences. J Clin Epidemiol 53:207–216
Acknowledgments
This work was supported by the National Research Foundation of Korea (NRF) Grant funded by the Korea government (MSIP) (No. 2010-0028631). The sponsor had no role in the design or conduct of this research.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflicts of interest
None of the authors has any financial interest in the subject under discussion in this paper.
Rights and permissions
About this article
Cite this article
Lee, CH., Chung, C.K. & Kim, C.H. Genetic differences on intracranial versus spinal cord ependymal tumors: a meta-analysis of genetic researches. Eur Spine J 25, 3942–3951 (2016). https://doi.org/10.1007/s00586-016-4745-4
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00586-016-4745-4