Abstract
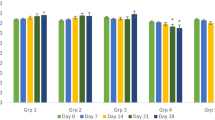
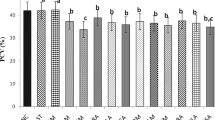
Caloncoba echinata is enlisted among the medicinal plants of African origin. However, little is known about its safety and general effects in the body. This study investigated the acute toxicity property, phytochemical profile, and haematological and histomorphometric changes of vital organs associated with its administration. Thirty-two albino mice assigned into eight groups of four mice per group were used for acute toxicity study. Groups 1–7 rats received 100, 500, 1000, 2000, 3000, 4000 and 5000 mg/kg of the extract orally, respectively, while distilled water (DW) was administered to the rats in group 8. The mice were observed for 48 h for signs of toxicity. Standard method was used to evaluate the phytochemical components of the plant, while 24 albino mice allotted to four groups of six mice each were used to study the effects of C. echinata on haematology. Mice in groups 1–3 were administered with 200, 100 and 50 mg/kg body weight of the extract respectively twice daily for 7 days, while group 4 mice received DW. Blood for haematology and vital organs for histomorphometric studies were harvested on day 8. One mouse died in group 7. Flavonoids, tannins, phenol, terpenes, glycosides and reducing sugars were abundantly present, while alkaloids and cardiac glycosides were present in moderate quantity. Resin and balsam were absent. There were no significant changes in haematological parameters. Mild degenerations were observed in the hepatocytes and renal tubular cells of group A mice. It was concluded that the extract may not be relatively very safe.













Similar content being viewed by others
References
Drury RA, Wallington A, Cameroun SR (1967) In Carlleton’s Histological Techniques. Oxford University Press, New York, pp 1–420
Fabricant DS, Farnsworths NR (2001) The value of plants used in traditional medicine for drug discovery. Environ Health Perspect Suppl 109(1):66–76
Ferguson LR (2001) Role of plant polyphenols in genomic stability. Mutat Res 475:89–111
Higgins T, Beutler E, Doumas BT (2008) Measurement of haemoglobin in blood. In: Burtis CA, Ashwood ER, Bruns DE (eds) Tietz fundamentals of clinical chemistry, 6th edn. Saunders Elsevier, Missouri, pp 514–515
Iwu MM (2013) Hand Book of African Medicinal Plants, 2nd edn. CRC Press, Taylor and Francis Group, Boca Raton, p 168
Magalhaes P, Appell H, Duarte J (2008) Involvement of advanced glycation end products in the pathogenesis of diabetes complication: the protective role of regular physical activity. Eur Rev Aging Phys Act 5(1):17–29
Mpetga JDS, Tene M, Wabo HK, Li SF, Kong LM, He HP, Hao XJ, Tane P (2012) Cytotoxic cycloartanes from fruits of Caloncoba glauca. Phytochem Lett 5:183–187
Nyenje M, Ndip RN (2011) In vitro antimicrobial activity of crude acetone extract of the stem bark of Combretum molle against selected bacterial pathogens. J Med Plant Res 5(21):53–56
Parasuraman S, Raveendran R, Kesavan R (2010) Blood sample collection in small laboratory animals. J Pharmacol Pharmacother 1(2):87–93
Soforowa EA (1993) Medicinal plants and traditional medicine in Africa 2nd edition. John and Wiley and Sons Ltd, England, pp 55–62
Spencer KC, Seigler DS (1985) Cyanogenic glycosides and the systematics of the Flacourtiaceae. Biochem Syst Ecol 13:421–431
Suleiman MM, Yusuf S (2008) Antidiarheal activity of the fruits of Vitex doniana in laboratory animals. Pharm Biol 46:387–392
Thrall MA, Weiser MG (2002) Haematology. In: Hendrix CM (ed) Laboratory procedures for veterinary technicians, 4th edition. Mosby Inc, Missouri, pp 29–74
Torell J, Cillard J, Cillard P (1986) Antioxidant activity of flavonoids and reactivity with peroxy radical. Phytochemistry 25(2):383–385
Trease GE, Evans WC (1989) Textbook of pharmacognosy, 14th edn. W.B Saunders, London, pp 40–47
Ziegler HL, Stark D, Christensen J, Olsen CE, Sittie AA, Jaroszewski JW (2002) New Dammarane and Malabaricane triterpenes from Caloncoba echinata. J Nat Prod 65:1764–1768
Funding
No funding was received for this study.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
Authors declare that they have no conflict of interest.
Ethical approval
All applicable international, national and/or institutional guidelines for the care and use of animals were followed.
Ethical statement
Authors hereby declare that “principles of laboratory animal care” (NIH publication No. 85-23, revised 1985) were followed, as well as specific national laws. All experiments have been examined and approved by the appropriate ethics committee.
Rights and permissions
About this article
Cite this article
Aba, P.E., Okorie-kanu, C.O. Preliminary phytochemical studies and biological effects of methanol extract of Caloncoba echinata in mice. Comp Clin Pathol 27, 1417–1425 (2018). https://doi.org/10.1007/s00580-018-2753-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00580-018-2753-y