Abstract
Background
The sensitivity of 2-[18F]fluoro-2-deoxyglucose positron emission tomography (FDG-PET) for advanced colorectal neoplasms among healthy subjects is not yet fully understood. The present study aimed to clarify the sensitivity by analyzing large-scale data from an asymptomatic screening population.
Methods
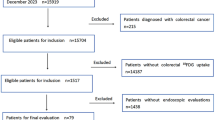
A total of 7505 asymptomatic screenees who underwent both FDG-PET and colonoscopy at our Cancer Screening Division between February 2004 and March 2013 were analyzed. FDG-PET and colonoscopy were performed on consecutive days, and each examination was interpreted in a blinded fashion. The results of the two examinations were compared for each of the divided six colonic segments, with those from colonoscopy being set as the reference. The relationships between the sensitivity of FDG-PET and clinicopathological features of advanced neoplasms were also evaluated.
Results
Two hundred ninety-one advanced neoplasms, including 24 invasive cancers, were detected in 262 individuals. Thirteen advanced neoplasms (advanced adenomas) were excluded from the analysis because of the coexistence of lesions in the same colonic segment. The sensitivity, specificity, and positive and negative predictive values of FDG-PET for advanced neoplasms were 16.9 % [95 % confidence interval (CI) 12.7–21.8 %], 99.3 % (95 % CI 99.2–99.4 %), 13.5 % (95 % CI 10.1–17.6 %), and 99.4 % (95 % CI 99.3–99.5 %), respectively. The sensitivity was lower for lesions with less advanced histological grade, of smaller size, and flat-type morphology, and for those located in the proximal part of the colon.
Conclusions
FDG-PET is believed to be difficult to use as a primary screening tool in population-based colorectal cancer screening because of its low sensitivity for advanced neoplasms. Even when it is used in opportunistic cancer screening, the limit of its sensitivity should be considered.


Similar content being viewed by others
References
International Agency for Research on Cancer: GLOBOCAN 2012: Estimated cancer incidence, mortality and prevalence worldwide in 2012. http://globocan.iarc.fr/Default.aspx. Accessed 25 Aug 2015.
Rex DK, Johnson DA, Anderson JC, et al. American College of Gastroenterology guidelines for colorectal cancer screening 2008. Am J Gastroenterol. 2009;104:739–50.
Cairns SR, Scholefield JH, Steele RJ, et al. Guidelines for colorectal cancer screening and surveillance in moderate and high risk groups (update from 2002). Gut. 2010;59:666–89.
Sung JJ, Ng SC, Chan FK, et al. An updated Asia Pacific consensus recommendations on colorectal cancer screening. Gut. 2015;64:121–32.
Kim DH, Pickhardt PJ, Taylor AJ, et al. CT colonography versus colonoscopy for the detection of advanced neoplasia. N Engl J Med. 2007;357:1403–12.
de Haan MC, van Gelder RE, Graser A, et al. Diagnostic value of CT-colonography as compared to colonoscopy in an asymptomatic screening population: a meta-analysis. Eur Radiol. 2011;21:1747–63.
Van Gossum A, Munoz-Navas M, Fernandez-Urien I, et al. Capsule endoscopy versus colonoscopy for the detection of polyps and cancer. N Engl J Med. 2009;361:264–70.
Spada C, Hassan C, Munoz-Navas M, et al. Second-generation colon capsule endoscopy compared with colonoscopy. Gastrointest Endosc. 2011;74:581–9.
Yasuda S, Ide M, Fujii H, et al. Application of positron emission tomography imaging to cancer screening. Br J Cancer. 2000;83:1607–11.
Terauchi T, Murano T, Daisaki H, et al. Evaluation of whole-body cancer screening using 18F-2-deoxy-2-fluoro-d-glucose positron emission tomography: a preliminary report. Ann Nucl Med. 2008;22:379–85.
Minamimoto R, Senda M, Jinnouchi S, et al. The current status of an FDG-PET cancer screening program in Japan, based on a 4-year (2006–2009) nationwide survey. Ann Nucl Med. 2013;27:46–57.
