Abstract
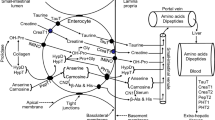
Zinc plays an important role in the biosynthesis and storage of insulin. Consequently, its deficiency may have a deleterious impact on the progression of diabetes and its associated consequences. Thus, this study was conducted to investigate the effect of hawthorn Crataegus azarolus on blood biochemical parameters, tissue zinc status, and oxidative stress biomarkers in streptozotocin-induced diabetic rats fed a zinc-insufficient diet. Thirty-two male albino Wistar rats were divided into four groups: two groups were fed a zinc-sufficient diet (one non-diabetic and the other diabetic), while the other two groups of diabetic rats were fed a zinc-insufficient diet. One group was not treated, and the other was treated with an extract of Crataegus azarolus (150 mg/kg body weight). Body weight and food intake were regularly recorded. After 4 weeks of dietary manipulation, the fasting animals were sacrificed. Zinc-deficient feed decreased body weight, insulin, and tissue zinc levels (in the femur, liver, kidney, and pancreas), as well as glutathione concentrations, lactic dehydrogenase, catalase, superoxide dismutase, glutathione peroxidase, and glutathione reductase activities. It was also noted that inadequate dietary zinc intake increased concentrations of glucose, cholesterol, triglycerides, urea, uric acid, creatinine, lipid peroxidation levels, and transaminase activities. However, oral administration of hawthorn extract was observed to improve all of the previously mentioned parameters, bringing them approximately back to their normal levels. The present study showed that Crataegus azarolus supplementation, presumably acting as an antioxidant, can be a natural source for reducing the development of diabetes caused by zinc deficiency.








Similar content being viewed by others
Data availability
All data underlying the reported findings are available in a publicly accessible repository. The data presented in this study are openly available in Research Square at https://doi.org/10.21203/rs.3.rs-2262461/v1.
References
Abdulmalek S, Eldala A, Awad D, Balbaa M (2021) Ameliorative effect of curcumin and zinc oxide nanoparticles on multiple mechanisms in obese rats with induced type 2 diabetes. Sci Rep 11:20677. https://doi.org/10.1038/s41598-021-00108-w
Abu-Gharbieh E, Shehab NG (2017) Therapeutic potentials of Crataegus azarolus var. eu-azarolus Maire leaves and its isolated compounds. BMC Complement Altern Med 17:218. https://doi.org/10.1186/s12906-017-1729-9
Aebi H (1984) Catalase in vitro. Meth Enzymol 105:121–126. https://doi.org/10.1016/S0076-6879(84)05016-3
Aierken A, Buchholz T, Chen C, Zhang X, Melzig MF (2017) Hypoglycemic effect of hawthorn in type II diabetes mellitus rat model. J Sci Food Agric 97(13):4557–4561. https://doi.org/10.1002/jsfa.8323
Alirezalu A, Salehi P, Ahmadi N, Sonboli A, Aceto S, Hatami Maleki H, Ayyari M (2018) Flavonoids profile and antioxidant activity in flowers and leaves of hawthorn species (Crataegus spp.) from different regions of Iran. Int J Food Prop 21:452–470. https://doi.org/10.1080/10942912.2018.1446146
Beloucif A, Kechrid Z, Bekada AM (2021) Effect of zinc deficiency on blood glucose, lipid profile, and antioxidant status in streptozotocin diabetic rats and the potential role of sesame oil. Biol Trace Elem Res. https://doi.org/10.1007/s12011-021-02934-5
Bonner-Weir S (1988) Morphological evidence for pancreatic polarity of beta-cell within islets of Langerhans. Diabetes 37:616–621. https://doi.org/10.2337/diab.37.5.616
Brand-Williams W, Cuvelier ME, Berset C (1995) Use of a free radical method to evaluate antioxidant activity. LWT Food Sci Technol 28:25–30. https://doi.org/10.1016/S0023-6438(95)80008-5
Buege JA, Aust SD (1978) Microsomal lipid peroxidation. In: Fleischer S, Packer L (eds) Methods in enzymology. Academic Press, pp 302–310
