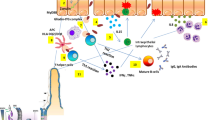
Abstract
Celiac disease has advanced from a medical rarity to a highly prevalent disorder. Patients with the disease show varying degrees of chronic inflammation within the small intestine due to an aberrant immune response to the digestion of gliadin found in wheat. As a result, cytokines and antibodies are produced in celiac patients that can be used as specific biomarkers for developing diagnostic tests. This review paper describes celiac disease in terms of its etiological cause, pathological effects, current diagnostic tests based on mucosal biopsy, and the genetic basis for the disease. In addition, it discusses the use of gliadin-induced cytokines, antibodies and autoantibodies as a diagnostic tool for celiac disease. Despite good initial results in terms of sensitivity and specificity, when these immunological tests were used on a large scale, even in combination with genetic testing, the results showed lower predictive value. This review addresses that issue and ends with an outlook on future work required to develop diagnostic tests with greater accuracy in predicting celiac disease in the general public, thus avoiding the need for endoscopy and mucosal biopsy.

Similar content being viewed by others
Abbreviations
- ELISA:
-
Enzyme-linked immunosorbent assay
- NK:
-
Natural killer
- HLA:
-
Human leukocyte antigen
- TCR:
-
T-cell receptor
- LMW:
-
Low molecular weight
- CDR:
-
Complementarity-determining region
- tTG:
-
Tissue transglutaminase
- AGA:
-
Anti-gliadin antibodies
- Anti-EMA:
-
Anti-endomysial antibodies
- Anti-tTG:
-
Anti-transglutaminase antibodies
- Anti-DGP:
-
Anti-deamidated gliadin peptide
- IFA:
-
Indirect immunofluorescence
- RIA:
-
Radioimmunoassay
- GFD:
-
Gluten-free diet
- SPR:
-
Surface plasmon resonance
- DIG:
-
Diffusion in gel
- IBD:
-
Inflammatory bowel disease
- GIP:
-
Gluten immunogenic peptides
References
Ludvigsson JF, Leffler DA, Bai JC, et al. The Oslo definitions for celiac disease and related terms. Gut. 2013;62:43–52.
Kang JY, Kang AHY, Green A, et al. Systematic review: worldwide variation in the frequency of celiac disease and changes over time. Aliment Pharmacol Ther. 2013;38:226–45.
Lionetti E, Gatti S, Pulvirenti A, et al. Celiac disease from a global perspective. Best Pract Res Clin Gastroenterol. 2015;29:365–79.
Hovell CJ, Collett JA, Vautier G, et al. High prevalence of celiac disease in a population-based study from Western Australia: a case for screening? Med J Aust. 2001;175:247–50.
Rubio-Tapia A, Ludvigsson JF, Brantner TL, et al. The prevalence of celiac disease in the United States. Am J Gastroenetrol. 2012;107:1538–44.
Woychik JH, Boundy JA, Dimler RJ. Starch gel-electrophoresis of wheat gluten proteins with concentrated urea. Arch Biochem Biophys. 1961;94:477–82.
Bushuk W, Zillman RR. Wheat cultivar identification by gliadin electrophoregrams: 1. Apparatus, method, and nomenclature. Can J Plant Sci. 1978;58:505–15.
Metakovsky EV, Branlard G, Chernakov VM, et al. Recombinant mapping of some chromosome 1A-, 1B-, 1D-, and 6B-controlled gliadins and low molecular weight glutenin subunits in common wheat. Theor Appl Genet. 1997;94:788–95.
Anderson OD, Dong L, Huo N, et al. A new class of wheat gliadin genes and proteins. PLoS ONE. 2012;7:12.
Kasarda DD, Okita TW, Bernardin JE, et al. Nucleic acid (cDNA) and amino acid sequences of α-type gliadins from wheat (Triticum aestivum). Proc Natl Acad Sci. 1984;81:4712–6.
Anderson OD, Greene FC. The α-gliadin gene family. DNA and protein sequence variation, subfamily structure, and origins of pseudogenes. Theor Appl Genet. 1997;95:59–65.