Rigo P, Paulus P, Kaschten BJ, et al. Oncological applications of positron emission tomography with fluorine-18 fluorodeoxyglucose. Eur J Nucl Med. 1996;23:1641–74.
Yasuda S, Fujii H, Nakahara T, et al. 18F-FDG PET detection of colonic adenomas. J Nucl Med. 2001;42:989–92.
Drenth JP, Nagengast FM, Oyen WJ. Evaluation of (pre-)malignant colonic abnormalities: endoscopic validation of FDG-PET findings. Eur J Nucl Med. 2001;28:1766–9.
Friedland S, Soetikno R, Carlisle M, et al. 18-Fluorodeoxyglucose positron emission tomography has limited sensitivity for colonic adenoma and early stage colon cancer. Gastrointest Endosc. 2005;61:395–400.
van Kouwen MC, Nagengast FM, Jansen JB, et al. 2-(18F)-fluoro-2-deoxy-d-glucose positron emission tomography detects clinical relevant adenomas of the colon: a prospective study. J Clin Oncol. 2005;23:3713–7.
Nakajo M, Jinnouchi S, Tashiro Y, et al. Effect of clinicopathologic factors on visibility of colorectal polyps with FDG PET. AJR Am J Roentgenol. 2009;192:754–60.
Weston BR, Iyer RB, Qiao W, et al. Ability of integrated positron emission and computed tomography to detect significant colonic pathology: the experience of a tertiary cancer center. Cancer. 2010;116:1454–61.
Ravizza D, Bartolomei M, Santoro L, et al. Positron emission tomography for the detection of colorectal adenomas. Dig Liver Dis. 2010;42:185–90.
Hirakawa T, Kato J, Okumura Y, et al. Detectability of colorectal neoplasia with fluorine-18-2-fluoro-2-deoxy-d-glucose positron emission tomography and computed tomography (FDG-PET/CT). J Gastroenterol. 2012;47:127–35.
Huang SW, Hsu CM, Jeng WJ, et al. A comparison of positron emission tomography and colonoscopy for the detection of advanced colorectal neoplasms in subjects undergoing a health check-up. PLoS One. 2013;8:e69111.
Minamimoto R, Senda M, Jinnouchi S, et al. Detection of colorectal cancer and adenomas by FDG-PET cancer screening program: results based on a nationwide Japanese survey. Ann Nucl Med. 2014;28:212–9.
Gollub MJ, Grewal RK, Panu N, et al. Diagnostic accuracy of 18F-FDG PET/CT for detection of advanced colorectal adenoma. Clin Radiol. 2014;69:611–8.
Hwang JP, Woo SK, Yoon SY, et al. The potential usefulness of 18F-FDG PET/CT for detecting colorectal carcinoma and adenoma in asymptomatic adults. Ann Nucl Med. 2015;29:157–63.
Shoda H, Kakugawa Y, Saito D, et al. Evaluation of 18F-2-deoxy-2-fluoro-glucose positron emission tomography for gastric cancer screening in asymptomatic individuals undergoing endoscopy. Br J Cancer. 2007;97:1493–8.
Hamashima C, Sobue T, Muramatsu Y, et al. Comparison of observed and expected numbers of detected cancers in the research center for cancer prevention and screening program. Jpn J Clin Oncol. 2006;36:301–8.
Hamiliton SR, Bosman FT, Boffetta P, et al. Carcinoma of the colon and rectum. In: Bosman FT, Carneiro F, Hruban RH, et al., editors. WHO classification of tumors of the digestive system. 4th ed. Lyon: IARC; 2010. p. 134–46.
Winawer SJ, Zauber AG. The advanced adenoma as the primary target of screening. Gastrointest Endosc Clin N Am. 2002;12:1–9.
Schlemper RJ, Itabashi M, Kato Y, et al. Differences in the diagnostic criteria used by Japanese and Western pathologists to diagnose colorectal carcinoma. Cancer. 1998;82:60–9.
Kudo S, Tamura S, Nakajima T, et al. Diagnosis of colorectal tumorous lesions by magnifying endoscopy. Gastrointest Endosc. 1996;44:8–14.
