Abstract
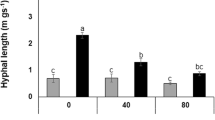
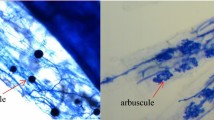
Three arbuscular mycorrhizal (AM) fungi (Glomus mosseae, Glomus claroideum, and Glomus intraradices) were compared for their root colonizing ability and activity in the root of Astragalus sinicus L. under salt-stressed soil conditions. Mycorrhizal formation, activity of fungal succinate dehydrogenase, and alkaline phosphatase, as well as plant biomass, were evaluated after 7 weeks of plant growth. Increasing the concentration of NaCl in soil generally decreased the dry weight of shoots and roots. Inoculation with AM fungi significantly alleviated inhibitory effect of salt stress. G. intraradices was the most efficient AM fungus compared with the other two fungi in terms of root colonization and enzyme activity. Nested PCR revealed that in root system of plants inoculated with a mix of the three AM fungi and grown under salt stress, the majority of mycorrhizal root fragments were colonized by one or two AM fungi, and some roots were colonized by all the three. Compared to inoculation alone, the frequency of G. mosseae in roots increased in the presence of the other two fungal species and highest level of NaCl, suggesting a synergistic interaction between these fungi under salt stress.




Similar content being viewed by others
Abbreviations
- ALP:
-
Alkaline phosphatase
- AM:
-
Arbuscular mycorrhizal
- LSU rDNA:
-
Large subunit ribosomal DNA
- PCR:
-
Polymerase chain reaction
- SDH:
-
Succinate dehydrogenase
References
Alkan N, Gadkar V, Yarden O, Kapulnik Y (2006) Analysis of quantitative interactions between two species of arbuscular mycorrhizal fungi, Glomus mosseae and G. intraradices, by real-time PCR. Appl Environ Microbiol 72:4192–4199
Al-Karaki GN (2000) Growth and mineral acquisition by mycorrhizal tomato grown under salt stress. Mycorrhiza 10:51–54
Al-Karaki GN, Hammad R, Rusan M (2001) Response of two tomato cultivars differing in salt tolerance to inoculation with mycorrhizal fungi under salt stress. Mycorrhiza 11:43–47
Chen XH, Zhao B (2007) Arbuscular mycorrhizal fungi mediated uptake of lanthanum in Chinese milk vetch (Astragalus sinicus L.). Chemosphere 68:1548–1555
Cho K, Toler H, Lee J, Ownley B, Stutz JC, Moore JL, Augé RM (2006) Mycorrhizal symbiosis and response of sorghum plants to combined drought and salinity stresses. J Plant Physiol 163:517–528
Daei G, Ardekani MR, Rejali F, Teimuri S, Miransari M (2009) Alleviation of salinity stress on wheat yield, yield components, and nutrient uptake using arbuscular mycorrhizal fungi under field conditions. J Plant Physiol 166:617–625
Dong XL, Zhao B (2006) Nested multiplex PCR—a feasible technique to study partial community of arbuscular mycorrhizal fungi in field-growing plant root. Sci China Ser C-Life Sci 49:354–361
El-Atrach F, Vierheilig H, Ocampo JA (1989) Influence of non-host plants on vesicular-arbuscular mycorrhizal infection of host plants and on spore germination. Soil Biol Biochem 21:161–163
Ghazi N, Al-Karaki (2000) Growth of mycorrhizal tomato and mineral acquisition under salt stress. Mycorrhiza 10:51–54
Gollotte A, van Tuinen D, Atkinson D (2004) Diversity of arbuscular mycorrhizal fungi colonising roots of the grass species Agrostis capillaris and Lolium perenne in a field experiment. Mycorrhiza 14:111–117
He ZQ, He CX, Zhang ZB, Zou ZR, Wang HS (2007) Changes of antioxidative enzymes and cell membrane osmosis in tomato colonized by arbuscular mycorrhizae under NaCl stress. Colloids Surf B 59:128–133
Hempel S, Renker C, Buscot F (2007) Differences in the species composition of arbuscular mycorrhizal fungi in spore, root and soil communities in a grassland ecosystem. Environ Microbiol 9(8):1930–1938
Hoagland DR, Arnon DI (1950) The water-culture method for growing plants without soil. Calif Agric Exp Stn Cir 347:32–37
