Abstract
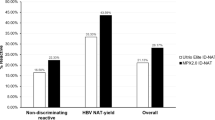
Blood transfusion is a life-saving approach, although it carries the risk of transfusion-transmitted viruses (TTVs). In Saudi Arabia (SA), nucleic acid amplification test (NAT) has been obligatory for HCV, HBV, and HIV by 2008. Our aim is to evaluate the effectiveness of NAT for detection of TTVs in the blood donated at Saudi German Hospitals (SGH) in SA. A total of 12,437 donor samples from SGH between January 2009 and June 2011 were subjected to routine serologic tests for HBV, HCV, and HIV followed by minipool NAT using Cobas Amplicor analyzer (Roche Diagnostics) for the seronegative samples. Out of 12,032 seronegative plasmas, five (0.042 %) were positive for HCV and two (0.017 %) for HIV-1 by NAT. None of our seronegative donors were NAT positive for HBV, highlighting the value of hepatitis B core antibody detection in donor testing. Our results provide evidence to support the effectiveness of NAT screening to detect TTVs in blood donations.
Similar content being viewed by others
References
Abdel Gader AM, Osman AA, Al Gahtani FH et al (2011) Attitude to blood donation in Saudi Arabia. Asian J Transfus Sci 5:121–126
Akhter J, Roberts GT, Perry A, Julie SG, Howman PA (2001) Use of nucleic acid testing for blood donor screening of human immunodeficiency virus and hepatitis C virus in the Saudi population. Saudi Med J 22(12):1073–1075
Alswaidi FM, O'Brien SJ (2009) Premarital screening programmes for haemoglobinopathies, HIV and hepatitis viruses: review and factors affecting their success. J Med Screen 16(1):22–28
Alswaidi FM, O'Brien SJ (2010) Is there a need to include HIV, HBV and HCV viruses in the Saudi premarital screening program on the basis of their prevalence and transmission risk factors? J Epid & Com Health 64:989–997
Ashshi AM (2012) Detection of occult hepatitis B virus in anti-HBc positive/anti-HBs positive blood donors in Saudi Arabia. Res J Med Sci 6(2):61–65
Bamaga MS, Bokhari FF, Aboud AM et al (2006) Nucleic acid amplification technology screening for hepatitis C virus and human immunodeficiency virus for blood donations. Saudi Med J 27(6):781–787
Biswas R, Tabor E, Hsia CC et al (2003) Comparative sensitivity of HBV NATs and HBsAg assays for detection of acute HBV infection. Transfusion 43:788–798
Busch MP, Glynn S, Stramer S, Strong DM, Caglioti S et al (2005) A new strategy for estimating risks of transfusion transmitted viral infections based on rates of infection of recently infected donors. Transfusion 45:254–264
Candotti D, El Chaar M, Allain J (2011) Transfusion transmission of hepatitis B virus: still learning more about it. ISBT Sci Ser 6:234–240
Center for Biologics Evaluation and Research (2009) Blood Products Advisory Committee, 94th meeting. Gaithersburg, April 1
Comanor L, Holland P (2006) Hepatitis B virus blood screening: unfinished agendas. Vox Sang 91:1–12
Dodd R, Notari E, Stramer S (2002) Current prevalence and incidence of infectious disease markers and estimated WP risk in the American Red Cross blood donor population. Transfusion 42:975–979
Duong Ly T, Laperche S, Brennan C et al (2004) Evaluation of the sensitivity and specificity of six HIV combined p24 antigen and antibody assays. J Virol Methods 122:185–194
Dwyre DM, Holland PV (2008) Hepatitis viruses. In: Barbara JAJ, Regan FAM, Contreras MC (eds) Transfusion microbiology. Cambridge University Press, Cambridge, pp 9–23
Dwyre DM, Fernando LP, Holland PV (2011) Hepatitis B, hepatitis C and HIV transfusion-transmitted infections in the 21st century. Vox Sang 100:92–98
El Chaar M, Candotti D, Crowther RA et al (2010) Impact of hepatitis B virus surface protein mutations on the diagnosis of occult hepatitis B virus infection. Hepatology 52:1600–1610
El-Hazmi M (2004) Prevalence of HBV, HCV, HIV-1, 2 and HTLV-I/II infections among blood donors in a teaching hospital in the central region of Saudi Arabia. Saudi Med J 25:26–33
Gerlich WH, Bremer C, Saniewski M, SchĂĽttler CG, Wend UC et al (2010) Occult hepatitis B virus infection: detection and significance. Dig Dis 28:116–125
Gonçales TT, Sabino EC, Salles NA et al (2009) HIV testing and confidential unit exclusion users in São Paulo. Brazil Transf 49(3):4A
Hajeer AH, Al Gharawi K, Al Rayes W et al (2008) Detection of HCV antibody-negative donations: Saudi experience with nucleic acid testing. British J Biomed Sci 65(2):103–104