Chen Z, Hu H, Chen M, Luo X, Yao W, Liang Q, Yang F, Wang X (2020) Association of triglyceride to high-density lipoprotein cholesterol ratio and incidence of diabetes mellitus: a secondary retrospective analysis based on a Chinese cohort study. Lipids Health Dis 19:33. https://doi.org/10.1186/s12944-020-01213-x
Choi S, Liu X, Pan Z (2018) Zinc deficiency and cellular oxidative stress: prognostic implications in cardiovascular diseases. Acta Pharmacol Sin 39:1120–1132. https://doi.org/10.1038/aps.2018.25
Daniele C, Mazzanti G, Pittler MH, Ernst E (2006) Adverse-event profile of Crataegus spp.: a systematic review. Drug Saf 29(6):523–535. https://doi.org/10.2165/00002018-200629060-00005
Dar R, Shahnawaz M, Qazi P (2019) General overview of medicinal plants: a review. Phytomorphology 69:349–351. https://doi.org/10.31254/phyto.2017.6608
Derai EH, Kechrid Z (2014) Combined effect of vitamins C and E on zinc status, carbohydrate metabolism and antioxidant values in diabetic rats fed zinc-deficient diet. J Metab Nutr 7:55–65. https://doi.org/10.3233/MNM-140005
Derouiche S, Kechrid Z (2016) Zinc supplementation overcomes effects of copper on zinc status, carbohydrate metabolism and some enzyme activities in diabetic and nondiabetic rats. Can J Diabetes 40:342–347. https://doi.org/10.1016/j.jcjd.2016.02.005
El Hendy HA, Yousef MI, Abo El-Naga NI (2001) Effect of dietary zinc deficiency on hematological and biochemical parameters and concentrations of zinc, copper, and iron in growing rats. Toxicology 167:163–170. https://doi.org/10.1016/S0300-483X(01)00373-0
Evans WC, Evans D (2009) General methods associated with the phytochemical investigation of herbal products. In: Evans WC, Evans D (eds) Trease and Evans’ pharmacognosy, 16th ed. W.B. Saunders, pp 135–147
Flohé L, Günzler WA (1984) Assays of glutathione peroxidase. Methods Enzymol 105:114–121. https://doi.org/10.1016/S0076-6879(84)05015-1
Gholamhoseinian A, Shahouzehi B, Mohammadi G (2020) Trace elements content of some traditional plants used for the treatment of diabetes mellitus. Biointerface Res Appl Chem 10(6):6167–6173. https://doi.org/10.33263/BRIAC105.61676173
Goldberg D, Spooner R (1987) Methods of enzymatic analysis. Bergmayer, VCH, New York
Greeley S, Sandstead HH (1983) Oxidation of alanine and β-hydroxybutyrate in late gestation by zinc-restricted rats. J Nutr 113:1803–1810. https://doi.org/10.1093/jn/113.9.1803
Hamdiken M, Bouhalit S, Kechrid Z (2018) Effect of Ruta chalepensis on zinc, lipid profile and antioxidant levels in the blood and tissue of streptozotocin-induced diabetes in rats fed zinc-deficient diets. Can J Diabetes 42:356–364. https://doi.org/10.1016/j.jcjd.2017.08.239
Hao W, Zhang Z, Guo Y, Sun P, Lv X, Zuo Y (2011) Hawthorn fruit increases the antioxidant capacity and reduces lipid peroxidation in senescence-accelerated mice. Eur Food Res Technol 232(4):743–751. https://doi.org/10.1007/s00217-011-1435-7
He K, Song S, Zou Z, Feng M, Wang D, Wang Y, Li X, Ye X (2016) The hypoglycemic and synergistic effect of loganin, morroniside, and ursolic acid isolated from the fruits of Cornus officinalis. Phytother Res 30:283–291. https://doi.org/10.1002/ptr.5529
Jiménez PG, Martín-Carmona J, Hernández EL (2020) Diabetes mellitus. Medicine - Programa De Formación Médica Continuada Acreditado 13:883–890. https://doi.org/10.1016/j.med.2020.09.010
Jollow DJ, Mitchell JR, Zampaglione N, Gillette JR (1974) Bromobenzene-induced liver necrosis. Protective role of glutathione and evidence for 3,4-bromobenzene oxide as the hepatotoxic metabolite. Pharmacology 11:151–169. https://doi.org/10.1159/000136485
Julkunen-Tiitto R (1985) Phenolic constituents in the leaves of northern willows: methods for the analysis of certain phenolics. J Agric Food Chem 33:213–217. https://doi.org/10.1021/jf00062a013