Noma S, Kawaura K, Hayakawa K, et al. Comprehensive molecular characterization of the α/β gliadin multigene family in hexaploid wheat. Mol Genet Genom. 2016;291:65–77.
Bronstein HD, Haeffner LJ, Kowlessar OD. Enzymatic digestion of gliadin: the effect of the resultant peptides in adult celiac disease. Clin Chim Acta. 1966;14:141–55.
Weiser H. Relation between gliadin structure and celiac toxicity. Acta Paediatr. 1996;412:3–9.
Shan L, Molberg O, Parrot I, et al. Structural basis for gluten intolerance in celiac disease. Science. 2002;297:2275–9.
Qi PF, Chen Q, Oullet T, et al. The molecular diversity of α-gliadin genes in the tribe Triticea. Genetica. 2013;141:303–10.
Oberhuber G, Granditsch G, Vogelsang H. The histopathology of celiac disease: time for a standardised report scheme for pathologists. Eur J Gastroentrol. 1999;11:1185–94.
Marsh MN, Vincenzo V, Srivastava A. Histology of gluten related disorders. Gastroenterol Hepatol Bed Bench. 2015;8:171–7.
Tonutti E, Bizzaro N. Diagnosis and classification of celiac disease and gluten sensitivity. Autoimmun Rev. 2014. doi:10.1016/j.autrev.2014.01.043.
Bao F, Green PH, Bhagat G. An update on celiac disease histopathology and the road ahead. Arch Pathol Lab Med. 2012;136:735–45.
Castillo NE, Theethira TG, Leffler DA. The present and the future in diagnosis and management of celiac disease. Gastroenterol Rep. 2015;3(1):3–11.
Hardy MY, Tye-Din JA. Celiac disease: a unique model for investigating broken tolerance in autoimmunity. Clin Transl Immunol. 2016. doi:10.1038/cti.2016.58.
Greco L, Romino R, Coto I, et al. The first large population-based twin study of celiac disease. Gut. 2002;50:624–8.
Howell MD, Austin RK, Kelleher D, et al. An HLA-D region restriction length polymorphism associated with celiac disease. J Exp Med. 1986;169:345–50.
Olivares M, Neef A, Castillejo G, et al. The HLA-DQ2 genotype selects for early intestinal microbiota composition in infants at high risk of developing celiac disease. Gut. 2015;64:406–17.
Lundin KEA, Scott H, Hansen T, et al. Gliadin-specific, HLA-DQ (alpha1*0501, beta1*0201) restricted T cells isolated from the small intestinal mucosa of celiac disease patients. J Exp Med. 1993;178:187–96.
Costantini S, Rossi M, Colonna G, et al. Modelling of HLA-DQ2 and its interaction with gluten peptides to explain molecular recognition in celiac disease. J Mol Graph Model. 2005;23:419–31.
Henderson KN, Tye-Din JA, Reid H, et al. A structural and immunological basis for the role of human leukocyte antigen DQ8 in celiac disease. Immunity. 2007;27:23–34.
Broughton SE, Petersen J, Theodossis A, et al. Biased T cell receptor usage directed against human leukocyte antigen DQ8-restricted gliadin peptides is associated with celiac disease. Immunity. 2012;37:611–21.
Hadithi M, von Blomberg BM, Crusius JB, et al. Accuracy of serologic tests and HLA-DQ typing for diagnosing celiac disease. Ann Intern Med. 2007;147:294–302.
Husby S, Koletzko S, Korponay-Szabó IR, et al. European Society for Pediatric Gastroenterology, Hepatology, and Nutrition guidelines for the diagnosis of celiac disease. J Pediatr Gastroenterol Nutr. 2012;54:136–60.
Fukunaga M, Ishimura N, Fukuyama C, et al. Celiac disease in non-clinical populations of Japan. J Gastroenterol. 2017. doi:10.1007/s00535-017-1339-9.
Dubois PC, Trynka G, Franke L, et al. Multiple common variants for celiac disease influencing immune gene expression. Nat Genet. 2010;42(4):295–302.
