Abstract
Background
Progressive hepatic fibrosis leads to hepatocellular carcinoma (HCC) and decompensated cirrhosis. The aim of this study was to identify the high-risk population for progressive hepatic fibrosis and the incidence of HCC and decompensated cirrhosis in chronic hepatitis B (CHB) patients with antiviral therapy.
Methods
The data came from a multicenter, center-randomized, double-blind clinical trial that analyzed only patients in the ETV-treated arm. There was 156 hepatitis B e antigen (HBeAg)-positive and 135 HBeAg-negative patients in 14 institutions. The primary endpoint was fibrosis reversal on 72-week Entecavir (ETV) treatment. The 7-year cumulative incidence of HCC and decompensated cirrhosis were analyzed. Multivariate logistic and LASSO regression analyses were used to screen variables associated with fibrosis reversal.
Results
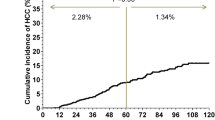
86/156 (55%) HBeAg-positive and 58/135 (43%) HBeAg-negative patients achieved fibrosis reversal on 72-week ETV treatment. Average age was 43 years, 203 (69.8%) was male, and 144 (49.5%) patients had cirrhosis. Age ≥ 40 years (OR: 0.46, 95% CI 0.23–0.93) and HBcrAg ≥ 8.23 log U/ml (OR: 2.72, 95% CI 1.33–5.54) in HBeAg-positive patients and HBV genotype C (OR: 0.44, 95% CI 0.21–0.97) in HBeAg-negative patients were independent factors of fibrosis reversal. It was confirmed in patients with cirrhosis. After 7-year ETV treatment, seven (4.5%) HBeAg-positive patients occurred HCC or decompensated cirrhosis, including four patients with age ≥ 40 years and six with HBcrAg 8.23log U/ml, while twelve (8.9%) HBeAg-negative patients occurred, including eleven with HBV genotype C.
Conclusions
HBeAg-positive patients with a low HBcrAg level or old age, and HBeAg-negative patients with HBV genotype C tended to develop progressive hepatic fibrosis and had a high incidence of HCC and decompensated cirrhosis, even on ETV treatment.




Similar content being viewed by others
References
Péneau C, Imbeaud S, La Bella T, et al. Hepatitis B virus integrations promote local and distant oncogenic driver alterations in hepatocellular carcinoma. Gut. 2022;71:616–26.
Marcellin P, Asselah T. Long-term therapy for chronic hepatitis B: hepatitis B virus DNA suppression leading to cirrhosis reversal. J Gastroenterol Hepatol. 2013;28:912–23.
Rong G, Chen Y, Yu Z, et al. Synergistic effect of Biejia-Ruangan on fibrosis regression in patients with chronic hepatitis B treated with entecavir: a multicenter, randomized, double-blind, placebo-controlled trial. J Infect Dis. 2022;225:1091–9.
Tong XF, Wang QY, Zhao XY, et al. Histological assessment based on liver biopsy: the value and challenges in NASH drug development. Acta Pharmacol Sin. 2022;43:1200–9.
Brunt EM. Liver biopsy reliability in clinical trials: thoughts from a liver pathologist. J Hepatol. 2020;73:1310–2.
Karlas T, Petroff D, Sasso M, et al. Impact of controlled attenuation parameter on detecting fibrosis using liver stiffness measurement. Aliment Pharmacol Ther. 2018;47:989–1000.
Kim WR, Berg T, Asselah T, et al. Evaluation of APRI and FIB-4 scoring systems for non-invasive assessment of hepatic fibrosis in chronic hepatitis B patients. J Hepatol. 2016;64:773–80.
Martinot-Peignoux M, Carvalho-Filho R, Lapalus M, et al. Hepatitis B surface antigen serum level is associated with fibrosis severity in treatment-naive, e antigen-positive patients. J Hepatol. 2013;58:1089–95.
Seto WK, Wong DK, Fung J, et al. High hepatitis B surface antigen levels predict insignificant fibrosis in hepatitis B e antigen positive chronic hepatitis B. PLoS One. 2012;7: e43087.
Tada T, Kumada T, Toyoda H, et al. HBcrAg predicts hepatocellular carcinoma development: an analysis using time-dependent receiver operating characteristics. J Hepatol. 2016;65:48–56.
Testoni B, Lebossé F, Scholtes C, et al. Serum hepatitis B core-related antigen (HBcrAg) correlates with covalently closed circular DNA transcriptional activity in chronic hepatitis B patients. J Hepatol. 2019;70:615–25.
Huang D, Wu D, Wang P, et al. End-of-treatment HBcrAg and HBsAb levels identify durable functional cure after Peg-IFN-based therapy in patients with CHB. J Hepatol. 2022;77:42–54.
Tseng TC, Liu CJ, Yang HC, et al. Higher proportion of viral basal core promoter mutant increases the risk of liver cirrhosis in hepatitis B carriers. Gut. 2015;64:292–302.
Chan HL, Wong ML, Hui AY, et al. Hepatitis B virus genotype C takes a more aggressive disease course than hepatitis B virus genotype B in hepatitis B e antigen-positive patients. J Clin Microbiol. 2003;4:1277–9.
Qu J, Yu Z, Li Q, et al. Blocking and reversing hepatic fibrosis in patients with chronic hepatitis B treated by traditional Chinese medicine (tablets of biejia ruangan or RGT): study protocol for a randomized controlled trial. Trials. 2014;15:438.
Ji D, Chen Y, Bi J, et al. Entecavir plus Biejia-Ruangan compound reduces the risk of hepatocellular carcinoma in Chinese patients with chronic hepatitis B. J Hepatol. 2022;77:1515–24.