Jurikova T, Sochor J, Rop O, Mlcek J, Balla S, Szekeres L, Adam V, Kizek R (2012) Polyphenolic profile and biological activity of Chinese hawthorn (Crataegus pinnatifida BUNGE) fruits. Molecules 17:14490–14509. https://doi.org/10.3390/molecules171214490
Kinlaw WB, Levine AS, Morley JE, Silvis SE, McClain CJ (1983) Abnormal zinc metabolism in type II diabetes mellitus. Am J Med 75:273–277. https://doi.org/10.1016/0002-9343(83)91205-6
Kwok CY, Li C, Cheng HL, Ng YF, Chan TY, Kwan YW, Leung GPH, Lee SMY, Mok DKW, Yu PHF, Chan SW (2013) Cholesterol lowering and vascular protective effects of ethanolic extract of dried fruit of Crataegus pinnatifida, hawthorn (Shan Zha), in diet-induced hypercholesterolaemic rat model. J Funct Foods 5:1326–1335. https://doi.org/10.1016/j.jff.2013.04.020
Li YV (2014) Zinc and insulin in pancreatic beta-cells. Endocrine 45:178–189. https://doi.org/10.1007/s12020-013-0032-x
Misra HP, Fridovich I (1977) Superoxide dismutase: “positive” spectrophotometric assays. Anal Biochem 79:553–560. https://doi.org/10.1016/0003-2697(77)90429-8
Neeta M, Mukta N, Bilwa K (2015) Comparative qualitative phytochemical analysis of Sesamum indicum L. Int J Curr Microbiol App Sci 4(3):172–181. https://doi.org/10.20546/ijcmas.2015.403.020
Ojuade FI, Olorundare OE, Akanbi OB, Afolabi SO, Njan AA (2021) Antidiabetic and antihyperlipidemic effects of aqueous extract of Parquetina nigrescens in streptozotocin–nicotinamide induced type 2 diabetic rats. Heliyon 7:e07363. https://doi.org/10.1016/j.heliyon.2021.e07363
Pal A, Khanum F, Bawa AS (2010) Nutritional, medicinal and industrial uses of sesame (Sesamum indicum L.) seeds - an overview. Agric Conspec Sci 75(4):159–168
Rahmati M, Keshvari M, Mirnasouri R, Chehelcheraghi F (2021) Exercise and Urtica dioica extract ameliorate hippocampal insulin signaling, oxidative stress, neuroinflammation, and cognitive function in STZ-induced diabetic rats. Biomed Pharmacother 139:111577. https://doi.org/10.1016/j.biopha.2021.111577
Shaheen AA, Abd El-Fattah AA (1995) Effect of dietary zinc on lipid peroxidation, glutathione, protein thiols levels and superoxide dismutase activity in rat tissues. Int J Biochem Cell Biol 27:89–95. https://doi.org/10.1016/1357-2725(94)00053-0
Shih CC, Lin CH, Lin YJ, Wu JB (2013) Validation of the antidiabetic and hypolipidemic effects of hawthorn by assessment of gluconeogenesis and lipogenesis related genes and AMP-activated protein kinase phosphorylation. Evid Based Complementary Altern Med 2013:597067. https://doi.org/10.1155/2013/597067
Southon S, Kechrid Z, Wright AJ, Fairweather-Tait SJ (1988) Effect of reduced dietary zinc intake on carbohydrate and Zn metabolism in the genetically diabetic mouse (C57BL/KsJ db+/db+). Br J Nutr 60:499–507. https://doi.org/10.1079/BJN19880122
Sun H, Saeedi P, Karuranga S, Pinkepank M, Ogurtsova K, Duncan BB, Stein C, Basit A, Chan JCN, Mbanya JC, Pavkov ME, Ramachandaran A, Wild SH, James S, Herman WH, Zhang P, Bommer C, Kuo S, Boyko EJ, Magliano DJ (2022) IDF diabetes atlas: global, regional and country-level diabetes prevalence estimates for 2021 and projections for 2045. Diabetes Res Clin Pract 183:109119. https://doi.org/10.1016/j.diabres.2021.109119
Suryawanshi NP, Bhutey AK, Nagdeote AN, Jadhav AA, Manoorkar GS (2006) Study of lipid peroxide and lipid profile in diabetes mellitus. Indian J Clin Biochem 21:126. https://doi.org/10.1007/BF02913080
Tebboub I, Kechrid Z (2021) Effect of curcuma on zinc, lipid profile and antioxidants levels in blood and tissue of streptozotocin-induced diabetic rats fed zinc deficiency diet. Arch Physiol Biochem 127:162–169. https://doi.org/10.1080/13813455.2019.1623820
Vélez-Alavez M, Labrada-Martagón V, Méndez-Rodriguez LC, Galván-Magaña F, Zenteno-Savín T (2013) Oxidative stress indicators and trace element concentrations in tissues of mako shark (Isurus oxyrinchus). Comp Biochem Physiol a: Mol Integr Physiol 165:508–514. https://doi.org/10.1016/j.cbpa.2013.03.006