Abraham G, Rohmer A, Tye-Din JA, et al. Genomic prediction of celiac disease targeting HLA-positive individuals. Genome Med. 2015;7:3–11.
Kruppa K. Endomysial antibodies predict celiac disease irrespective of the titers or clinical presentation. World J Gastroenterol. 2012;18:2511–6.
Fasano A. Zonulin and its regulation of intestinal barrier function: the biological door to inflammation, autoimmunity, and cancer. Physiol Rev. 2011;91:151–75.
Lammers KM, Lu R, Brownley J, et al. Gliadin induces an increase in intestinal permeability and zonulin release by binding to the chemokine receptor CXCR3. Gastroenterology. 2008;135:194–204.
Schumann M, Richter JF, Wedell I, et al. Mechanisms of epithelial translocation of the alpha(2)-gliadin-33 mer in celiac sprue. Gut. 2008;57:747–54.
Matysiak-Budnik T, Moura IC, Arcos-Fajardo M, et al. Secretory IgA mediates retrotranscytosis of intact gliadin peptides via the transferrin receptor in celiac disease. J Exp Med. 2008;205:143–54.
Bodd M, Ráki M, Tollefsen S, et al. HLA-DQ2-restricted gluten-reactive T cells produce IL-21 but not IL-17 or IL-22. Mucosal Immunol. 2010;3:594–601.
Peluso I, Fantini MC, Fina D, et al. IL-21 counteracts the regulatory T cell-mediated suppression of human CD4+ T lymphocytes. J Immunol. 2007;178:732–9.
Hüe S, Mention J-J, Monteiro RC, et al. A direct role for NKG2D/MICA interaction in villous atrophy during celiac disease. Immunity. 2004;21:367–77.
Meresse B, Curran SA, Ciszewski C, et al. Reprogramming of CTLs into natural killer-like cells in celiac disease. J Exp Med. 2006;203:1343–55.
Sollid LM, Jabri B. Triggers and drivers of autoimmunity: lessons from celiac disease. Nat Rev Immunol. 2013;13:294–302.
Sabatino AD, Vanoli A, Giuffrida P, et al. The function of tissue transglutaminase in celiac disease. Autoimmun Rev. 2012;11:746–53.
Evans DF, Pye G, Bramley R, et al. Measurement of gastrointestinal pH profiles in normal ambulant human subjects. Gut. 1988;29:1035–41.
Sollid LM. Molecular basis of celiac disease. Annu Rev Immunol. 2000;18:53–81.
Du Pre’ MF, Sollid LM. T-cell and B-cell immunity in celiac disease. Best Pract Res Clin Gastroenterol. 2015;29:413–23.
Dieterich W, Laag E, Schopper H, et al. Autoantibodies to tissue transglutaminase as predictors of celiac disease. Gastroenterology. 1998;115:1317–21.
O’Farrelly C, Kelly J, Hekkens W, et al. Alpha gliadin antibody levels: a serological test for celiac disease. Br Med J (Clin Res Ed). 1983;25:2007–10.
Savilahti E, Viander M, Perkkio M, et al. IgA antigliadin antibodies: a marker of mucosal damage in childhood celiac disease. Lancet. 1983;1:320–2.
Tucker NT, Barghuthy FS, Prihoda TJ, et al. Antigliadin antibodies detected by enzyme-linked immunosorbent assay as a marker of childhood celiac disease. J Pediatr. 1988;113:286–9.
Bodé S, Weile B, Krasilnikoff PA, et al. The diagnostic value of the gliadin antibody test in celiac disease in children: a prospective study. J Pediatr Gastroenterol Nutr. 1993;17:260–4.
Lerner A, Kumar V, Iancu TC. Immunological diagnosis of childhood celiac disease: comparison between antigliadin, antireticulin and antiendomysial antibodies. Clin Exp Immunol. 1994;95:78–82.
