Abstract
Purpose
Volatile anesthetics are a potential risk for cognitive impairment in the developing brain. Isoflurane causes cell death, reduces neurogenesis, and changes neuronal differentiation. In this study, the effects of a single isoflurane exposure on the developing human brain were evaluated using human embryonic stem cell (hESC)-derived neural progenitor cells (NPCs).
Methods
Multipotent NPCs were derived from hESCs and randomly exposed to either 5 vol% isoflurane (4 h) or no isoflurane (control group). The cells were fixed after 1, 3, 5, and 7 days to evaluate differentiation using the ratio of β-III tubulin to nestin. Neuronal cell survival and proliferation were assessed using the WST-1 and bromodeoxyuridine (BrdU) cell proliferation assays, respectively. To evaluate the mechanism of isoflurane neurotoxicity, we added TAT-Pep5, a p75 neurotrophic receptor (p75NTR) inhibitor, to each of the groups.
Results
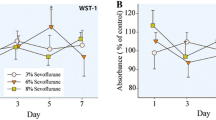
Isoflurane had minimal or no effect on the cell survival of NPCs in vitro. Proliferation, assessed on the basis of BrdU incorporation, was inhibited in the isoflurane group on days 3 (p = 0.033) and 5 (p = 0.001). The ratios of β-III tubulin to nestin in the isoflurane group on days 1 and 3 were significantly lower (p = 0.004 and p = 0.029, respectively) than those in the control group. Anti-proliferative and differentiation-reducing effect did not persist. TAT-Pep5 pretreatment significantly blocked the isoflurane-mediated decrease in the β-III tubulin to nestin ratio (p = 0.012) on day 1.
Conclusion
Exposing NPCs to isoflurane hampered proliferation and differentiation but not neuronal survival. Isoflurane may be a powerful neuronal modulator during the early developmental period, partly mediated by activation of p75NTR.




Similar content being viewed by others
References
Jevtovic-Todorovic V, Hartman RE, Izumi Y, Benshoff ND, Dikranian K, Zorumski CF, Olney JW, Wozniak DF. Early exposure to common anesthetic agents causes widespread neurodegeneration in the developing rat brain and persistent learning deficits. J Neurosci. 2003;23:876–82.
Zhu C, Gao J, Karlsson N, Li Q, Zhang Y, Huang Z, Li H, Kuhn HG, Blomgren K. Isoflurane anesthesia induced persistent, progressive memory impairment, caused a loss of neural stem cells, and reduced neurogenesis in young, but not adult, rodents. J Cereb Blood Flow Metab. 2010;30:1017–30.
Sall JW, Stratmann G, Leong J, McKleroy W, Mason D, Shenoy S, Pleasure SJ, Bickler PE. Isoflurane inhibits growth but does not cause cell death in hippocampal neural precursor cells grown in culture. Anesthesiology. 2009;110:826–33.
Loepke AW, Soriano SG. An assessment of the effects of general anesthetics on developing brain structure and neurocognitive function. Anesth Analg. 2008;106:1681–707.
Stratmann G, Sall JW, May LD, Bell JS, Magnusson KR, Rau V, Visrodia KH, Alvi RS, Ku B, Lee MT, Dai R. Isoflurane differentially affects neurogenesis and long-term neurocognitive function in 60-day-old and 7-day-old rats. Anesthesiology. 2009;110:834–48.
Sun L. Early childhood general anaesthesia exposure and neurocognitive development. Br J Anaesth. 2010;105[Suppl 1]:i61–8.
DiMaggio C, Sun LS, Kakavouli A, Byrne MW, Li G. A retrospective cohort study of the association of anesthesia and hernia repair surgery with behavioral and developmental disorders in young children. J Neurosurg Anesthesiol. 2009;21:286–91.
Dhara SK, Stice SL. Neural differentiation of human embryonic stem cells. J Cell Biochem. 2008;105:633–40.
Trujillo CA, Schwindt TT, Martins AH, Alves JM, Mello LE, Ulrich H. Novel perspectives of neural stem cell differentiation: from neurotransmitters to therapeutics. Cytom A. 2009;75:38–53.
Head BP, Patel HH, Niesman IR, Drummond JC, Roth DM, Patel PM. Inhibition of p75 neurotrophin receptor attenuates isoflurane-mediated neuronal apoptosis in the neonatal central nervous system. Anesthesiology. 2009;110:813–25.
Oh SK, Kim HS, Park YB, Seol HW, Kim YY, Cho MS, Ku SY, Choi YM, Kim DW, Moon SY. Methods for expansion of human embryonic stem cells. Stem Cells. 2005;23:605–9.
Cho MS, Hwang DY, Kim DW. Efficient derivation of functional dopaminergic neurons from human embryonic stem cells on a large scale. Nat Protoc. 2008;3:1888–94.
