Abstract
Purpose
To evaluate the clinical and radiographic outcomes following total disc arthroplasty using the XL TDR® Lumbar Disc in the treatment of patients with symptomatic degenerative disc disease at one level between L1–2 and L4–5.
Methods
Data were compiled from two centers participating in a prospective, multi-center Food and Drug Administration-approved investigational device exemption clinical trial enrolling patients with single-level lumbar degenerative disc disease unresponsive to non-operative treatment. Longitudinal outcomes were evaluated through 3-year follow-up and included patient-reported pain, function, and general health, as well as radiographic measures such as maintenance of disc height and range of motion.
Results
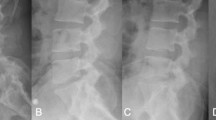
The two-center cohort included 64 treated patients, 42 % female, averaging 45.3 years of age (range 26–67). The majority of procedures were performed at the L4–5 level (75 %), uncomplicated, with minimal blood loss (88 % 0–50 cc), and in an outpatient setting (93.8 %). Postoperative events included 10 patients (15.6 %) with new hip flexion weakness, 7 (10.9 %) with new lower extremity weakness, and 10 (15.6 %) with new lower extremity sensory deficits, all resolved by 3 months in all but two patients whose deficits were prolonged but eventually resolved. Average disc height increased postoperatively from 7.2 to 12.1 mm (69 %), and was 10.7 mm (49 % increase from preoperative) at 3 years. Flexion/extension range of motion averaged 5.9° (SD 4.8°) at 3 years, and was not statistically different from preoperative (p = 0.471). Heterotopic ossification interfering with segmental motion was noted in 3 patients (10.3 %) at 3 years, none ankylosed (Grade IV). Postoperative improvement in patient-reported outcomes was significant (p < 0.01 for all measures) and maintained through 3-year follow-up. Satisfaction with results was reported by 85 % (51/60) of patients at 2 years and 93 % (28/30) at 3 years. There were no revisions through 3 years postoperative.
Conclusions
The results following XL TDR show good clinical and radiographic outcomes out to 3 years postoperative, with clinically significant improvements in pain, function, and general health, few complications, and high patient satisfaction.






Similar content being viewed by others
References
Boselie TF, Willems PC, van MH, de Bie RA, Benzel EC, van SH (2013) Arthroplasty versus fusion in single-level cervical degenerative disc disease: a Cochrane review. Spine 38:E1096–E1107
Jacobs WC, van der Gaag NA, Kruyt MC et al (2013) Total disc replacement for chronic discogenic low back pain: a Cochrane review. Spine 38:24–36
Blumenthal S, McAfee PC, Guyer RD et al (2005) A prospective, randomized, multicenter Food and Drug Administration investigational device exemptions study of lumbar total disc replacement with the CHARITE artificial disc versus lumbar fusion: part I: evaluation of clinical outcomes. Spine 30:1565–1575
Delamarter R, Zigler JE, Balderston RA, Cammisa FP, Goldstein JA, Spivak JM (2011) Prospective, randomized, multicenter Food and Drug Administration investigational device exemption study of the ProDisc-L total disc replacement compared with circumferential arthrodesis for the treatment of two-level lumbar degenerative disc disease: results at twenty-four months. J Bone Joint Surg Am 93:705–715
Gornet MF, Burkus JK, Dryer RF, Peloza JH (2011) Lumbar disc arthroplasty with MAVERICK disc versus stand-alone interbody fusion: a prospective, randomized, controlled, multicenter investigational device exemption trial. Spine 36:E1600–E1611
Zigler J, Delamarter R, Spivak JM et al (2007) Results of the prospective, randomized, multicenter Food and Drug Administration investigational device exemption study of the ProDisc-L total disc replacement versus circumferential fusion for the treatment of 1-level degenerative disc disease. Spine 32:1155–1162
Patel VC, Park DK, Herkowitz HN (2012) Lateral transpsoas fusion: indications and outcomes. ScientificWorldJournal 2012:893608
Pimenta L, Diaz RC, Guerrero LG (2006) Charite lumbar artificial disc retrieval: use of a lateral minimally invasive technique. Technical note. J Neurosurg Spine 5:556–561
Pimenta L, Oliveira L, Schaffa T, Coutinho E, Marchi L (2011) Lumbar total disc replacement from an extreme lateral approach: clinical experience with a minimum of 2 years’ follow-up. J Neurosurg Spine 14:38–45
Ozgur BM, Aryan HE, Pimenta L, Taylor WR (2006) Extreme Lateral Interbody Fusion (XLIF): a novel surgical technique for anterior lumbar interbody fusion. Spine J 6:435–443
Peterson MD, Youssef JA (2013) Extreme Lateral Interbody Fusion (XLIF): Lumbar Surgical Technique. In: Goodrich J, Volcan I (eds) Extreme Lateral Interbody Fusion (XLIF), 2nd edn. QMP, St. Louis, pp 159–178
Cleaver N, Afshar H, Oliveira L et al (2013) Lateral Total Disc Replacement (XL TDR). In: Goodrich JA, Volcan IJ (eds) eXtreme Lateral Interbody Fusion (XLIF), 2nd edn. QMP, St. Louis, pp 341–353
Pimenta L, Schaffa TD, Lhamby JT et al (2008) Total Disc Replacement Through the XLIF Approach. In: Goodrich JA, Volcan IJ (eds) eXtreme Lateral Interbody Fusion (XLIF). QMP, St. Louis, pp 275–283