Chartrand LJ, Aglunik J, Vanounou T, et al. Effectiveness of anti-Gliadin antibodies as a screening test for celiac disease in children. Can Med Assoc J. 1997;1997(157):527–33.
Lagerqvist C, Ingrid D, Tony H, et al. Antigliadin immunoglobulin A best in finding celiac disease in children younger than 18 months of age. J Pediatr Gastroenterol Nutr. 2008;47:428–35.
Pereira SV, Raba J, Messina GA. IgG anti-Gliadin determination with an immunological microfluidic system applied to the automated diagnostic of the celiac disease. Anal Bioanal Chem. 2010;396:2921–7.
Ladinser B, Rossipal E, Pittschieler K. Endomysium antibodies in celiac disease: an improved method. Gut. 1994;35:776–8.
Picarelli A. 31-43 amino acid sequence of the a-Gliadin induces anti-Endomysial antibody production during in vitro challenge. Scand J Gastroenterol. 1999;34:1099–102.
Rostami K, Jo Kerckhaert, Tiemessen R, et al. Sensitivity of antiendomysium and antigliadin antibodies in untreated celiac disease: disappointing in clinical practice: sensitivity of IgA EMA and AGA in celiac disease. Am J Gastroenterol. 1994;94:888–94.
Leffler DA, Schuppan D. Update on serologic testing in celiac disease. Am J Gastroenterol. 2010;105:2520–4.
Bruce SE, Bjarnason I, Peter I. Human jejunal transglutaminase: demonstration of activity, enzyme kinetics and substrate specificity with special relation to gliadin and celiac disease. Clin Sci. 1985;68:573–9.
Hill PG, Holmes GK. Celiac disease: a biopsy is not always necessary for diagnosis. Aliment Pharmacol Ther. 2008;27:572–7.
Vivas S, Ruiz de Morales JG, Riestra S, et al. Duodenal biopsy may be avoided when high transglutaminase antibody titres are present. World J Gastroenterol. 2009;15:4775–80.
Tortara R, Imperatore N, Capone P, et al. The presence of anti-Endomysial antibodies and the level of anti-tissue transglutaminase can be used to diagnose adult celiac disease without duodenal biopsy. Aliment Pharmacol Ther. 2014;40:122–9.
Dahlbom I, Nyberg BI, Bernston L, et al. Simultaneous detection of IgA and IgG antibodies against tissue transglutaminase: the preferred pre-biopsy test in childhood celiac disease. Scand J Clin Lab Invest. 2016;76:208–16.
Sblattero D, Berti I, Trevisiol C, et al. Human recombinant tissue transglutaminase ELISA: an innovative diagnostic assay for celiac disease. Am J Gastroenterol. 2000;95:1253–7.
Lock RJ, Gilmour JEM, Unsworth DJ. Anti-tissue transglutaminase, anti-endomysium and anti-R1-reticulin autoantibodies: the antibody trinity of celiac disease. Clin Exp Immunol. 1999;116:258–62.
Lewis NR, Scott BB. Meta-analysis: deamidated gliadin peptide antibody and tissue transglutaminase antibody compared as screening tests for celiac disease. Aliment Pharmacol Ther. 2010;31:73–81.
Dahlbom I, Korponay-Szabo IR, Kovacs JB, et al. Prediction of clinical and mucosal severity of celiac disease and dermatitis herpetiformis by quantification of IgA/IgG serum antibodies to tissue transglutaminase. J Pediatr Gastroenterol Nutr. 2010;50:140–6.
Volta U, Granito A, Fiorini E, et al. Usefulness of antibodies to deamidated gliadin peptides in celiac disease diagnosis and follow-up. Dig Dis Sci. 2008;53:1582–8.
Dahle C, Hagman A, Ignatova S, et al. Antibodies against deamidated gliadin peptides identify adult celiac disease patients negative for antibodies against endomysium and tissue transglutaminase. Aliment Pharmacol Ther. 2010;32:254–60.
Barbato M, Maiellaa G, Camilloa CD, et al. The anti-deamidated gliadin peptide antibodies unmask celiac disease in small children with chronic diarrhoea. Dig Liver Dis. 2011;43:465–9.
