Abstract
Background
Attention has recently focused on mitochondrial-mediated apoptosis as a signaling pathway that is disrupted in many diseases involving excessive cell proliferation. Medicinal plants that induce apoptosis may be useful in chemoprevention. Cajanus cajan (C. cajan) is cultivated in East and West Africa. It is used in folk medicine for the treatment of liver injury, anemia, dysentery, measles, jaundice, and tumor.
Purpose
This study aims to investigate the in vitro antioxidant and mitochondrial-mediated apoptotic effects of C. cajan.
Methods
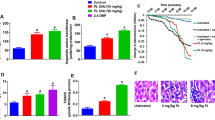
The mitochondrial permeability transition (mPT), mitochondrial ATPase activity, (2, 2-diphenylpicrylhydrazyl) DPPH radical-scavenging activity, total antioxidant capacity, and total phenol and apoptotic biomarkers were assessed while the phytochemicals present in the extract were determined by gas chromatography-mass spectrometry (GC–MS). Methanol extract of Cajanus cajan (MECC) triggered mitochondrial-mediated apoptosis by induction of pore opening, enhancement of mitochondrial ATPase activities, and increased immunohistochemical expression of caspase 3, caspase 9, and cytochrome c.
Results
MECC triggered mitochondrial-mediated apoptosis by induction of pore opening and increased expression of caspase 3, caspase 9, and cytochrome c. Enhancement of ATPase activities inhibition of lipid peroxidation and DPPH scavenging activity of DPPH were observed.
Conclusions
These findings suggest that C. cajan-induced mitochondrial-mediated apoptosis in normal rat liver through the induction of mPT and eventual release of cytochrome c which is a prelude to the progression of apoptosis. Hence, certain bioactive components of C. cajan may be useful in chemoprevention and management of conditions associated with insufficient apoptosis.












Similar content being viewed by others
References
Ajaiyeoba EO, Ogbole OO, Abiodun OO, Ashidi JS, Houghton PJ, Wright CW (2013) Cajachalcone: an antimalarial compound from Cajanus cajan leaf extract. J Parasitol Res 2:1–5
Ashidi JS, Houghton PJ, Hylands PJ, Efferth T (2010) Ethnobotanical survey and cytotoxicity testing of plants of South-western Nigeria used to treat cancer, with isolation of cytotoxic constituents from Cajanus cajan Millsp. Leaves J Ethnopharmacol 128:501–512
Benhar M (2020) Oxidants, antioxidants and thiol redox switches in the control of regulated cell death pathways. Antioxidants 9:309
Biasutto L, Azzolini M, Szabo I (1863) Zoratti M (2016) The mitochondrial permeability transition pore in AD 2016: An update. Biochim Biophys Acta Mol Cell Res 10:2515–2530
Brand-Williams W, Cuvelier ME, Berset C (1995) Use of free radical method to evaluate activity. Lebensm Wiss Technol 28:25–30
Burkhill HM (1978) Tropical Africa, 2nd Ed Vol 1 families A-D. Royal Botanic Gardens, Kew, Richmond, UK
Díaz-Quintana A, Pérez-Mejías G, Guerra-Castellano A, De La Rosa MA, Díaz-Moreno I (2020) Wheel and deal in the mitochondrial inner membranes: the tale of cytochrome c and cardiolipin. Oxid Med Cell Longev 2020:1–20
Efferth T, Li PC, Konkimalla VS, Kaina B (2007) From traditional Chinese medicine to rational cancer therapy.
Endlicher R, Drahota Z, Štefková K, Červinková Z, Kučera O (2023) The mitochondrial permeability transition pore—current knowledge of its structure, function, and regulation, and optimized methods for evaluating its functional state. Cells 12(9):1273
Fulda S (2010) Evasion of apoptosis as a cellular stress response in cancer. Int J of Cell Biol 2010:1–6. https://doi.org/10.1155/2010/370835
Fu YJ, Zu YG, Liu W, Efferth T, Zhang NJ, Liu XN, Kong Y (2006) Optimization of luteolin separation from pigeon pea [Cajanus cajan (L.) Millsp.] leaves by macroporous resins. J Chromatogr A 1137:145–152
Grover JK, Yadav S, Vats VJ (2002) Medicinal plants of India with antidiabetic potential. J Ethnopharmacol 81:81–100
Halliwell B, Gutteridge JMC (1998) The antioxidants of human extracellular fluids. Arch Biochem Biophys 280:1–8. https://doi.org/10.1016/0003-9861(90)90510-6. PMID: 2191627
Heddle JA, Hite M, Kirkhart B, Mavournin K, MacGregor JT, Newell GW, Salamone MF (1983) The induction of micronuclei as a measure of genotoxicity. A report of the U.S. Environmental Protection Agency Gene-Tox Program. Mutat Res. Sep; 123 (1):61–118. https://doi.org/10.1016/0165-1110(83)90047-7. PMID: 6888413
Iweala EEJ, Evbakhavbokun WO, Maduagwu EN (2019) Antioxidant and hepatoprotective effect of Cajanus cajan in N-nitrosodiethylamine-induced liver damage. Sci Pharm 87(3):24
Jaliwala YA, Mohanti PK, Jain N (2014) Antiulcer and anti-inflammatory activity of Cajanus cajan. Int Res J Pharm 5(4):313–316
Kundu R, Dasgupta S, Biswas A, Bhattacharya A, Pal BC, Bandyopadhyay D, Bhattacharya S, Bhattacharya S (2008) Cajanus cajan Linn. (Leguminosae) prevents alcohol-induced rats liver damage and augument cytoprotective function. J Ethnopharmacol 118:440–447
