Abstract
Background
Galectin-3 has been independently correlated with malignant behavior in human colon cancer. The involvement of galectin-3 in the invasiveness of colon cancer cells remains to be determined. We investigated whether galectin-3 was involved in the colon cancer cell migration mediated by certain kinase pathways.
Methods
We studied 2 colon cancer cell lines (DLD-1 and Caco2) and clinical samples. