Abstract
Background
Excessive type I IFN (IFN-I) production by plasmacytoid dendritic cells (pDCs) promotes autoimmunity. Recently, we reported that a prominent feature of both experimental autoimmune pancreatitis (AIP) and human type 1 AIP is pDC activation followed by enhanced production of IFN-I and IL-33. However, the roles played by interferon regulatory factor 7 (IRF7), a critical transcription factor for IFN-I production in pDCs, in these disorders have not been clarified.
Methods
Whole and nuclear extracts were isolated from pancreatic mononuclear cells (PMNCs) from MRL/MpJ mice exhibiting AIP. Expression of phospho-IRF7 and nuclear translocation of IRF7 was examined in these extracts by immunoblotting. Pancreatic expression of IRF7 was assessed by immunofluorescence analysis in experimental AIP. Nuclear translocation of IRF7 upon exposure to neutrophil extracellular traps (NETs) was assessed in peripheral blood pDCs from type 1 AIP patients. Pancreatic IRF7 expression was examined in surgically operated specimens from type 1 AIP patients.
Results
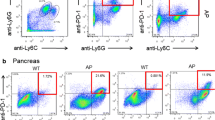
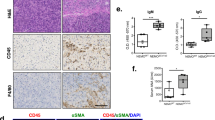
IRF7 activation was induced in pancreatic pDCs in experimental AIP. siRNA-mediated knockdown of IRF7 expression prevented AIP development, which was accompanied by a marked reduction in both pancreatic accumulation of pDCs and production of IFN-α and IL-33. Notably, in peripheral blood pDCs isolated from patients with type 1 AIP, nuclear translocation of IRF7 was enhanced as compared with the translocation in pDCs from healthy controls. Furthermore, IRF7-expressing pDCs were detected in the pancreas of patients with type 1 AIP.
Conclusions
These findings suggest that the IRF7-IFN-I-IL-33 axis activated in pDCs drives pathogenic innate immune responses associated with type 1 AIP.





Similar content being viewed by others
Abbreviations
- Ab:
-
Antibody
- Ag:
-
Antigen
- AIP:
-
Autoimmune pancreatitis
- BDCA2:
-
Blood dendritic cell antigen 2
- CP:
-
Chronic pancreatitis
- ELISA:
-
Enzyme-linked immunosorbent assay
- H&E:
-
Hematoxylin and eosin
- IFN-I:
-
Type I IFN
- IgG4-RD:
-
IgG4-related disease
- IP:
-
Intraperitoneal
- IRF:
-
Interferon regulatory factor
- IV:
-
Intravenous
- MSU:
-
Monosodium urate
- NET:
-
Neutrophil extracellular trap
- PC:
-
Pancreatic cancer
- pDC:
-
Plasmacytoid dendritic cell
- PDCA:
-
pDC antigen
- pIRF3:
-
Phospho-IRF3
- pIRF7:
-
Phospho-IRF7
- PMNC:
-
Pancreatic mononuclear cell
- poly(I:C):
-
Polyinosinic–polycytidylic acid
- SLE:
-
Systemic lupus erythematosus
- TF:
-
Transcription factor
- TLR:
-
Toll-like receptor
References
Swiecki M, Colonna M. The multifaceted biology of plasmacytoid dendritic cells. Nat Rev Immunol. 2015;15:471–85.
Gilliet M, Cao W, Liu YJ. Plasmacytoid dendritic cells: sensing nucleic acids in viral infection and autoimmune diseases. Nat Rev Immunol. 2008;8:594–606.
Honda K, Takaoka A, Taniguchi T. Type I interferon [corrected] gene induction by the interferon regulatory factor family of transcription factors. Immunity. 2006;25:349–60.
Wang Y, Swiecki M, McCartney SA, et al. dsRNA sensors and plasmacytoid dendritic cells in host defense and autoimmunity. Immunol Rev. 2011;243:74–90.
Ganguly D. Do type I interferons link systemic autoimmunities and metabolic syndrome in a pathogenetic continuum? Trends Immunol. 2018;39:28–43.
Watanabe T, Minaga K, Kamata K, et al. Mechanistic insights into autoimmune pancreatitis and IgG4-related disease. Trends Immunol. 2018;39:874–89.
Garcia-Romo GS, Caielli S, Vega B, et al. Netting neutrophils are major inducers of type I IFN production in pediatric systemic lupus erythematosus. Sci Transl Med. 2011;3:73ra20.
Lande R, Ganguly D, Facchinetti V, et al. Neutrophils activate plasmacytoid dendritic cells by releasing self-DNA-peptide complexes in systemic lupus erythematosus. Sci Transl Med. 2011;3:73ra19.
Furie R, Khamashta M, Merrill JT, et al. Anifrolumab, an anti-interferon-alpha receptor monoclonal antibody, in moderate-to-severe systemic lupus erythematosus. Arthritis Rheumatol. 2017;69:376–86.
Khamashta M, Merrill JT, Werth VP, et al. Sifalimumab, an anti-interferon-alpha monoclonal antibody, in moderate to severe systemic lupus erythematosus: a randomised, double-blind, placebo-controlled study. Ann Rheum Dis. 2016;75:1909–16.