Kuranaga E, Matsunuma T, Kanuka H, Takemoto K, Koto A, Kimura K, Miura M (2011) Apoptosis the speed of looping morphogenesis in Drosophila male terminalia. Devel 138:1493–1499
Kerr JFR, Wyllie AH, Currie AR (1972) Apoptosis: a basic biological phenomenon with wide ranging implications in tissue kinetics. Br J Cancer 26:239–257
Lapidus RG, Sokolove PM (1993) Spermine inhibition of the permeability transition of isolated rat liver mitochondria: an investigation of mechanisms. Biochem Biophys J 64:246–253
Lee JY, Kim H, Song YS (2012) Genistein as a potential anticancer agent against ovarian cancer. J Tradit Complement Med 2:96–104
Lim HW, Lim HY, Wong KP (2009) Uncoupling of oxidative phosphorylation by curcumin: implication of its cellular mechanism of action. Biochem Biophys Res Commun 389(1):187–192
Liu Y, Bi T, Wang G, Dai W, Wu G, Qian L, Lupeol, (2015) inhibits proliferation and induces apoptosis of human pancreatic cancer PCNA-1 cells through AKT/ERK pathways, Naunyn Schmiedebergs Arch. Pharmacol 388:295–304
Lockshin RA, Zakeri Z (2001) Programmed cell death and apoptosis: origins of the theory. Nat Rev Mol Cell Biol 2:5455–5450
Lowry OH, Rosebrough NJ, Farr AL, Randall RJ (1951) Protein measurement with the Folin phenol reagent. J Biol Chem 193:262–275
Martin KR (2006) Targeting apoptosis with dietary bioactive agents. Exp Biol Med 231117–129
Musa HH, Musa TH, Oderinde O et al (2022) Traditional herbal medicine: overview of research indexed in the Scopus database. Adv Tradit Med
Nwaechefu OO, Olaolu TD, Akinwunmi IR, Ojezele OO, Olorunsogo OO (2022) Cajanus cajan ameliorated CCl4-induced oxidative stress in Wistar rats via the combined mechanisms of anti-inflammation and mitochondrial-membrane transition pore inhibition. J Ethnopharmacol 10:114920
Olanlokun OJ, Lawal OS, Olorunsogo OO (2020) Modulatory effects of ethyl acetate and methanol fractions of the stem bark extract of Alstonia boonei on mitochondrial-mediated apoptosis. J Herbs Spices Med Plants 26(4):340–355
Olorunsogo OO, Bababunmi EA, Bassir O (1979) Uncoupling effect of Nphosphonomethylglycine on at liver mitochondria. Biochem Pharm 27:925–927. https://doi.org/10.1016/0006-2952(79)90377-0. PMID: 156546
Ozsoy N, Can A, Yanardag R, Akev N (2008) Antioxidant activity of Smilax excels L. leaf extracts. Food Chem 110:571–583
Prieto P, Pineda M, Aguilar M (1999) Spectrophotometric quantitation of antioxidant capacity through the formation of a phosphomolybdenum complex: specific application to the determination of vitamin E. Anal Biochem 269(2):337–341
Poe K (2017) Plant-based diets and phytonutrients: potential health benefits and disease prevention. Arch Med 9(6:7)
Ruberto G, Baratta M, Deans S, Dorman H (2000) Antioxidant and antimicrobial activity of Foeniculum vulgare and Crithmum maritimum essential oils. Planta Med 66:687–693. https://doi.org/10.1055/s-2000-9773
Shrivastava S, Jeengar MK, Reddy VS, Reddy GB, Naidu VG (2015) Anticancer effect of celastrol on human triple negative breast cancer: possible involvement of oxidative stress, mitochondrial dysfunction, apoptosis and PI3K/Akt pathways Exp. Mol Pathol 98(3):313–327
Singleton VL, Rossi JA Jr (1965) (1965) Colorimetry of total phenolics with phosphomolybdic-phosphotungstic acid reagents. American Journal of Enology and Vitculture 16(3):144–158
Sundaram R, Mitra SK (2007) Antioxidant activity of ethyl acetate soluble fraction of Acacia.arabica bark in rats. Indian J Pharmacol 39:33–38
Talib WH, Mahasneh AM (2010) Antiproliferative activity of plant extracts used against cancer in traditional medicine. Sci Pharm 78:133–146
van Loo G, Saelens X, van Gurp M et al (2002) The role of mitochondrial factors in apoptosis: a Russian roulette with more than one bullet. Cell Death Differ 9:1031–1042
Vitale I, Pietrocola F, Guilbaud E, Aaronson SA, Abrams JM, Adam D, Pasparakis M (2023) Apoptotic cell death in disease—current understanding of the NCCD 2023. Cell Death Differ 1–58
Yuan L, Cai Y, Zhang L, Liu S, Li P, Li X (2022) Promoting apoptosis, a promising way to treat breast cancer with natural products: a comprehensive review. Front Pharmacol 12:801662. https://doi.org/10.3389/fphar.2021.801662
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Funding
This study was not supported by any funding.
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
All procedures performed in studies involving animals were in accordance with the ethical standards of the institution or practice at which the studies were conducted.”
Informed consent
For this type of study informed consent is not required.
Consent for publication
For this type of study consent for publication is not required.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Nwaechefu, O., Adeoye, B., Lateef, I. et al. Cajanus cajan induces mitochondrial-mediated apoptosis via caspase activation and cytochrome c release. Comp Clin Pathol 33, 207–222 (2024). https://doi.org/10.1007/s00580-023-03541-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00580-023-03541-7