Fu KI, Sano Y, Kato S, et al. Chromoendoscopy using indigo carmine dye spraying with magnifying observation is the most reliable method for differential diagnosis between non-neoplastic and neoplastic colorectal lesions: a prospective study. Endoscopy. 2004;36:1089–93.
Katagiri A, Fu KI, Sano Y, et al. Narrow band imaging with magnifying colonoscopy as diagnostic tool for predicting histology of early colorectal neoplasia. Aliment Pharmacol Ther. 2008;27:1269–74.
Matsuda T, Fujii T, Saito Y, et al. Efficacy of the invasive/non-invasive pattern by magnifying chromoendoscopy to estimate the depth of invasion of early colorectal neoplasms. Am J Gastroenterol. 2008;103:2700–6.
Sano Y, Ikematsu H, Fu KI, et al. Meshed capillary vessels by use of narrow-band imaging for differential diagnosis of small colorectal polyps. Gastrointest Endosc. 2009;69:278–83.
Uraoka T, Saito Y, Ikematsu H, et al. Sano’s capillary pattern classification for narrow-band imaging of early colorectal lesions. Dig Endosc. 2011;23(Suppl 1):112–5.
The Paris endoscopic classification of superficial neoplastic lesions: esophagus, stomach, and colon: November 30 to December 1, 2002. Gastrointest Endosc 2003;58(Suppl):S3–43.
Colon and rectum. In: Edge SB, Byrd DR, Compton CC, et al., editors. AJCC cancer staging manual. 7th ed. New York: Springer; 2010. p. 143–164.
Hanley JA, McNeil BJ. The meaning and use of the area under a receiver operating characteristic (ROC) curve. Radiology. 1982;143:29–36.
Nakama H, Yamamoto M, Kamijo N, et al. Colonoscopic evaluation of immunochemical fecal occult blood test for detection of colorectal neoplasia. Hepatogastroenterology. 1999;46:228–31.
Morikawa T, Kato J, Yamaji Y, et al. A comparison of the immunochemical fecal occult blood test and total colonoscopy in the asymptomatic population. Gastroenterology. 2005;129:422–8.
Allison JE, Sakoda LC, Levin TR, et al. Screening for colorectal neoplasms with new fecal occult blood tests: update on performance characteristics. J Natl Cancer Inst. 2007;99:1462–70.
Park DI, Ryu S, Kim YH, et al. Comparison of guaiac-based and quantitative immunochemical fecal occult blood testing in a population at average risk undergoing colorectal cancer screening. Am J Gastroenterol. 2010;105:2017–25.
Kapidzic A, Grobbee EJ, Hol L, et al. Attendance and yield over three rounds of population-based fecal immunochemical test screening. Am J Gastroenterol. 2014;109:1257–64.
Jass JR, Whitehall VL, Young J, et al. Emerging concepts in colorectal neoplasia. Gastroenterology. 2002;123:862–76.
Jass JR. Classification of colorectal cancer based on correlation of clinical, morphological and molecular features. Histopathology. 2007;50:113–30.
Bar-Shalom R, Yefremov N, Guralnik L, et al. Clinical performance of PET/CT in evaluation of cancer: additional value for diagnostic imaging and patient management. J Nucl Med. 2003;44:1200–9.
Antoch G, Saoudi N, Kuehl H, et al. Accuracy of whole-body dual-modality fluorine-18-2-fluoro-2-deoxy-d-glucose positron emission tomography and computed tomography (FDG-PET/CT) for tumor staging in solid tumors: comparison with CT and PET. J Clin Oncol. 2004;22:4357–68.
Acknowledgment
This study was supported in part by the National Cancer Center Research and Development Fund (27-A-5).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Sekiguchi, M., Kakugawa, Y., Terauchi, T. et al. Sensitivity of 2-[18F]fluoro-2-deoxyglucose positron emission tomography for advanced colorectal neoplasms: a large-scale analysis of 7505 asymptomatic screening individuals. J Gastroenterol 51, 1122–1132 (2016). https://doi.org/10.1007/s00535-016-1201-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00535-016-1201-5