Abstract
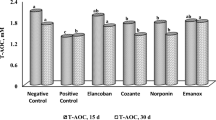
In the interests of food safety and public health, plants and their compounds are now reemerging as an alternate approach to treat gastrointestinal diseases in chickens. In this study, we evaluated the impact of two edible multi-plant extract compounds on growth performance, gut bacteria, and hematological parameters, anticoccidial index (ACI), and optimum anticoccidial activity (OAA) in 17-day-old domestic chickens experimentally infected with protozoan Eimeria. The highest weight gain was recorded in the uninfected unmedicated group followed by a mixture of medicinal plants 0.01 (T3) group. The basophil and total red blood cells were significantly higher in control groups in comparison with those in all other groups, and the total white blood cells were significantly lower in control groups compared with those in all other groups. Higher oocyst per gram (OPG) was recorded in the infected unmedicated group (T2), followed by Karela 0.01 (T5), and lower OPG in the mixture of medicinal plants 0.01 (T3), T3, and Karela 0.001 (T6), followed by sulfaclozine treatment (T7). T3 had ACI values of 129.79. According to the OAA index, the T3, T4, and T5 groups were partially resistant and sensitive to T6. T7 groups were sensitive to sulfaclozine. In this study, we found that the levels of Lactobacillus, which are positively associated with gut health, increased, whereas Clostridium, Salmonella, and total declined over time in response to the mixture of medicinal plants and Karela. These results indicated that the mixture of medicinal plants and Karela can be effectively used for the treatment of coccidiosis in domestic chickens.


Similar content being viewed by others
References
Abera S, Ibrahim N, Kabeta T (2016) Prevalence of poultry coccidiosis in addis ababa poultry farms. Br J Poult Sci 5:21–25. https://doi.org/10.5897/JVMAH2016.0505
Abou-Elkhair R, Gaafar KM, Elbahy NM, Helal MA, Mahboub HDH, Sameh G (2014) Bioactive effect of dietary supplementation with essential oils blend of oregano, thyme and garlic oils on performance of broilers infected with Eimeria species. Glob Vet 13:977–985. https://doi.org/10.5829/idosi.gv.2014.13.06.9137
Ahad S, Tanveer S, Malik TA, Nawchoo IA (2018) Anticoccidial activity of fruit peel of Punica granatum L. Microb Pathog 116:78–83. https://doi.org/10.1016/j.micpath.2018.01.015
Al-Kassie GAM (2009) Influence of two plant extracts derived from thyme and cinnamon on broiler performance. Pakistan Vet J 29: 169–173. http://pvj.com.pk/pdf-files/29_4/169-173.pdf
Al-Shaibani IRM, Al-Khadher AMA, Al-Hibah AZH (2020) Anticoccidial activity of Allium sativum and Punica granatum against experimentally induced Eimeria tenella infection in broiler chickens. Asian J Res Anim Vet Sci 5: 20–29. https://doi.org/10.13140/RG.2.2.17167.20642
Arabkhazaeli F, Modrisanei M, Nabian S, Mansoori B, Madani A (2013) Evaluating the resistance of Eimeria spp. field isolates to anticoccidial drugs using three different indices. Iran J Parasitol 8(2): 231–241 (PMCID: PMC3724148)
Bjerrum L, Engberg RM, Leser TD, Jensen BB, Finster K, Pedersen K (2006) Microbial community composition of the ileum and cecum of broiler chickens as revealed by molecular and culture-based techniques. Poult Sci 85:1151–1164. https://doi.org/10.1093/ps/85.7.1151
Chandrakesan P, Muralidharan K, Kumar VD, Ponnudurai G, Harikrishnan TJ, Rani KSVN (2009) Efficacy of a herbal complex against caecal coccidiosis in broiler chickens. Veterinarski Arhiv 79:199–203
Chang CLT, Chung CY, Kuo CH, Kuo TF, Yang CW, Yang WC (2016a) Beneficial effect of Bidens pilosa on body weight gain, food conversion ratio, gut bacteria and coccidiosis in chickens. PLoS ONE 11:1–15. https://doi.org/10.1371/journal.pone.0146141
Chang CLT, Yang CY, Muthamilselvan T, Yang WC (2016b) Field trial of medicinal plant, Bidens pilosa, against eimeriosis in broilers. Sci Rep 6:1–7. https://doi.org/10.1038/srep24692