Amarri S, Alvisi P, De Giorgio R, et al. Antibodies to deamidated gliadin peptides: an accurate predictor of celiac disease in infancy. J Clin Immunol. 2013;33:1027–30.
Schwertz E, Kahlberg F, Sack U, et al. Serologic assay based on gliadin-related nonapeptides as a highly sensitive and specific diagnostic aid in celiac disease. Clin Chem. 2004;50:2370–5.
Agardh A. Antibodies against synthetic deamidated gliadin peptides and tissue transglutaminase for the identification of childhood celiac disease. Clin Gastroenterol Hepatol. 2007;5:1276–81.
Ankelo M, Kleimola V, Simell S, et al. Antibody responses to deamidated gliadin peptide show high specificity and parallel antibodies to tissue transglutaminase in developing celiac disease. Clin Exp Immunol. 2007;150:285–93.
Rashtak S, Ettore MW, Homburger HA, et al. Comparative usefulness of deamidated gliadin antibodies in the diagnosis of celiac disease. Clin Gastroenetrol Hepatol. 2008;6:426–32.
Basso D, Guariso G, Fogar P, et al. Antibodies against synthetic deamidated gliadin peptides for celiac disease diagnosis and follow-up in children. Clin Chem. 2008;55:150–7.
Sugai E, Moreno ML, Hwang HJ, et al. Celiac disease serology in patients with different pretest probabilities: is biopsy avoidable? World J Gastroenterol. 2010;16:3144–52.
Anderson RP, van Heel DA, Tye-Din JA, et al. T cells in peripheral blood after gluten challenge in celiac disease. Gut. 2005;54:1217–23.
Brottveit M, Ráki M, Bergseng E, et al. Assessing possible celiac disease by an HLA-DQ2-gliadin tetramer test. Am J Gastroenterol. 2011;106:1318–24.
Goletti D, Vincenti D, Carrara S, et al. Selected RD1 peptides for active tuberculosis diagnosis: comparison of a gamma interferon whole-blood enzyme-linked immunosorbent assay and an enzyme-linked immunospot assay. Clin Diagn Lab Immunol. 2005;12:1311–6.
Ruhwald M, Bjerregaard-Anderson M, Rabna P, et al. CXCL10/IP-10 release is induced by incubation of whole blood from tuberculosis patients with ESAT-6, CFP10 and TB7.7. Microbes Infect. 2007;9:806–12.
Lalvani A, Meroni PL, Millington KA, et al. Recent advances in diagnostic technology: applications in autoimmune and infectious diseases. Clin Exp Rheumatol. 2008;26:S62–6.
Anderson RP, Degano P, Godkin AJ, et al. In vivo antigen challenge in celiac disease identifies a single transglutaminase-modified peptide as the dominant A-gliadin T cell epitope. Nat Med. 2000;6:337–42.
Tye-Din JA, Stewart JA, Dromey JA, et al. Comprehensive, quantitative mapping of T cell epitopes in gluten in celiac disease. Sci Transl Med. 2010;2:41.
Ontiveros N, Tye-Din JA, Hardy MY, et al. Ex-vivo whole blood secretion of interferon (IFN)-γ and IFN-γ-inducible protein-10 measured by enzyme-linked immunosorbent assay are as sensitive as IFN-γ enzyme-linked immunospot for the detection of gluten-reactive T cells in human leucocyte antigen (HLA)-DQ2.5+-associated celiac disease. Clin Exp Immunol. 2014;175:305–15.
Lehmann PV, Zhang W. Unique strengths of ELISPOT for T cell diagnostics. Methods Mol Biol. 2012;792:3–23.
Salazer C, Nagadia R, Pandit P, et al. A novel saliva-based micro-RNA biomarker panel to detect head and neck cancers. Cell Oncol. 2014;37:331–8.
Baldini C, Giusti L, Ciregia F, et al. Proteomic analysis of saliva: a unique tool to distinguish primary Sjogren’s syndrome from secondary Sjogren’s syndrome and other sicca syndromes. Arthritis Res Ther. 2011;13(6):R194.
