Abstract
Purpose
When neostigmine is used to reverse muscle relaxants in patients with asthma without signs of airway inflammation, asthma attack is occasionally encountered. It is likely that abnormally increased electrical impulses traveling from the brain through cholinergic nerves to airway smooth muscles may be one of the pathogeneses of asthma attack. We applied continuous electrical field stimulation (c-EFS) or continuous electrical stimulation (c-ES) of low frequency to the vagal nerve of the rat in vitro and in vivo to determine the role of cholinergic nerve activation in inducing airway constriction.
Methods
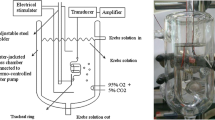
Fifty-seven male Wistar rats were used. In an in vitro study we examined whether tetrodotoxin (TTX), an Na+-channel blocker, 4-DAMP, a muscarinic M3 receptor antagonist, or neostigmine could affect c-EFS-induced contraction of the tracheal ring. In an in vivo study, we examined whether c-ES of the vagal nerve could increase maximum airway pressure (P max) and whether neostigmine could potentiate c-ES-induced P max.
Results
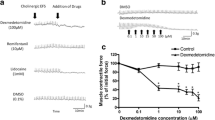
TTX and 4-DAMP completely inhibited c-EFS-induced contraction whereas neostigmine potentiated c-EFS-induced contraction dose-dependently. P max was not increased by neostigmine. P max was not increased by 2-Hz c-ES, but was increased by the addition of neostigmine. P max was increased by 5-Hz c-ES, and further increased by the addition of neostigmine.
Conclusion
The contractile response of the tracheal ring to c-EFS is potentiated by neostigmine. P max is increased by c-ES of the vagal nerve, and is potentiated by neostigmine. These data suggest that increased activity of the cholinergic nerve could be involved in asthma attack.







Similar content being viewed by others
References
Hoang BX, Levine SA, Graeme Shaw D, Pham P, Hoang C. Bronchial epilepsy or broncho-pulmonary hyper-excitability as a model of asthma pathogenesis. Med Hypotheses. 2006;67:1042–51.
Jain S, Jain KC. Effect of phenytoin sodium in the management of poorly controlled bronchial asthma at a rural health center in Phalodi, Rajasthan, India. J Asthma. 1991;28:201–11.
Sayar B, Polvan O. Epilepsy and bronchial asthma. Lancet. 1968;1:1038.
Nishioka K, Shibata O, Yamaguchi M, Yoshimura M, Makita T, Sumikawa K. Succinylcholine potentiates acetylcholine-induced contractile and phosphatidylinositol responses of rat trachea. J Anesth. 2007;21:171–5.
Yamaguchi M, Shibata O, Nishioka K, Makita T, Sumikawa K. Propofol attenuates ovalbumin-induced smooth muscle contraction of the sensitized rat trachea: inhibition of serotonergic and cholinergic signaling. Anesth Analg. 2006;103:594–600.
Lefkowitz RJ, Hoffman BB, Taylor P. Neurohumoral transmission: the autonomic and somatic motor nerve systems. In: Gilman AG, editor. Goodman and Gilman’s the pharmacological basis of therapeutics. 8th ed. New York: Pergamon Press Inc; 1990. p. 97.
Toda N, Hatano Y. Contractile responses of canine tracheal muscle during exposure to fentanyl and morphine. Anesthesiology. 1980;53:93–100.
Maclagan J, Barnes PJ. Muscarinic pharmacology of the airways. Trends Pharmacol Sci. 1989; Suppl:88–92.
Ten Berge REJ, Roffel AdF, Zaagsma J. The interaction of selective and non-selective antagonists with pre- and postjunctional muscarinic receptor subtypes in the guinea pig trachea. Eur J Pharmacol. 1993;233:279–84.
Kirkpatrick CT, Rooney PJ. Contractures produced by carbamate anticholinesterases in bovine tracheal smooth muscle. Clin Exp Pharmacol Physiol. 1982;9:603–11.
Shibata O, Tsuda A, Makita T, Iwanaga S, Hara T, Shibata S, Sumikawa K. Contractile and phosphatidylinositol responses of rat trachea to anticholinesterase drugs. Can J Anaesth. 1998;45:1190–5.
Hazizaj A, Hatija A. Bronchospasm caused by neostigmine. Eur J Anaesthesiol. 2006;23:85–6.
Gouge SF, Daniels DJ, Smith CE. Exacerbation of asthma after pyridostigmine during Operation Desert Storm. Mil Med. 1994;159:108–11.
Author information
Authors and Affiliations
Corresponding author
About this article
Cite this article
Ishii, K., Shibata, O., Nishioka, K. et al. Effects of neostigmine on bronchoconstriction with continuous electrical stimulation in rats. J Anesth 26, 80–84 (2012). https://doi.org/10.1007/s00540-011-1258-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00540-011-1258-5