Wang SZ, Wu M, Chen KJ, Liu Y, Sun J, Sun Z, Ma H, Liu LT (2019) Hawthorn extract alleviates atherosclerosis through regulating inflammation and apoptosis related factors: an experimental study. Chin J Integr Med 25(2):108–115. https://doi.org/10.1007/s11655-018-3020-4
Xin C, Zhao M, Wang J, Wang Z (2021) Hawthorn polyphenols, D-chiro-inositol, and epigallocatechin gallate exert a synergistic hypoglycemic effect. J Food Biochem 45:e13771. https://doi.org/10.1111/jfbc.13771
Yahyaoui A, Arfaoui MO, Rigane G, Hkir A, Amari K, Ben Salem R, Ammari Y (2019) Investigation on the chemical composition and antioxidant capacity of extracts from Crataegus azarolus L.: effect of growing location of an important Tunisian medicinal plant. Chem Afr 2(2):361–365. https://doi.org/10.1007/s42250-019-00054-1
Yang B, Liu P (2012) Composition and health effects of phenolic compounds in hawthorn (Crataegus spp.) of different origins. J Sci Food Agric 92(8):1578–1590. https://doi.org/10.1002/jsfa.5671
Yousef MI, El-Hendy HA, El-Demerdash FM, Elagamy EI (2002) Dietary zinc deficiency induced-changes in the activity of enzymes and the levels of free radicals, lipids and protein electrophoretic behavior in growing rats. Toxicology 175:223–234. https://doi.org/10.1016/s0300-483x(02)00049-5
Wong SP, Leong LP, Koh WH (2006) Antioxidant activities of aqueous extracts of selected plants. Food Chem 99(4):775–783. https://doi.org/10.1016/j.foodchem.2005.07.058
Wu M, Liu L, Xing Y, Yang S, Li H, Cao Y (2020) Roles and mechanisms of hawthorn and its extracts on atherosclerosis: a review. Front Pharmacol 11:118. https://doi.org/10.3389/fphar.2020.00118
Zhang B, Yue R, Chen Y, Yang M, Huang X, Shui J, Peng Y, Chin J (2019) Gut microbiota, a potential new target for Chinese herbal medicines in treating diabetes mellitus. Evid Based Complementary Altern Med 2019:2634898. https://doi.org/10.1155/2019/2634898
Zhang P, Li T, Wu X, Nice EC, Huang C, Zhang Y (2020) Oxidative stress and diabetes: antioxidative strategies. Front Med 14:583–600. https://doi.org/10.1007/s11684-019-0729-1
Zhang Y, Ti S, Liu S, Zhao J, Chen W, Wang H (2014) Effect of hawthorn on Drosophila melanogaster antioxidant-related gene expression. Trop J Pharm Res 13(3):353. https://doi.org/10.4314/tjpr.v13i3.6
Zhishen J, Mengcheng T, Jianming W (1999) The determination of flavonoid contents in mulberry and their scavenging effects on superoxide radicals. Food Chem 64:555–559. https://doi.org/10.1016/S0308-8146(98)00102-2
Acknowledgements
The authors express their gratitude to the Algiers Pasteur Institute for providing the rats used in this study and to ONAB EL-Harouch in Skikda, Algeria, for their assistance with diet component supplementation. Their contributions were essential to the successful completion of this research project.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Funding
This research was supported by a grant from the Ministry of Higher Education and Scientific Research, Algeria (Grant Number: D01N01UN230120190003).
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
All procedures were approved by the Ethical Committee of Annaba University, Algeria (PNR-ANDRS 8/u23/332). The Guide for the Care and Use of Laboratory Animals was followed.
Informed consent
All the authors read the manuscript and approved for submission.
Consent for publication
For this type of study, consent for publication is not required.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Triki, R., Boughediri, K., Chelaghmia, M.L. et al. The therapeutic potential of “Crataegus azarolus” on zinc, lipid profile, and antioxidant status in streptozotocin-induced diabetic rats with zinc-deficient diet. Comp Clin Pathol 33, 7–19 (2024). https://doi.org/10.1007/s00580-023-03517-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00580-023-03517-7