Murtagh R, Castellvi AE (2014) Motion preservation surgery in the spine. Neuroimaging Clin N Am 24:287–294
Bertagnoli R, Zigler J, Karg A, Voigt S (2005) Complications and strategies for revision surgery in total disc replacement. Orthop Clin North Am 36:389–395
Leary SP, Regan JJ, Lanman TH, Wagner WH (2007) Revision and explantation strategies involving the CHARITE lumbar artificial disc replacement. Spine 32:1001–1011
McAfee PC, Geisler FH, Saiedy SS et al (2006) Revisability of the CHARITE artificial disc replacement: analysis of 688 patients enrolled in the U.S. IDE study of the CHARITE Artificial Disc. Spine 31:1217–1226
Patel AA, Brodke DS, Pimenta L et al (2008) Revision strategies in lumbar total disc arthroplasty. Spine 33:1276–1283
Scott-Young M (2005) Strategy for revision disc replacement surgery. Roundtables in Spine Surgery 1:23–72
van Ooij A, Oner FC, Verbout AJ (2003) Complications of artificial disc replacement: a report of 27 patients with the SB Charite disc. J Spinal Disord Tech 16:369–383
Wagner WH, Regan JJ, Leary SP et al (2006) Access strategies for revision or explantation of the Charite lumbar artificial disc replacement. J Vasc Surg 44:1266–1272
Marchi L, Oliveira L, Coutinho E, Pimenta L (2012) The importance of the anterior longitudinal ligament in lumbar disc arthroplasty: 36-month follow-up experience in extreme lateral total disc replacement. Int J Spine Surg 6:18–23
Dooris AP, Goel VK, Grosland NM, Gilbertson LG, Wilder DG (2001) Load-sharing between anterior and posterior elements in a lumbar motion segment implanted with an artificial disc. Spine 26:E122–E129
Rundell SA, Auerbach JD, Balderston RA, Kurtz SM (2008) Total disc replacement positioning affects facet contact forces and vertebral body strains. Spine 33:2510–2517
White A, Panjabi M (2001) Clinical Biomechanics of the Spine, 2nd edn. JB Lippincott, Philadelphia
Rajaraman V, Vingan R, Roth P, Heary RF, Conklin L, Jacobs GB (1999) Visceral and vascular complications resulting from anterior lumbar interbody fusion. J Neurosurg 91:60–64
Le Huec JC, Lafage V, Bonnet X et al (2010) Validated finite element analysis of the maverick total disc prosthesis. J Spinal Disord Tech 23:249–257
Lemaire JP, Carrier H, Sariali E, Skalli W, Lavaste F (2005) Clinical and radiological outcomes with the Charite artificial disc: a 10-year minimum follow-up. J Spinal Disord Tech 18:353–359
McAfee PC, Cunningham B, Holsapple G et al (2005) A prospective, randomized, multicenter Food and Drug Administration investigational device exemption study of lumbar total disc replacement with the CHARITE artificial disc versus lumbar fusion: part II: evaluation of radiographic outcomes and correlation of surgical technique accuracy with clinical outcomes. Spine 30:1576–1583
Schmidt H, Galbusera F, Rohlmann A, Zander T, Wilke HJ (2012) Effect of multilevel lumbar disc arthroplasty on spine kinematics and facet joint loads in flexion and extension: a finite element analysis. Eur Spine J 21(Suppl 5):S663–S674
Tournier C, Aunoble S, Le Huec JC et al (2007) Total disc arthroplasty: consequences for sagittal balance and lumbar spine movement. Eur Spine J 16:411–421
Cheng WK, Jadhav V, Palmer DK (2011) A novel modification for removal of the polyethylene core in artificial disc retrieval using a transpsoas minimally invasive technique. J Neurosurg Spine 14:466–469
McAfee PC, Cunningham BW, Devine J, Williams E, Yu-Yahiro J (2003) Classification of heterotopic ossification (HO) in artificial disk replacement. J Spinal Disord Tech 16:384–389
Tortolani PJ, Cunningham BW, Eng M, McAfee PC, Holsapple GA, Adams KA (2007) Prevalence of heterotopic ossification following total disc replacement. A prospective, randomized study of two hundred and seventy-six patients. J Bone Joint Surg Am 89:82–88
Tohmeh AG, Rodgers WB, Peterson MD (2011) Dynamically evoked, discrete-threshold electromyography in the extreme lateral interbody fusion approach. J Neurosurg Spine 14:31–37
Geisler FH, Blumenthal SL, Guyer RD et al (2004) Neurological complications of lumbar artificial disc replacement and comparison of clinical results with those related to lumbar arthrodesis in the literature: results of a multicenter, prospective, randomized investigational device exemption study of Charite intervertebral disc. Invited submission from the joint section meeting on disorders of the spine and peripheral nerves, March 2004. J Neurosurg Spine 1:143–154
Acknowledgments
The XL TDR US IDE clinical trial was funded by NuVasive, Inc. The authors would like to acknowledge NuVasive Clinical Affairs and Clinical Resources for their operational and administrative support of the XL TDR IDE trial and data.
Conflict of interest
Antoine Tohmeh and William Smith are both consultants of NuVasive, Inc.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Tohmeh, A.G., Smith, W.D. Lumbar total disc replacement by less invasive lateral approach: a report of results from two centers in the US IDE clinical trial of the XL TDR® device. Eur Spine J 24 (Suppl 3), 331–338 (2015). https://doi.org/10.1007/s00586-015-3843-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00586-015-3843-z