Stone JH, Zen Y, Deshpande V. IgG4-related disease. N Engl J Med. 2012;366:539–51.
Kamisawa T, Zen Y, Pillai S, et al. IgG4-related disease. Lancet. 2015;385:1460–71.
Shimosegawa T, Chari ST, Frulloni L, et al. International consensus diagnostic criteria for autoimmune pancreatitis: guidelines of the International Association of Pancreatology. Pancreas. 2011;40:352–8.
Okazaki K, Uchida K, Koyabu M, et al. Recent advances in the concept and diagnosis of autoimmune pancreatitis and IgG4-related disease. J Gastroenterol. 2011;46:277–88.
Kawa S, Okazaki K, Kamisawa T, Working members of Research Committee for Intractable Pancreatic D, et al. Japanese consensus guidelines for management of autoimmune pancreatitis: II. Extrapancreatic lesions, differential diagnosis. J Gastroenterol. 2010;45:355–69.
Arai Y, Yamashita K, Kuriyama K, et al. Plasmacytoid dendritic cell activation and IFN-alpha production are prominent features of murine autoimmune pancreatitis and human IgG4-related autoimmune pancreatitis. J Immunol. 2015;195:3033–44.
Watanabe T, Yamashita K, Arai Y, et al. Chronic fibro-inflammatory responses in autoimmune pancreatitis depend on IFN-alpha and IL-33 produced by plasmacytoid dendritic cells. J Immunol. 2017;198:3886–96.
Kamata K, Watanabe T, Minaga K, et al. Autoimmune pancreatitis mouse model. Curr Protoc Immunol. 2018;120:15 31 1–15 31 8.
Puthia M, Ambite I, Cafaro C, et al. IRF7 inhibition prevents destructive innate immunity-A target for nonantibiotic therapy of bacterial infections. Sci Transl Med. 2016;8:336ra59.
Watanabe T, Asano N, Fichtner-Feigl S, et al. NOD1 contributes to mouse host defense against Helicobacter pylori via induction of type I IFN and activation of the ISGF3 signaling pathway. J Clin Invest. 2010;120:1645–62.
Tsuji Y, Watanabe T, Kudo M, et al. Sensing of commensal organisms by the intracellular sensor NOD1 mediates experimental pancreatitis. Immunity. 2012;37:326–38.
Watanabe T, Sadakane Y, Yagama N, et al. Nucleotide-binding oligomerization domain 1 acts in concert with the cholecystokinin receptor agonist, cerulein, to induce IL-33-dependent chronic pancreatitis. Mucosal Immunol. 2016;9:1234–49.
The Japan Pancreas Society. Japanese clinical diagnostic criteria for autoimmune pancreatitis, 2018 (Proposal) revision of japanese clinical diagnostic criteria for autoimmune pancreatitis, 2011. Suizo. 2018;33:902–13.
Sun L, Zhu Z, Cheng N, et al. Serum amyloid A induces interleukin-33 expression through an IRF7-dependent pathway. Eur J Immunol. 2014;44:2153–64.
Kamata K, Watanabe T, Minaga K, et al. Intestinal dysbiosis mediates experimental autoimmune pancreatitis via activation of plasmacytoid dendritic cells. Int Immunol. 2019;31:795–809.
Kaplan MJ, Radic M. Neutrophil extracellular traps: double-edged swords of innate immunity. J Immunol. 2012;189:2689–95.
Shiokawa M, Kodama Y, Sekiguchi K, et al. Laminin 511 is a target antigen in autoimmune pancreatitis. Sci Transl Med. 2018;10:eaaq0997.
Hubers LM, Vos H, Schuurman AR, et al. Annexin A11 is targeted by IgG4 and IgG1 autoantibodies in IgG4-related disease. Gut. 2018;67:728–35.
Perugino CA, AlSalem SB, Mattoo H, et al. Identification of galectin-3 as an autoantigen in patients with IgG4-related disease. J Allergy Clin Immunol. 2019;143(736–45):e6.
Furukawa S, Moriyama M, Miyake K, et al. Interleukin-33 produced by M2 macrophages and other immune cells contributes to Th2 immune reaction of IgG4-related disease. Sci Rep. 2017;7:42413.
Awada A, Nicaise C, Ena S, et al. Potential involvement of the IL-33-ST2 axis in the pathogenesis of primary Sjogren's syndrome. Ann Rheum Dis. 2014;73:1259–63.
Minaga K, Watanabe T, Kamata K, et al. The IFN-alpha-IL-33 Axis as Possible Biomarkers in IgG4-Related Disease. Am J Gastroenterol. 2019;114:1002–3.
Acknowledgements
This work was supported by Grants-in-Aid for Scientific Research (19K08455, 19K17506) from Japan Society for the Promotion of Science, Takeda Science Foundation, Smoking Research Foundation, Yakult Bio-Science Foundation, SENSHIN Medical Research Foundation, and Japan Agency for Medical Research and Development (AMED) for Research on Intractable Diseases.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declared that they have no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Minaga, K., Watanabe, T., Arai, Y. et al. Activation of interferon regulatory factor 7 in plasmacytoid dendritic cells promotes experimental autoimmune pancreatitis. J Gastroenterol 55, 565–576 (2020). https://doi.org/10.1007/s00535-020-01662-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00535-020-01662-2