Heimbach JK, Kulik LM, Finn RS, et al. AASLD guidelines for the treatment of hepatocellular carcinoma. Hepatology. 2018;67:358–80.
Marcellin P, Gane E, Buti M, et al. Regression of cirrhosis during treatment with tenofovir disoproxil fumarate for chronic hepatitis B: a 5-year open-label follow-up study. Lancet (London, England). 2013;381:468–75.
WHO Guidelines Approved by the Guidelines Review Committee. Guidelines for the prevention, care and treatment of persons with chronic hepatitis B infection. Geneva: World Health Organization; 2015.
Rockey DC, Caldwell SH, Goodman ZD, et al. Liver biopsy. Hepatology. 2009;49:1017–44.
Ishak K, Baptista A, Bianchi L, et al. Histological grading and staging of chronic hepatitis. J Hepatol. 1995;22:696–9.
Wong DK, Tanaka Y, Lai CL, et al. Hepatitis B virus core-related antigens as markers for monitoring chronic hepatitis B infection. J Clin Microbiol. 2007;45:3942–7.
Kimura T, Rokuhara A, Sakamoto Y, et al. Sensitive enzyme immunoassay for hepatitis B virus core-related antigens and their correlation to virus load. J Clin Microbiol. 2002;40:439–45.
Xu Z, Ren X, Liu Y, et al. Association of hepatitis B virus mutations in basal core promoter and precore regions with severity of liver disease: an investigation of 793 Chinese patients with mild and severe chronic hepatitis B and acute-on-chronic liver failure. J Gastroenterol. 2011;46:391–400.
Graupera I, Thiele M, Serra-Burriel M, et al. Low accuracy of FIB-4 and NAFLD fibrosis scores for screening for liver fibrosis in the population. Clin Gastroenterol Hepatol. 2022;20:2567-2576.e6.
Boursier J, de Ledinghen V, Leroy V, et al. A stepwise algorithm using an at-a-glance first-line test for the non-invasive diagnosis of advanced liver fibrosis and cirrhosis. J Hepatol. 2017;66:1158–65.
Chang XJ, Sun C, Chen Y, et al. On-treatment monitoring of liver fibrosis with serum hepatitis B core-related antigen in chronic hepatitis B. World J Gastroenterol. 2019;25:4764–78.
Vachon A, Osiowy C. Novel biomarkers of hepatitis B virus and their use in chronic hepatitis B patient management. Viruses. 2021;13:951.
Wang W, Shu Y, Bao H, et al. Genotypes and hot spot mutations of hepatitis B virus in northwest Chinese population and its correlation with diseases progression. Biomed Res Int. 2019;2019:3890962.
Chu CM, Lin CC, Chen YC, et al. Basal core promoter mutation is associated with progression to cirrhosis rather than hepatocellular carcinoma in chronic hepatitis B virus infection. Br J Cancer. 2012;107:2010–5.
Tseng TC, Liu CJ, Yang HC, et al. Serum hepatitis B surface antigen levels help predict disease progression in patients with low hepatitis B virus loads. Hepatology. 2013;57:441–50.
Croagh CM, Bell SJ, Slavin J, et al. Increasing hepatitis B viral load is associated with risk of significant liver fibrosis in HBeAg-negative but not HBeAg-positive chronic hepatitis B. Liver Int. 2010;30:1115–22.
Ji D, Chen Y, Shang Q, et al. Unreliable estimation of fibrosis regression during treatment by liver stiffness measurement in patients with chronic hepatitis B. Am J Gastroenterol. 2021;116:1676–85.
Tseng TC, Liu CJ, Hsu CY, et al. High level of hepatitis B core-related antigen associated with increased risk of hepatocellular carcinoma in patients with chronic HBV infection of intermediate viral load. Gastroenterology. 2019;157:1518-29.e3.
Liang LY, Wong VW, Toyoda H, et al. Serum hepatitis B core-related antigen predicts hepatocellular carcinoma in hepatitis B e antigen-negative patients. J Gastroenterol. 2020;55:899–908.
Hosaka T, Suzuki F, Kobayashi M, et al. Impact of hepatitis B core-related antigen on the incidence of hepatocellular carcinoma in patients treated with nucleos(t)ide analogues. Aliment Pharmacol Ther. 2019;49:457–71.
Mak LY, Huang Q, Wong DK, et al. Residual HBV DNA and pgRNA viraemia is associated with hepatocellular carcinoma in chronic hepatitis B patients on antiviral therapy. J Gastroenterol. 2021;56:479–88.
Acknowledgements
We appreciate the Major Science and Technology Special Project of China (2018ZX10725-506) and Beijing Natural Science Foundation (7212101) for funding support. We are very grateful to professor Jingfeng Bi, a statistician, for the statistical guidance.
Author information
Authors and Affiliations
Contributions
All authors approved the final version of the manuscript. YY and XC had full access to all data in the study and assume responsibility for the integrity of the data and accuracy of the data analysis. YY conceptualized and designed the study. XL and CS helped with the acquisition, analysis, and interpretation of data. XC, XL, and WD drafted the manuscript. All authors critically revised the manuscript for important intellectual content. XC helped with statistical analysis. YY obtained funding. YL, QS, LS, QL, QL, HL, JW, ZY, JL, GX, LL, LC, LT, and YC provided administrative, technical, and material support. YY supervised the study.
Corresponding authors
Ethics declarations
Conflict of interest
The authors who have taken part in this study declared that they do not have anything to disclose regarding funding or conflict of interest concerning this manuscript.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Chang, X., Li, Y., Sun, C. et al. High-risk population of progressive hepatic fibrosis in chronic hepatitis B patients on antiviral therapy. J Gastroenterol 58, 481–493 (2023). https://doi.org/10.1007/s00535-023-01970-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00535-023-01970-3