Jacobs R, Maasdorp E, Malherbe S, et al. Diagnostic potential of novel salivary host biomarkers as candidates for the immunological diagnosis of tuberculosis disease and monitoring of tuberculosis treatment response. PLoS ONE. 2016;12:1311–6.
Sueki A, Matsuda K, Yamaguchi A, et al. Evaluation of saliva as a diagnostic materials for influenza virus infection by PCR-based assay. Clin Chim Acta. 2016;453:71–4.
Hakeem V, Fifield R, Al-Bayaty HF, et al. Salivary IgA antigliadin antibody as a marker for celiac disease. Arch Dis Child. 1992;67:724–7.
Lenander-Lumikari Ihalin R, Lahteenoja H. Changes in whole saliva in patients with celiac disease. Arch Oral Biol. 2000;45(5):347–54.
Bonamico M, Nenna R, Montuori M, et al. First salivary screening of celiac disease by detection of anti-transglutaminase autoantibody radioimmunoassay in 5000 Italian primary schoolchildren. J Pediatr Gastroenterol Nutr. 2011;52(1):17–20.
Adornetto G, Fabiani L, Volpe G, et al. An electrochemical immunoassay for the screening of celiac disease in saliva samples. Anal Bioanal Chem. 2015;407:7189–96.
Actis AB, Perovic NR, Defagó D, et al. Fatty acid profile of human saliva: a possible indicator of dietary fat intake. Arch Oral Biol. 2005;50:1–6.
Benkebil F, Combescure C, Anghel SI, et al. Diagnostic accuracy of a new point-of-care screening assay for celiac disease. World J Gastroenterol. 2013;19:5111–7.
Bienvenu F, Duvanel CB, Seignovert C, et al. Evaluation of a point-of-care test based on deamidated gliadin peptides for celiac disease screening in a large pediatric population. Eur J Gastroenterol Hepatol. 2012. doi:10.1097/meg.0b013e3283582d95.
Watanabe C, Komoto S, Hokari R, et al. Prevalence of serum celiac antibody in patients with IBD in Japan. J Gastroenterol. 2014;49:825–34.
Wungjiranirun M, Kelly CP, Leffler DA. Current status of celiac disease drug development. Am J Gastroenterol. 2016;111:779–86.
Soler M, Estevez MC, Moreno Mde L, et al. Label-free SPR detection of gluten peptides in urine for non-invasive celiac disease follow-up. Biosens Bioelectron. 2016;79:158–64.
Comino I, Fernández-Bañares F, Esteve M, et al. Fecal gluten peptides reveal limitations of serological tests and food questionnaires for monitoring gluten-free diet in celiac disease patients. Am J Gastroenterol. 2016;111:1456–65.
Rohrman BA, Richards-Kortum RR. A paper and plastic device for performing recombinase polymerase amplification of HIV DNA. Lab Chip. 2012;12:3082–8.
Fu E, Liang T, Spicar-Mihalic P, et al. Two-dimensional paper network format that enables simple multistep assays for use in low-resource settings in the context of malaria antigen detection. Anal Chem. 2012;84:4574–9.
Veigas B, Jacob JM, Costa MN, et al. Gold on paper–paper platform for Au-nanoprobe TB detection. Lab Chip. 2012;12:4802–8.
Acknowledgements
The authors thank Dr. Jason Tye-Din, MBBS, PhD, FRACP, Immunology Division, The Walter and Eliza Hall Institute of Medical Research, Parkville, Victoria, Australia, for his critical review and feedback on the manuscript. This research is supported by an Australian Government Research Training Program Scholarship. O.S. acknowledges the Australian Research Council and National Health and Medical Research Council for financial support (APP1101258, IH150100028).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Kaur, A., Shimoni, O. & Wallach, M. Celiac disease: from etiological factors to evolving diagnostic approaches. J Gastroenterol 52, 1001–1012 (2017). https://doi.org/10.1007/s00535-017-1357-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00535-017-1357-7