Jacquot E, van Tuinen D, Gianinazzi S, Gianinazzi-Pearson V (2000) Monitoring species of arbuscular mycorrhizal fungi in plants and in soil by nested PCR: application to the study of the impact of sewage sludge. Plant Soil 226:179–188
Jiang XY, Huang Y (2003) Mechanism of contribution of mycorrhizal fungi to plant saline-alkali tolerance. Ecol Environ 12:353–356
Kjøller R, Rosendahl S (2000) Detection of arbuscular mycorrhizal fungi (Glomales) in roots by nested PCR SSCP (single stranded conformation polymorphism). Plant Soil 226:189–196
Koide RT (2000) Functional complementarity in the arbuscular mycorrhizal symbiosis. New Phytol 147:233–235
Li Y, Peng J, Shi P, Zhao B (2009) The effect of Cd on mycorrhizal development and enzyme activity of Glomus mosseae and Glomus intraradices in Astragalus sinicus L. Chemosphere 75:894–899
Naidoo G, Naidoo Y (2001) Effects of salinity and nitrogen on growth, ion relations and proline accumulation in Triglochinbulbosa. Wetlands Ecol Manage 9:491–497
Niu DL, Wang QJ (2002) Research progress on saline—alkali field control. Chinese J Soil Sci 33:449–455 (in Chinese)
Porras-Soriano A, Soriano-Martin ML, Porras-Piedra A, Azcón R (2009) Arbuscular mycorrhizal fungi increased growth, nutrient uptake and tolerance to salinity in olive trees under nursery conditions. J Plant Physiol 166:1350–1359
Porter WM (1979) The “Most Probable Number” method for enumerating infective propagules of VAM fungi in soil. Aust J Soil Res 17:515–519
Reddy SR, Pindi PK, Reddy SM (2005) Molecular methods for research on arbuscular mycorrhizal fungi in India: problems and prospects. Curr Sci 89:1699–1709
Renker C, Heinrichs J, Kaldorf M, Buscot F (2003) Combining nested PCR and restriction digest of the internal transcribed spacer region to characterize arbuscular mycorrhizal fungi on roots from the field. Mycorrhiza 13:191–198
Rosendahl S, Stukenbrock EH (2004) Community structure of arbuscular mycorrhizal fungi in undisturbed vegetation revealed by analyses of LSU rDNA sequences. Mol Ecol 13:3179–3186
Stukenbrock EH, Rosendahl S (2005) Distribution of dominant arbuscular mycorrhizal fungi among five plant species in undisturbed vegetation of a coastal grassland. Mycorrhiza 15:497–503
Tisserant B, Gianinazzi-Pearson V, Gianinazzi S, Gollotte A (1993) In planta histochemical staining of fungal alkaline phosphatase activity for analysis of efficient arbuscular mycorrhizal infections. Mycol Res 97:245–250
Triantafilis J, Odeh IOA, McBratney AB (2001) Five geostatistical models to predict soil salinity from electromagnetic induction data across irrigated cotton. Soil Sci Soc Am J 65:869–878
Trouvelot A, Kough J, Gianinazzi-Pearson V (1986) Evaluation of VAM infection levels in root systems. Research for estimation methods having a functional significance. In: Gianinazzi-Pearson V, Gianinazzi S (eds) Physiological and Genetical Aspects of Mycorrhizae. INRA, Paris, pp 217–221
van Tuinen D, Jacquot E, Zhao B, Gollotte A, Gianinazzi-Pearson V (1998) Characterization of root colonization profiles by a microcosm community of arbuscular mycorrhizal fungi using 25S rDNA-targeted nested PCR. Mol Ecol 7:879–887
Zhao B, Trouvelot A, Gianinazzi S, Gianinazzi-Pearson V (1997) Influence of two legume species on hyphal production and activity of two arbuscular mycorrhizal fungi. Mycorrhiza 7:179–185
Acknowledgments
The authors are grateful to Prof. Lyn. Abbott (UWA, Australia) for the invaluable advice and to Dr. V. Gianinazzi-Pearson (INRA, Dijon, France) for the critical review of this manuscript. This study was partly supported by the National Natural Science Foundation of China (grant no. 30270051 and grant no. 4004-084011).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Peng, J., Li, Y., Shi, P. et al. The differential behavior of arbuscular mycorrhizal fungi in interaction with Astragalus sinicus L. under salt stress. Mycorrhiza 21, 27–33 (2011). https://doi.org/10.1007/s00572-010-0311-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00572-010-0311-9