Abstract
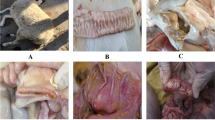
Paratuberculosis (Johne’s disease) is the chronic infectious granulomatous enteritis of ruminants caused by Mycobacterium avium subspecies paratuberculosis. Due to the lack of definite diagnostic tests, the early detection of the disease is difficult. The present study was undertaken to assess comparative efficacy of diagnostic methods routinely used in the identification and confirmation of Mycobacterium avium subspecies paratuberculosis (MAP) in the tissue samples of naturally infected clinical and subclinical sheep. The ileum and mesenteric lymph node (MLN) tissues were collected from 20 paratuberculous sheep from organized farm of Rajasthan. These animals were further classified as paucibacillary (PB) (n = 8) or multibacillary (MB) (n = 12) on the basis of histopathological findings and mycobacterial loads. The ileum and MLN sections from PB and MB and uninfected control groups were tested by indirect immunoperoxidase technique (IPT), Ziehl Neelsen’s (ZN) method, bacterial culture, IS900 PCR and 251 gene PCR. In PB sheep, IPT and ZN were positive in 87.5 and 50% of intestinal sections and 75 and 37.5% of MLN sections, respectively. In MB sheep, IPT and ZN had equal sensitivity. The overall sensitivity of IPT was found to be superior (95%) than ZN method (80%) in demonstration of acid-fast bacteria in intestinal tissues. On the bacterial cultural examination of the intestinal and MLN tissues using Herrold’s egg yolk medium, MAP was isolated in eight (66.6%) MB and in four (50%) PB sheep. Among 20 sheep, IS900 PCR detected 13 (65.0%) and 251 gene PCR detected 16 (80%) sheep as positive for MAP. In the PB sheep, the culture, ZN test and PCR sensitivity was found low in comparison to MB sheep. The sensitivity of IPT in PB sheep was better than other tests in detecting the MAP infection, which underlines its utility in confirmation of subclinical and clinical cases of paratuberculosis in sheep. The sensitivity of 251 gene PCR assay was found better than IS900 gene PCR and has potential to be used as screening test for clinical and subclinical paratuberculosis in sheep flocks.



Similar content being viewed by others
References
Bauerfeind R, Benazzi S, Weiss R, Schliesser T, Willems H, Balger G (1996) Molecular characterization of Mycobacterium paratuberculosis isolates from sheep, goats, and cattle by hybridization with a DNA probe to insertion element IS900. J Clin Microbiol 34:1617–1621
Behr MA, Kapur V (2008) The evidence for Mycobacterium paratuberculosis in Crohn’s disease. Curr Opin Gastroenterol 24:17–21
Clarke CJ, Little D (1996) The pathology of ovine paratuberclosis : gross and histopathological changes in the intestine and other tissues. J Comp Pathol 114:410–437
Delgado F, Etchechoury D, Gioffre A et al (2009) Comparison between two in situ methods for Mycobacterium avium subsp. paratuberculosis detection in tissue samples from infected cattle. Vet Microbiol 134:383–387
Delgado L, Garcia Marin JF, Munoz M, Benavides J, Juste RA, Garcia-Pariente C, Fuertes M, Gonzalez J, Ferreras MC, Perez V (2013) Pathological findings in young and adult sheep following experimental infection with two different doses of Mycobacterium avium subspecies paratuberculosis. Vet Pathol 50:857–866. https://doi.org/10.1177/0300985813476066
Eisenberg SW, Rutten VP, Koets AP (2015) Dam Mycobacterium avium subspecies paratuberculosis (MAP) infection status does not predetermine calves for future shedding when raised in a contaminated environment: a cohort study. Vet Res 46(70). https://doi.org/10.1186/s13567-015-0191-2.
Englund S, Bolske G, Johansson KE (2002) An IS900-like sequence found in a Mycobacterium sp. other than Mycobacterium avium subsp. paratuberculosis. FEMS Microbiol Lett 209:167–171
Gwozdz JM, Thompson KG, Murray A, Reichel MP, Manktelow BW, West DM (2000) Comparison of three serological tests and an interferon-γ assay for the diagnosis of paratuberculosis in experimentally infected sheep. Aust Vet J 78:779–783
Hemida H, Kihal M (2015) Detection of paratuberculosis using histopathology, immunohistochemistry, and ELISA in West Algeria. Comp Clin Pathol 24:1621–1629. https://doi.org/10.1007/s00580-015-2126-8
Imirzalioglu C, Dahemen H, Hain T, Billion A, Kuenne C, Chakraborty T, Domann E (2011) Highly specific and quick detection of Mycobacterium avium subsp. Paratuberculosis in feces and gut tissues of cattle and humans by multiple real-time PCR assays. J Clin Microbiol 49:1843–1852
Kumar AA (2004) Moleular pathology and diagnosis of paratuberculosis in sheep. Ph.D. thesis, IVRI, Deemed University, Izatnagar, UP, India.
Kumar AA, Tripathi BN, Jolhe DK (2006) Immunohistochemical demonstraton of mycobacterial antigents in sheep experimentally infected with Mycobacterium avium subsp. paratuberculosis. Indian J Vet Pathol 30:1–4
Kurade NP, Tripathi BN, Rajukumar K, Parihar NS (2004) Sequential development of histologic lesions and their relationship with bacterial isolation, fecal shedding, and immune responses during progressive stage of experimental infection of lambs with Mycobacterium avium subsp. paratuberculosis. Vet Pathol 41:378–387
Lombard JE (2011) Epidemiology and economics of paratuberculosis. Vet Clin North Am Food Anim Pract 27:525–535
Martinson SA, Hanna PE, Ikede BO, Lewis JP, Miller LM, Keefe GP, McKenna SLB (2008) Comparison of bacterial culture, histopathology, and immunohistochemistry for the diagnosis of Johne’s disease in culled dairy cows. J Vet Diagn Investig 20:51–57
Olsen I, Boysen P, Kulberg S, Hope JC, Jungersen G, Storset AK (2005) IFN-γ production from NK cells: implication for diagnostic testing. Proc 8th ICP: 118–121
Rajeev S, Zhang Y, Sreevatsan S, Alifiya S, Motiwala A, Byrum B (2005) Evaluation of multiple genomic targets for identification and confirmation of Mycobacterium avium subsp. paratuberculosis isolates using real time PCR. Vet Microbiol 105:215–221
Rajendra Kumar T (2008) Immunopathoogical studies on paratuberculosis (Johne’s disease) in goats. M.V.Sc. thesis, IVRI, Deemed University, Izatnagar, UP, India
Sibley JA, Woodbury MR, Appleyard GD, Elklin B (2007) Mycobacterium avium subsp paratuberculosis in Bison (Bison bison) from northern Canada. J Wildl Dis 43:775–779
Sirak A (2010) Isolation of Mycobacterium paratuberculosis from apparently healthy sheep and goats. Ethiop Vet J 14:113–120
Sivakumar P, Tripathi BN, Singh N, Sharma AK (2006) Pathology of naturally occurring paratuberculosis in water buffaloes (bubalus bubalis). Vet Pathol 43:455–462
Stabel JR (1997) An improved method for cultivation of Mycobacterium avium subsp. paratuberculosis from bovine fecal samples and comparison to three other methods. J Vet Diagn Investig 9:375–380
Stabel JR, Bannantine JP (2005) Development of a nested PCR method targeting a unique multicopy element, ISMap02, for detection of Mycobacterium avium subsp. paratuberculosis in faecal samples. J Clin Microbiol 43:4744–4750
Tripathi BN, Sivakumar P, Paliwal OP, Singh N (2006) Comparison of IS900 tissue PCR, bacterial culture, johnin and serological tests for diagnosis of naturally occurring paratuberculosis in goats. Vet Microbiol 166:4244–4250
Verin R, Perroni M, Rossi G, De Grossi L, Botta R, De Sanctis B, Rocca S, Cubeddu T, Crosby-Durrani H, Taccini E (2016) Paratuberculosis in sheep: histochemical, immunohistochemical and in situ hybridization evidence of in utero and milk transmission. Res Vet Sci 106:173–179
Verna AE, Garcia-Pariente C, Munoz M, Moreno O, Garcia Martin JF et al (2007) Variations in the immuno-pathological responses of lambs after experimental infection with different strains of Mycobacterium avium subsp. paratuberculosis. Zoonoses Public Health 54:243–252
Whitlock RH, Wells SJ, Sweeney RW, Van TJ (2000) ELISA and fecal culture for paratuberculosis (Johne’s disease): sensitivity and specificity of each method. Vet Microbiol 77:387–398
Whittington RJ, Sergeant ESG (2001) Progress towards understanding the spread, detection and control of Mycobacterium avium subsp. paratuberculosis in animal subpopulations. Aust Vet J 79:267–278
Acknowledgements
We are thankful to the Director IVRI, Izatnagar, Bareilly (UP) and the Director, CSWRI, Avikanagar, Rajasthan for providing necessary help and facility to carry out this study.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical statement
All animal experimental procedures were performed with approval of Institute Animal Ethics Committee (IAEC), ICAR-CSWRI, Avikanagar constituted by Committee for the Purpose of Control and Supervision of Experiments on Animals (CPCSEA) India. All applicable institutional guidelines for the care and use of animals were followed.
Informed consent
Informed consent was obtained from all individual participants included in the study.
Statement on human and animal rights
All institutional and national guidelines for the care and use of laboratory animals were followed.
Rights and permissions
About this article
Cite this article
Sonawane, G.G., Tripathi, B.N. Comparative diagnosis of Mycobacterium avium subspecies paratuberculosis in the tissues of clinical and subclinical sheep of paratuberculosis endemic farm. Comp Clin Pathol 28, 549–555 (2019). https://doi.org/10.1007/s00580-018-2837-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00580-018-2837-8