Abstract
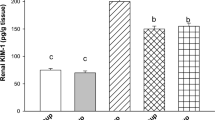
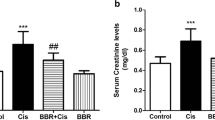
All components of Ziziphus jujube (ZJ) were used in medicine and food in ancient Iran and China. Cisplatin is one of the most important drugs in the treatment of solid tumors. Taking this drug can result in nephrotoxicity through several mechanisms. The purpose of this study was to evaluate the effects of ZJ on the nephrotoxicity induced by cisplatin in rats. Thirty-five experimental rats were randomly divided into the following five groups (n = 7 per group) for an 8-day study: 1- Group C1 was the control group and received distilled water (1 ml/day); 2- Group C2 received a single dose of intraperitoneal cisplatin (5 mg/kg); 3- Group ZJ received 1500 mg/kg/day Ziziphus Jujube extract orally; 4- Group ZJ1 was given 1500 mg/kg/day of Ziziphus Jujube extract orally with taking a single dose of cisplatin (5 mg/kg) on the first day only; 5- Group ZJ2 received 3000 mg/kg/day of Ziziphus jujube extract orally with taking a single dose of cisplatin (5 mg/kg) on the first day only. Eventually, histopathological parameters, blood urea nitrogen (BUN), malondialdehyde (MDA), alanine transaminase (ALT), and aspartate transaminase (AST) were assessed. The findings showed that cisplatin administration resulted in severe degeneration in all parts of the nephron tubules. Also, the present study showed that MDA levels were significantly lower in both ZJ1 and ZJ2 groups compared with those in group C2 (p < 0.01). Moreover, the cisplatin-induced elevation of serum BUN levels significantly decreased in both ZJ1 and ZJ2 groups in comparison with that in group C2 (p < 0.01). In addition, serum levels of both ALT and AST were significantly higher in group C2 in comparison with those in group C1 (p < 0.05). Extensive tubular necrosis was seen in group C2. In brief, results of this research indicated that ZJ could prevent cisplatin-induced kidney injury in rats.




Similar content being viewed by others
References
Awad DS, Ali RM, Mhaidat NM, Shotar AM (2014) Zizyphus jujuba protects against ibuprofen-induced nephrotoxicity in rats. Pharm Biol 52(2):182–186
Chirino YI, Pedraza-Chaverri J (2009) Role of oxidative and nitrosative stress in cisplatin-induced nephrotoxicity. Exp Toxicol Pathol 61(3):223–242
Ghaly IS, Said A, Abdel-Wahhab MA (2008) Zizyphus jujuba and Origanum majorana extracts protect against hydroquinone-induced clastogenicity. Environ Toxicol Pharmacol 25(1):10–19
Guo S, Duan J-a, Tang Y, Su S, Shang E, Ni S et al (2009) High-performance liquid chromatography—two wavelength detection of triterpenoid acids from the fruits of Ziziphus jujuba containing various cultivars in different regions and classification using chemometric analysis. J Pharm Biomed Anal 49(5):1296–1302
Ingale KG, Thakurdesai PA, Vyawahare NS (2013) Protective effect of Hygrophila spinosa against cisplatin induced nephrotoxicity in rats. Indian J Pharm 45(3):232
İşeri S, Ercan F, Gedik N, Yüksel M, Alican I (2007) Simvastatin attenuates cisplatin-induced kidney and liver damage in rats. Toxicology 230(2-3):256–264
Jiang JG, Huang XJ, Chen J (2007) Separation and purification of saponins from Semen Ziziphus jujuba and their sedative and hypnotic effects. J Pharm Pharmacol 59(8):1175–1180
Jung K, An JM, Eom D-W, Kang KS, Kim S-N (2017) Preventive effect of fermented black ginseng against cisplatin-induced nephrotoxicity in rats. J Ginseng Res 41(2):188–194
Kaeidi A, Taati M, Hajializadeh Z, Jahandari F, Rashidipour M (2015) Aqueous extract of Zizyphus jujuba fruit attenuates glucose induced neurotoxicity in an in vitro model of diabetic neuropathy. Iran J Basic Med Sci 18(3):301
Karakoc H, Altintas R, Parlakpinar H, Polat A, Samdanci E, Sagir M et al (2014) Protective effects of molsidomine against cisplatin-induced nephrotoxicity. Adv Clin Exp Med 24(4):585–593
Kuhad A, Tirkey N, Pilkhwal S, Chopra K (2006) Renoprotective effect of Spirulina fusiformis on cisplatin-induced oxidative stress and renal dysfunction in rats. Ren Fail 28(3):247–254
Kumar SP, Asdaq SM, Kumar NP, Asad M, Khajuria DK (2009) Protective effect of Zizyphus jujuba fruit extract against paracetamol and thioacetamide induced hepatic damage in rats. The Internet Journal of Pharmacology. 7(1):13667
Mahajan RT, Chopda M (2009) Phyto-pharmacology of Ziziphus jujuba Mill-a plant review. Pharmacogn Rev 3(6):320
Miller R, Tadagavadi R, Ramesh G, Reeves W (2010) Mechanisms of cisplatin nephrotoxicity. Toxins (Basel) 2(11):2490–2518. https://doi.org/10.3390/toxins2112490
Movassaghi S, Oudarji AY, Sharifi ZN (2016) Effect of pentoxifylline on apoptosis of kidney’s cells following acute methamphetamine administration in male Wistar rats. Galen Med J 5(3):131–138
Pabla N, Dong Z (2008) Cisplatin nephrotoxicity: mechanisms and renoprotective strategies. Kidney Int 73(9):994–1007
Taati M, Alirezaei M, Meshkatalsadat M, Rasoulian B, Kheradmand A, Neamati S (2011) Antioxidant effects of aqueous fruit extract of Ziziphus jujuba on ethanol-induced oxidative stress in the rat testes. Iran J Veterinary Res 12(1):39–45
Wafaa M, Abozied I, Helmy NA (2011) Production and development of new products from local and Chinese Ber fruits. Aust J Basic Appl Sci 5:652–659
Acknowledgments
The authors wish to acknowledge the Vice Chancellery of Research and Technology in Shiraz University of Medical Sciences, Shiraz, I.R. Iran for provision of animal facilities, biochemistry, and histopathology analyses services.
Funding
The authors received provision of enough funds (Grant no 92-01-21-6906) for our project from the Vice Chancellery of Research and Technology in Shiraz University of Medical Sciences, Shiraz, I.R. Iran.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Tanideh, N., Zarifi, F., Rafiee, S. et al. Ziziphus jujube protects against cisplatin-induced nephrotoxicity in rats. Comp Clin Pathol 29, 321–326 (2020). https://doi.org/10.1007/s00580-019-03055-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00580-019-03055-1