Abstract
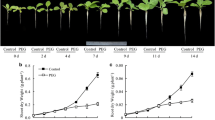
We investigated in two tobacco (Nicotiana tabacum) plant lines (wildtype or antisense mutant) whether impairment in expression of the plasma membrane aquaporin gene (NtAQP1) affects the arbuscular mycorrhizal (AM) fungal colonisation pattern or the symbiotic efficiency of AM fungi. These two objectives were investigated under well-watered and drought stress conditions. Both plant lines had a similar pattern of root colonisation under well-watered and drought stress conditions. In contrast, under drought stress, AM wildtype plants grew faster than mycorrhizal antisense plants. Plant gas exchange also appeared to depend on the expression of NtAQP1 and parallelled the determined growth increments. The implications of enhanced symplastic water transport via NtAQP1 for the efficiency of the AM symbiosis under drought stress conditions are further discussed.



Similar content being viewed by others
References
Aharon R, Shahak Y, Wininger S, Bendov R, Kapulnik Y, Galili G (2003) Overexpression of plasma membrane aquaporins in transgenic tobacco improves plant vigour under favourable growth conditions but not under drought or salt stress. Plant Cell 15:439–447
Augé RM (2001) Water relations, drought and vesicular-arbuscular mycorrhizal symbiosis. Mycorrhiza 11:3–42
Barrieu F, Chaumont F, Chrispeels MJ (1998) High expression of the tonoplast aquaporin ZmTIP1 in epidermal and conducting tissues of maize. Plant Physiol 117:1153–1163
Bates LS, Waldren RP, Teare ID (1973) Rapid determination of free proline for water stress studies. Plant Soil 39:205–207
Biela A, Grote K, Otto B, Hoth S, Hedrich R, Kaldenhoff R (1999) The Nicotiana tabacum plasma membrane aquaporin NtAQP1 is mercury-insensitive and permeable for glycerol. Plant J 18:565–570
Bligh EG, Dyer WJ (1959) A rapid method of total lipid extraction and purification. Can J Biochem Physiol 37:911–917
Bonfante P, Perotto S (1995) Strategies of arbuscular mycorrhizal fungi when infecting host plants. New Phytol 130:3–21
Chrispeels MJ, Agre P (1994) Aquaporins: water channel proteins of plant and animal cells. Trends Biochem Sci 19:421–425
Duncan DB (1955) Multiple range and multiple F-tests. Biometrics 11:1–42
Eckert M, Biela A, Siefritz F, Kaldenhoff R (1999) New aspects of plant aquaporin regulation and specificity. J Exp Bot 50:1541–1545
Gianinazzi-Pearson V (1996) Plant cell response to arbuscular mycorrhizal fungi: getting to the roots of the symbiosis. Plant Cell 8:1871–1883
Graham JH, Drouillard DL, Hodge NC (1996) Carbon economy of sour orange in response to different Glomus spp. Tree Physiol 16:1023–1029
Grote K, Trzebiatovski P, Kaldenhoff R (1998) RNA levels of plasma membrane aquaporins in Arabidopsis thaliana. Protoplasma 204:139–144
Irigoyen JJ, Emerich DW, Sánchez-Díaz M (1992) Water stress induced changes in concentrations of proline and total soluble sugars in nodulated alfalfa (Medicago sativa) plants. Physiol Plant 84:67–72
Javot H, Lauvergeat V, Santoni V, Martin-Laurent F, Guclu J, Vinh J, Heyes J, Franck KI, Schaffner AR, Bouchez D, Maurel C (2003) Role of a single aquaporin isoform in root water uptake. Plant Cell 15:509–522
Johansson I, Karlsson M, Johansson U, Larsson C, Kjellbom P (2000) The role of aquaporins in cellular and whole plant water balance. Biochim Biophys Acta 1465:324–342
Johnson KD, Höfte H, Chrispeels MJ (1990) An intrinsic tonoplast protein of proteins storage vacuoles in seeds is structurally related to a bacterial solute transporter (GlpF). Plant Cell 2:525–532
Kameli A, Lösel DM (1993) Carbohydrates and water status in wheat plants under water stress. New Phytol 125:609–614
Kammerloher W, Fischer U, Pienchottka GP, Schäffner AR (1994) Water channels in the plant plasma membrane cloned by immunoselection from a mammalian expression system. Plant J 6:187–199
