Abstract
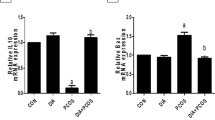
Polycystic ovary syndrome (PCOS) is a complex syndrome that causes infertility in women. Licorice is one of the medicinal plants with antioxidant properties used to treat diseases. This study investigated the anti-inflammatory effect of licorice extract on PCOS. After induction of PCOS by subcutaneous injection (SC) of dehydroepiandrosterone (DHEA) (60 mg/kg) in BALB/C mice for 21 days, the mice were treated with licorice extract (50 mg/kg) for 21 days. At the end of experimental procedures, ovarian morphology, blood antioxidant capacity, nitric oxide (NO), the abundance of Treg cells, and expression of matrix metalloproteinase-9 (MMP-9), transforming growth factor-ß (TGF-ß), cyclooxygenase-2 genes (COX-2), and tumor necrosis factor-alpha (TNF-α) were measured. The estrus cycle returned to normal after treatment with licorice extract. It was observed that the licorice number of ovarian cysts was significantly higher in the licorice group than in the PCOS group, and the corpus luteum was lower than in the control group. NO, FRAP, and Treg cell numbers did not significantly alter. The TGF- gene’s expression level significantly increased, but the expression level of TNF-α, COX-2, and MMP-9 did not change significantly. Immune responses play an important role in the development of inflammation. Licorice extract, with anti-inflammatory properties, may be an effective supplement in improving the histological and immunological changes of PCOS.






Similar content being viewed by others
Data availability
Data will be available on reasonable request from the corresponding author.
References
Ab Rahman MR, Abdul Razak F, Mohd Bakri M (2014) Evaluation of wound closure activity of nigella sativa, melastoma malabathricum, pluchea indica, and piper sarmentosum extracts on scratched monolayer of human gingival fibroblasts. Evidence-based Complement Altern Med 2014:. https://doi.org/10.1155/2014/190342
Ahmadi A, Mostafavi M, kalantari hesari A (2019) Histological studies of the effect of licorice root hydroalcoholic extract on the structure of polycystic ovary syndrome in mice following hyperandrogenism induced by letrozole. J Urmia Univ Med Sci 29:857–868
Al-Muala IK, Al-Jiboori BM (2010) Effect of licorice extract on ovulation induction in immature female mice. J Biotechnol Res Cent 4:118–129. https://doi.org/10.24126/jobrc.2010.4.1.106
Armanini D, Mattarello MJ, Fiore C et al (2004) Licorice reduces serum testosterone in healthy women. Steroids 69:763–766. https://doi.org/10.1016/j.steroids.2004.09.005
Bekyürek T, Lima N, Bayra G (2002) Diagnosis of sexual cycle by means of vaginal smear method in the chinchilla (Chinchilla lanigera). Lab Anim 36:51–60. https://doi.org/10.1258/0023677021911768
Brazdova A, Senechal H, Peltre G, Poncet P (2016) Immune aspects of female infertility. Int J Fertil Steril 10:1–10
Cao D, Jiang J, You L, et al (2016) The protective effects of 18β-glycyrrhetinic acid on helicobacter pylori -infected gastric mucosa in mongolian gerbils. Biomed Res Int 2016. https://doi.org/10.1155/2016/4943793
Cella M, Farina MG, Dominguez Rubio AP et al (2010) Dual effect of nitric oxide on uterine prostaglandin synthesis in a murine model of preterm labour. Br J Pharmacol 161:844–855. https://doi.org/10.1111/j.1476-5381.2010.00911.x
González-Reyes S, Santillán-Cigales JJ, Jiménez-Osorio AS et al (2016) Glycyrrhizin ameliorates oxidative stress and inflammation in hippocampus and olfactory bulb in lithium/pilocarpine-induced status epilepticus in rats. Epilepsy Res 126:126–133. https://doi.org/10.1016/j.eplepsyres.2016.07.007
Guerin LR, Prins JR, Robertson SA (2009) Regulatory T-cells and immune tolerance in pregnancy: a new target for infertility treatment? Hum Reprod Update 15:517–535
Hatziagelaki E, Pergialiotis V, Kannenberg JM et al (2020) Association between biomarkers of low-grade inflammation and sex hormones in women with polycystic ovary syndrome. Exp Clin Endocrinol Diabetes 128:723–730. https://doi.org/10.1055/a-0992-9114
Hernández-Jiménez JL, Barrera D, Espinoza-Simón E et al (2022) Polycystic ovarian syndrome: signs and feedback effects of hyperandrogenism and insulin resistance. Gynecol Endocrinol 38:2–9. https://doi.org/10.1080/09513590.2021.2003326