Kim JH, Oh AY, Choi YM, Ku SY, Kim YY, Lee NJ, Sepac A, Bosnjak ZJ. Isoflurane decreases death of human embryonic stem cell-derived, transcriptional marker Nkx2.5(+) cardiac progenitor cells. Acta Anaesthesiol Scand. 2011;55:1124–31.
Lehmann J, Retz M, Sidhu SS, Suttmann H, Sell M, Paulsen F, Harder J, Unteregger G, Stockle M. Antitumor activity of the antimicrobial peptide magainin II against bladder cancer cell lines. Eur Urol. 2006;50:141–7.
Michalczyk K, Ziman M. Nestin structure and predicted function in cellular cytoskeletal organisation. Histol Histopathol. 2005;20:665–71.
Lemkuil BP, Head BP, Pearn ML, Patel HH, Drummond JC, Patel PM. Isoflurane neurotoxicity is mediated by p75NTR-RhoA activation and actin depolymerization. Anesthesiology. 2011;114:49–57.
Culley DJ, Boyd JD, Palanisamy A, Xie Z, Kojima K, Vacanti CA, Tanzi RE, Crosby G. Isoflurane decreases self-renewal capacity of rat cultured neural stem cells. Anesthesiology. 2011;115:754–63.
Jevtovic-Todorovic V, Boscolo A, Sanchez V, Lunardi N. Anesthesia-induced developmental neurodegeneration: the role of neuronal organelles. Front Neurol. 2012;3:141.
Sakai H, Sheng H, Yates RB, Ishida K, Pearlstein RD, Warner DS. Isoflurane provides long-term protection against focal cerebral ischemia in the rat. Anesthesiology. 2007;106:92–9.
LoTurco JJ, Owens DF, Heath MJ, Davis MB, Kriegstein AR. GABA and glutamate depolarize cortical progenitor cells and inhibit DNA synthesis. Neuron. 1995;15:1287–98.
Gould E, Tanapat P, McEwen BS, Flugge G, Fuchs E. Proliferation of granule cell precursors in the dentate gyrus of adult monkeys is diminished by stress. Proc Natl Acad Sci USA. 1998;95:3168–71.
Lin N, Moon TS, Stratmann G, Sall JW. Biphasic change of progenitor proliferation in dentate gyrus after single dose of isoflurane in young adult rats. J Neurosurg Anesthesiol. 2013;25:306–10.
Barberi T, Klivenyi P, Calingasan NY, Lee H, Kawamata H, Loonam K, Perrier AL, Bruses J, Rubio ME, Topf N, Tabar V, Harrison NL, Beal MF, Moore MA, Studer L. Neural subtype specification of fertilization and nuclear transfer embryonic stem cells and application in parkinsonian mice. Nat Biotechnol. 2003;21:1200–7.
Wang C, Slikker W Jr. Strategies and experimental models for evaluating anesthetics: effects on the developing nervous system. Anesth Analg. 2008;106:1643–58.
Zhao X, Yang Z, Liang G, Wu Z, Peng Y, Joseph DJ, Inan S, Wei H. Dual effects of isoflurane on proliferation, differentiation, and survival in human neuroprogenitor cells. Anesthesiology. 2013;118(3):537–49.
Arnold JH, Truoq RD, Rice SA. Prolonged administration of isoflurane to pediatric patients during mechanical ventilation. Anesth Analg. 1993;76(3):520–6.
McBeth C, Watkins TG. Isoflurane for sedation in a case of congenital myasthenia gravis. Br J Anaesth. 1996;77(5):672–4.
Kelsall AW, Ross-Russell R, Herrick MJ. Reversible neurologic dysfunction following isoflurane sedation in pediatric intensive care. Crit Care Med. 1994;22(6):1032–4.
Newey SE, Velamoor V, Govek EE, Van Aelst L. Rho GTPases, dendritic structure, and mental retardation. J Neurobiol. 2005;64:58–74.
Orliaguet G, Vivien B, Langeron O, Bouhemad B, Coriat P, Riou B. Minimum alveolar concentration of volatile anesthetics in rats during postnatal maturation. Anesthesiology. 2001;95:734–9.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Funding
This work was supported by Grant 02-2012-029 from the Seoul National University Bundang Hospital Research Fund, Republic of Korea.
Conflict of interest
None.
Additional information
H.-M. Sohn and H. Y. Kim contributed equally as co-first authors.
Electronic supplementary material
Below is the link to the electronic supplementary material.
About this article
Cite this article
Sohn, HM., Kim, H.Y., Park, S. et al. Isoflurane decreases proliferation and differentiation, but none of the effects persist in human embryonic stem cell-derived neural progenitor cells. J Anesth 31, 36–43 (2017). https://doi.org/10.1007/s00540-016-2277-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00540-016-2277-z