Harritshoj LH, Dickmeiss E, Hansen MB et al (2008) Transfusion-transmitted HIV infection by a Danish blood donor with a very low viral load in the preseroconversion window phase. Transfusion 48:2026–2028
Kapoor D, Saxena R, Sood B, Sarin SK (2000) Blood transfusion practices in India: results of a national survey. Indian J Gastroenterol 19:64–67
Kleinman SH, Busch MP (2006) Assessing the impact of HBV NAT on window period reduction and residual risk. J Clin Vir 36(1):S23–S29
Kleinman SH, Strong DM, Tegtmeier G et al (2005) Hepatitis B virus (HBV) DNA screening of blood donations in minipools with the COBAS AmpliScreen HBV test. Transfusion 45:1247–1257
Kretzschmar E, Chudy M, Nuebling CM et al (2007) First case of hepatitis C virus transmission by a red blood cell concentrate after introduction of nucleic acid amplification technique screening in Germany: a comparative study with various assays. Vox Sang 92:297–301
Kuliya-Gwarzo A (2011) Screening for blood transfusion transmissible viruses in resource limited settings. The Internet J of Infectious Dis; 9(1). doi: 10.5580/132e
Laperche S (2005) Blood safety and nucleic acid testing in Europe. Euro Surveill 10(2):3–4
Makroo RN, Choudary N, Jagannathan L et al (2008) Multicenter evaluation of individual donor nucleic acid testing (NAT) for simultaneous detection of human immunodeficiency virus-1 & hepatitis B & C viruses in Indian blood donors. Ind J Med Res 127:140–147
Melve GK, Myrmel H, Eide GE et al (2009) Evaluation of the persistence and characteristics of indeterminate reactivity against hepatitis C virus in blood donors. Transfusion 49:2359–2365
Nübling CM, Heiden M, Chudy M, Kress J et al (2009) Experience of mandatory NAT screening across all blood organizations in Germany: NAT yield versus breakthrough transmissions. Transfusion 49:1850–1858
Phelps R, Robbins K, Liberti T et al (2004) Window-period human immunodeficiency virus transmission to two recipients by an adolescent blood donor. Transfusion 44:929–933
Quresh M, Mohammed AS, Malik S (2007) Seroprevalence of HBsAg, HCV and HIV antibodies in healthy individuals in Makkah region, KSA. Biomedica 23:12–16
Raghuraman S, Subramaniam T, Daniel D et al (2003) Occurrence of false positives during testing for antibodies to HCV among volunteer blood donors in India. J Cli Microbiol 41(4):1788–1790
Raimondo G, Allain JP, Brunetto MR et al (2008) Statements from the Taormina expert meeting on occult hepatitis B virus infection. J Hepatol 49:654–659
Scheiblauer H, Soboll H, Nick S (2006) Evaluation of 17 CE-marked HBsAg assays with respect to clinical sensitivity, analytical sensitivity, and hepatitis B virus mutant detection. J Med Virol 78(1):S66–S70
Shang G, Yan Y, Yang B et al (2009) Two HBV DNA+/HBsAg − blood donors identified by HBV NAT in Shenzhen. China Tran Apher Sci 41(1):3–7
Stramer SL (2007a) Current risks of transfusion-transmitted agents: a review. Arch Pathol Lab Med 131:702–707
Stramer SL (2007b) Current risks of transfusion-transmitted agents: a review. Archives of Path & Lab Med 131(5):702–707
Tomono T, Murokawa H, Minegishi K, Yamanaka R, Lizuka HY et al (2002) Status of NAT screening for HCV, HIV and HBV: experience in Japan. Dev Biol (Basel) 108:29–39
Velati C, Fomiatti L, Baruffi L et al (2005) Impact of nucleic acid amplification technology (NAT) in Italy in the three years following implementation (2001–2003). Euro Surveill 10:2–14
Wiedmann M, Kluwick S, Walter M et al (2007) HIV-1, HCV and HBV seronegative window reduction by the new Roche cobas® TaqScreen MPX test in seroconverting donors. J Clin Vir 39:282–287
World Health Organization (2009) Global blood safety and availability: facts and figures from the 2007 blood safety survey. Fact Sheet no. 279. WHO, Geneva
World Health Organization (2010) Screening donated blood from transfusion transmissible infections: recommendations. WHO, Geneva
Zekri AR, Awlia AA, El Mahalawi H, Ismail EF, Mabrouk GM (2002) Evaluation of blood units with isolated anti HBc for the presence of HBV DNA. Dis Markers 18(3):107–110
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Selim, H.M., ElBashaar, M.A. & ElWakil, S.G. Donor minipool NAT screening for HBV, HCV, and HIV: a 2-year experience in a private hospital in Saudi Arabia. Comp Clin Pathol 23, 1125–1132 (2014). https://doi.org/10.1007/s00580-013-1751-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00580-013-1751-3