Collier CT, Hofacre CL, Payne AM, Anderson DB, Kaiser P, Mackie RI, Gaskins HR (2008) Coccidia-induced mucogenesis promotes the onset of necrotic enteritis by supporting Clostridium perfringens growth. Vet Immunol Immunopathol 122:104–115. https://doi.org/10.1016/j.vetimm.2007.10.014
Cui N, Wang X, Wang Q, Li H, Wang F, Zhao X (2017) Effect of dual infection with Eimeria tenella and subgroup J Avian leukosis virus on the cecal microbiome in specific-pathogen-free chicks. Front Vet Sci 4:1–10. https://doi.org/10.3389/fvets.2017.00177
De Pablos LM, Dos Santos MFB, Montero E, Garcia-Granados A, Parra A, Osuna A (2010) Anticoccidial activity of maslinic acid against infection with Eimeria tenella in chickens. Parasitol Res 107:601–604. https://doi.org/10.1007/s00436-010-1901-3
del Cacho E, Gallego M, Lee SH, Lillehoj HS, Quilez J, Lillehoj EP, Sanchez-Acedo C (2012) Induction of protective immunity against Eimeria tenella, Eimeria maxima, and Eimeria acervulina infections using dendritic cell-derived exosomes. Infect Immun 80:1909–1916. https://doi.org/10.1128/IAI.06413-11
Elkatcha M, Soltan M, ElShall N, El-Desoky A (2018) Effect of high dietary level of some amino acids and coccidial infection on growth performance and health status of broiler chicken. Alexandria J Vet Sci 59:147–165. https://doi.org/10.5455/ajvs.281585
Fatemi A, Asasi K, Razavi SM (2017) Anticoccidial effects of Artemisia annua ethanolic extract: prevention, simultaneous challenge-medication, and treatment. Parasitol Res 116:2581–2589. https://doi.org/10.1007/s00436-017-5567-y
Habibi H, Firouzi S, Nili H, Razavi M, Asadi SL, Daneshi S (2016) Anticoccidial effects of herbal extracts on Eimeria tenella infection in broiler chickens: in vitro and in vivo study. J Parasit Dis 40:401–407. https://doi.org/10.1007/s12639-014-0517-4
Huang G, Tang X, Bi F, Hao Z, Han Z, Suo J, Zhang S, Wang S, Duan C, Yu Z, Yu F, Yu Y, Lv Y, Suo X, Liu X (2018) Eimeria tenella infection perturbs the chicken gut microbiota from the onset of oocyst shedding. Vet Parasitol 258:30–37. https://doi.org/10.1016/j.vetpar.2018.06.005
Kaa S (2019) Medicinal plants and its effect on some physiological traits in birds: a review. Res J Pharm Biol Chem Sci 10:1194–1198
Kogut MH (2017) Issues and consequences of using nutrition to modulate the avian immune response. J Appl Poult Res 26:1–8. https://doi.org/10.3382/japr/pfx028
Long PL, Millard BJ, Joyner LP, Norton CC (1976) A guide to laboratory techniques used in the study and diagnosis of avian coccidiosis. Folia Vet Lat 6:201–217
Macdonald SE, Nolan MJ, Harman K, Boulton K, Hume DA, Tomley FM, Stabler RA, Blake DP (2017) Effects of Eimeria tenella infection on chicken caecal microbiome diversity, exploring variation associated with severity of pathology. PLoS ONE 12:1–17. https://doi.org/10.1371/journal.pone.0184890
Mushtaq M, Durrani FR, Imtiaz N, Sadique U, Hafeez A, Akhtar S, Ahmad S (2012) Effect of administration of Withania somnifera on some hematological and immunological profile of broiler chicks. Pak Vet J 32:70–72. http://www.pvj.com.pk/pdf-files/32_1/70-72.pdf
Muthamilselvan T, Kuo TF, Wu YC, Yang WC (2016) Herbal remedies for coccidiosis control: a review of plants, compounds, and anticoccidial actions. Evidence-based Complement. Evid Based Complemen Alternat Med 2657981:1–19. https://doi.org/10.1155/2016/2657981
Mwale M, Masika PJ (2009) Ethno-veterinary control of parasites, management and role of village chickens in rural households of Centane district in the Eastern Cape, South Africa. Trop Anim Health Prod 41:1685–1693. https://doi.org/10.1007/s11250-009-9366-z
National Research Council (1994) Nutrient requirements of poultry, 9th rev. National Academy Press. Washington, DC, p 1994