Hsieh MJ, Lin CW, Yang SF et al (2014) Glabridin inhibits migration and invasion by transcriptional inhibition of matrix metalloproteinase 9 through modulation of NF-κB and AP-1 activity in human liver cancer cells. Br J Pharmacol 171:3037–3050. https://doi.org/10.1111/bph.12626
Kauffman AS, Thackray VG, Ryan GE et al (2015) A novel letrozole model recapitulates both the reproductive and metabolic phenotypes of polycystic ovary syndrome in female mice. Biol Reprod 93. https://doi.org/10.1095/biolreprod.115.131631
Klein KO, Janfaza M, Wong JA, Chang RJ (2003) Estrogen bioactivity in Fo-Ti and other herbs used for their estrogen-like effects as determined by a recombinant cell bioassay. J Clin Endocrinol Metab 88:4077–4079. https://doi.org/10.1210/jc.2003-030349
Krishna MB, Joseph A, Subramaniam AG et al (2015) Reduced tregs in peripheral blood of PCOS patients - a consequence of aberrant Il2 signaling. J Clin Endocrinol Metab 100:282–292. https://doi.org/10.1210/jc.2014-2401
Lee HS, Kang P, Kim KY, Seol GH (2015) Foeniculum vulgare mill. Protects against lipopolysaccharide-induced acute lung injury in mice through ERK-dependent NF-kB activation. Korean J Physiol Pharmacol 19:183–189. https://doi.org/10.4196/kjpp.2015.19.2.183
Li C, Eom T, Jeong Y (2015) Glycyrrhiza glabra L. extract inhibits LPS-induced inflammation in RAW macrophages. J Nutr Sci Vitaminol (tokyo) 61:375–381. https://doi.org/10.3177/jnsv.61.375
Li M, Ruan X, Mueck AO (2022) Management strategy of infertility in polycystic ovary syndrome. Glob Heal J 6:70–74. https://doi.org/10.1016/j.glohj.2022.03.002
Luchetti CG, Mikó E, Szekeres-Bartho J et al (2008) Dehydroepiandrosterone and metformin modulate progesterone-induced blocking factor (PIBF), cyclooxygenase 2 (COX2) and cytokines in early pregnant mice. J Steroid Biochem Mol Biol 111:200–207. https://doi.org/10.1016/j.jsbmb.2008.06.007
Ma C, Ma Z, Liao XL et al (2013) Immunoregulatory effects of glycyrrhizic acid exerts anti-asthmatic effects via modulation of Th1/Th2 cytokines and enhancement of CD4+CD25 +Foxp3+ regulatory T cells in ovalbumin-sensitized mice. J Ethnopharmacol 148:755–762. https://doi.org/10.1016/j.jep.2013.04.021
Minnetti M, De Alcubierre D, Bonaventura I et al (2022) Effects of licorice on sex hormones and the reproductive system. Nutrition 103–104:111727. https://doi.org/10.1016/j.nut.2022.111727
Mokhtarian F, Shi Y, Shirazian D et al (1994) Defective production of anti-inflammatory cytokine, TGF-beta by T cell lines of patients with active multiple sclerosis. J Immunol 152:6003–6010. https://doi.org/10.4049/jimmunol.152.12.6003
Motta AB (2010) Dehydroepiandrosterone to induce murine models for the study of polycystic ovary syndrome. J Steroid Biochem Mol Biol 119:105–111
OECD (2001) OECD guidelines for the testing of chemicals, Section 4, Test No. 425: Acute Oral Toxicity - Up-and-Down Procedure. Guidel Test Chem 26
Padua MB, Tekin Ş, Spencer TE, Hansen PJ (2005) Actions of progesterone on uterine immunosuppression and endometrial gland development in the uterine gland knockout (UGKO) ewe. Mol Reprod Dev 71:347–357. https://doi.org/10.1002/mrd.20301
Salimi-Asl M, Mozdarani H, Kadivar M (2016) Up-regulation of miR-21 and 146a expression and increased DNA damage frequency in a mouse model of polycystic ovary syndrome (PCOS). BioImpacts 6:85–91. https://doi.org/10.15171/bi.2016.12
Sekhon LH, Gupta S, Kim Y, Agarwal A (2010) Female infertility and antioxidants. Curr Womens Health Rev 6:84–95. https://doi.org/10.2174/157340410791321381
Severin ME, Lee PW, Liu Y et al (2016) MicroRNAs targeting TGFb signalling underlie the regulatory T cell defect in multiple sclerosis. Brain 139:1747–1761. https://doi.org/10.1093/brain/aww084
Shalev E, Goldman S, Ben-Shlomo I (2001) The balance between MMP-9 and MMP-2 and their tissue inhibitor (TIMP)-1 in luteinized granulosa cells: comparison between women with PCOS and normal ovulatory women. Mol Hum Reprod 7:325–331. https://doi.org/10.1093/molehr/7.4.325