Kjellbom P, Larsson C, Johansson I, Karlsson M, Johanson U (1999) Aquaporins and water homeostasis in plants. Trends Plant Sci 4:308–314
Krajinski F, Biela A, Schubert D, Gianinazzi-Pearson V, Kaldenhoff R, Franken P (2000) Arbuscular mycorrhiza development regulates the mRNA abundance of Mtaqp1 encoding a mercury-insensitive aquaporin of Medicago truncatula. Planta 211:85–90
Kramer PJ, Boyer JS (1997) Water relations of plants and soils. Academic, San Diego
Long SP, Hällgren JE (1987) Measurement of CO2 assimilation by plants in the field and the laboratory. In: Combs J, Hall DO, Long SP, Scurlock JMO (eds) Techniques in bioproductivity and photosynthesis, 2nd edn. Pergamon, Oxford, pp 62–94
Martre P, Morillon R, Barrieu F, North GB, Nobel PS, Chrispeels MJ (2002) Plasma membrane aquaporins play a significant role during recovery from water deficit. Plant Physiol 130:2101–2110
Maurel C, Javot H, Lauvergeat V, Gerbeau P, Tournaire C, Santoni V, Heyes J (2003) Molecular physiology of aquaporins in plants. Int Rev Cytol 215:105–148
Murashige T, Skoog F (1962) A revised medium for rapid growth and bioassays with tobacco tissue cultures. Physiol Plant 15:473–497
Otto B, Kaldenhoff R (2000) Cell-specific expression of the mercury-insensitive plasma-membrane aquaporin NtAQP1 from Nicotiana tabacum. Planta 211:167–172
Phillips JM, Hayman DS (1970) Improved procedure of clearing roots and staining parasitic and vesicular-arbuscular mycorrhizal fungi for rapid assessment of infection. Trans Br Mycol Soc 55:159–161
Roth CH, Malicki MA, Plagge R (1992) Empirical evaluation of the relationship between soil dielectric constant and volumetric water content as the basis for calibrating soil moisture measurements. J Soil Sci 43:1–13
Roussel H, Bruns S, Gianinazzi-Pearson V, Hahlbrock K, Franken P (1997) Induction of a membrane intrinsic protein-encoding mRNA in arbuscular mycorrhiza and elicitor-stimulated cell suspension cultures of parsley. Plant Sci 126:203–210
Ruiz-Lozano JM (2003) Arbuscular mycorrhizal symbiosis and alleviation of osmotic stress. New perspectives for molecular studies. Mycorrhiza 13:309–317
Siefritz F, Tyree MT, Lovisolo C, Schubert A, Kaldenhoff R (2002) PIP1 plasma membrane aquaporins in tobacco: from cellular effects to function in plants. Plant Cell 14:869–876
Siefritz F, Otto B, van der Krol A, Kaldenhoff R (2004) The plasma membrane aquaporin NtAQP1 is a key component of the leaf movement mechanism in tobacco. Plant J 37:147–155
Smith SE, Read DJ (1997) Mycorrhizal symbiosis. Academic, San Diego
Trouvelot A, Kough JL, Gianinazzi-Pearson V (1986) Mesure du taux de mycorhization VA d’un système radiculaire. Recherche de méthodes d’estimation ayant une signification fonctionnelle. In: Gianinazzi-Pearson V, Gianinazzi S (eds) Physiological and genetical aspects of mycorrhizae. INRA Press, Paris, pp 217–221
Uehlein N, Lovisolo C, Siefritz F, Kaldenhoff R (2003) The tobacco aquaporin NtAQP1 is a membrane CO2 pore with physiological functions. Nature 425:734–737
White I, Knight JH, Zegelin SJ, Topp GC (1994) Comments to “Considerations on the use of time-domain reflectometry (TDR) for measuring soil water content” by WR Whalley. J Soil Sci 45:503–508
Acknowledgement
This work and R. Porcel were financed by CICYT-FEDER (Project AGL2002-03952).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Porcel, R., Gómez, M., Kaldenhoff, R. et al. Impairment of NtAQP1 gene expression in tobacco plants does not affect root colonisation pattern by arbuscular mycorrhizal fungi but decreases their symbiotic efficiency under drought. Mycorrhiza 15, 417–423 (2005). https://doi.org/10.1007/s00572-005-0346-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00572-005-0346-5