Oloruntola OD, Ayodele SO, Agbede JO, Oloruntola DA (2016) Effect of feeding broiler chickens with diets containing Alchornea cordifolia leaf meal and enzyme supplementation. Archivos de Zootecnia 65: 489–498. https://www.redalyc.org/pdf/495/49549091003.pdf
Peek HW, Landman WJM (2011) Coccidiosis in poultry: anticoccidial products, vaccines and other prevention strategies. Vet Quarter 31:143–161. https://doi.org/10.1080/01652176.2011.605247
Pereira JV, Bergamo DCB, Pereira JO, Franca SDC, Pietro RCLR, Silva-Sousa YTC (2005) Antimicrobial activity of Arctium lappa constituents against microorganisms commonly found in endodontic infections. Braz Dent J 16:192–196. https://doi.org/10.1590/s0103-64402005000300004
Pop LM, Varga E, Coroian M, Nedisan ME, Mircean V, Dumitrache MO, Farczadi L, Fulop I, Croitoru MD, Fazakas M, Gyorke A (2019) Efficacy of a commercial herbal formula in chicken experimental coccidiosis. Paras Vectors 12:1–9. https://doi.org/10.1186/s13071-019-3595-4
Rathinam T, Chapman HD (2009) Sensitivity of isolates of Eimeria from Turkey flocks to the anticoccidial drugs amprolium, clopidol, diclazuril, and monensin. Avian Dis 53:405–408. https://doi.org/10.1637/8679-030509-Reg.1
Shargh MS, Dastar B, Zerehdaran S, Khomeiri M, Moradi A (2012) Effects of using plant extracts and a probiotic on performance, intestinal morphology, and microflora population in broilers. J Appl Poult Res 21:201–208. https://doi.org/10.3382/japr.2010-00145
Simaraks S, Chinrasri O, Aengwanich W (2004) Hematological, electrolyte and serum biochemical values of the Thai indigenous chickens (Gallus domesticus) in northeastern, Thailand. Songklanakarin J Sci Technol 26: 425–430. https://rdo.psu.ac.th/sjstweb/journal/26-3/14Thai-chicken.pdf
Singh VS, Palod J, Shukla SK, Shukla PK (2008) Influence of an herbal anticoccidials on growth, parasitological, hematological parameters and carcass traits of broilers experimentally infected with mixed field isolates of Eimeria species. Indian j Anim Sci 78:1057–1062
Sugiharto S (2016) Role of nutraceuticals in gut health and growth performance of poultry. J Saudi Soc Agric Sci 15:99–111. https://doi.org/10.1016/j.jssas.2014.06.001
Sundar SB, Harikrishnan TJ, Latha BR, Chandra GS, Kumar TM (2017) Anticoccidial drug resistance in chicken coccidiosis and promising solutions : a review. J Entomol Zool Stud 5:1526–1529. https://www.entomoljournal.com/archives/2017/vol5issue4/PartT/5-4-96-998.pdf
Wunderlich F, Al-Quraishy S, Steinbrenner H, Sies H, Dkhil MA (2014) Towards identifying novel anti-Eimeria agents: trace elements, vitamins, and plant-based natural products. Parasitol Res 113:3547–3556. https://doi.org/10.1007/s00436-014-4101-8
Yang WC, Tien YJ, Chung CY, Chen YC, Chiou WH, Hsu SY, Liu HY, Liang CL, Chang CLT (2015) Effect of Bidens pilosa on infection and drug resistance of Eimeria in chickens. Res Vet Sci 98:74–81. https://doi.org/10.1016/j.rvsc.2014.11.002
Zhou Z, Nie K, Huang Q, Li K, Sun Y, Zhou R, Wang Z, Hu S (2017) Changes of cecal microflora in chickens following Eimeria tenella challenge and regulating effect of coated sodium butyrate. Exp Parasitol 177:73–81. https://doi.org/10.1016/j.exppara.2017.04.007
Author information
Authors and Affiliations
Contributions
HH and NGh were involved in the collection of data, statistical analysis, and drafting of the manuscript. All the authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Consent for publication
All the authors gave their informed consent prior to their inclusion in the study.
Competing interests
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Habibi, H., Ghahtan, N., Tohidi, S. et al. Effect of composition of medicinal plants on growth performance, gut bacteria, hematological parameters, anticoccidial index, and optimum anticoccidial activity in domestic chicken. Comp Clin Pathol 31, 737–745 (2022). https://doi.org/10.1007/s00580-022-03352-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00580-022-03352-2