Shamsi M, Ghazavi A, Saeedifar AM et al (2022) The immune system’s role in PCOS. Mol Biol Rep 49:10689–10702. https://doi.org/10.1007/s11033-022-07695-5
Shamsi M, Nejati V, Najafi G (2016) Therapeutic effects of licorice extract on in vitro maturation and in vitro fertilization in mice model of polycystic ovary syndrome. J Maz Univ Med Sci 25:113–121
Shamsi M, Nejati V, Najafi G, Pour SK (2020) Protective effects of licorice extract on ovarian morphology, oocyte maturation, and embryo development in pcos-induced mice: an experimental study. Int J Reprod Biomed 18:865–876. https://doi.org/10.18502/ijrm.v13i10.7771
Sheikh-El-Arab Elsedeek M, Elmaghraby HAH (2011) Predictors and characteristics of letrozole induced ovulation in comparison with clomiphene induced ovulation in anovulatory PCOS women. Middle East Fertil Soc J 16:125–130. https://doi.org/10.1016/j.mefs.2010.11.004
Sugiyama H, Gyulai R, Toichi E et al (2005) Dysfunctional blood and target tissue CD4+CD25high regulatory T cells in psoriasis: mechanism underlying unrestrained pathogenic effector T cell proliferation. J Immunol 174:164–173. https://doi.org/10.4049/jimmunol.174.1.164
Tamir S, Eizenberg M, Somjen D et al (2001) Estrogen-like activity of glabrene and other constituents isolated from licorice root. J Steroid Biochem Mol Biol 78:291–298. https://doi.org/10.1016/S0960-0760(01)00093-0
Wahab S, Annadurai S, Abullais SS et al (2021) Glycyrrhiza glabra (Licorice): a comprehensive review on its phytochemistry, biological activities, clinical evidence and toxicology. Plants 10. https://doi.org/10.3390/plants10122751
Wang X, rong, Hao H guang, Chu L, (2017) Glycyrrhizin inhibits LPS-induced inflammatory mediator production in endometrial epithelial cells. Microb Pathog 109:110–113. https://doi.org/10.1016/j.micpath.2017.05.032
Yazdi A, Sardari S, Sayyah M, Ezzati MH (2011) Evaluation of the anticonvulsant activity of the leaves of glycyrrhiza glabra var. glandulifera grown in Iran, as a possible renewable source for anticonvulsant compounds. Iran J Pharm Res 10:75–82
Yilmaz N, Inal HA, Gorkem U et al (2013) Follicular fluid total antioxidant capacity in patient with PCOS. Fertil Steril 100:S356. https://doi.org/10.1016/j.fertnstert.2013.07.838
Zhou R, Xu L, Ye M et al (2014) Formononetin inhibits migration and invasion of MDA-MB-231 and 4T1 breast cancer cells by suppressing MMP-2 and MMP-9 through PI3K/AKT signaling pathways. Horm Metab Res 46:753–760. https://doi.org/10.1055/s-0034-1376977
Author information
Authors and Affiliations
Contributions
Ensieh Seif Amirhoseiny: designed the experiments, performed most of the experiments, wrote the manuscript and Editing, and performed the formal analysis of results. Ali Ganji: performed the data analyses, conceptualization, and review. Ghasem Mosayebi: conceptualization, review. Maryam Shamsi: performed most of the experiment, wrote the manuscript, and editing, review. Ali Ghazavi: designed the methodology, analysis, interpretation of data, critical revising manuscript, and funding acquisition. All authors have read and approved the manuscript.
Corresponding author
Ethics declarations
Funding
This work was supported by the Research Council of Arak University of Medical Sciences (grant number: 243).
Conflict of interest
The authors declare no competing interests.
Ethics approval
All procedures performed in studies involving animals were in accordance with the ethical standards of the institutional Ethics Committee of the Arak University of Medical Sciences (IR.ARAKMU.REC.1398.243). All experiments in this study were based on the guidelines of the World Health Organization for the care and use of laboratory animals.
Informed consent
For this type of study informed consent is not required.
Consent for publication
For this type of study consent for publication is not required.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Amirhoseiny, E.S., Ganji, A., Mosayebi, G. et al. Improves immunological and histological alterations associated with Polycystic ovarian syndrome in DHEA -induced mice by Licorice extract. Comp Clin Pathol 32, 827–835 (2023). https://doi.org/10.1007/s00580-023-03492-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00580-023-03492-z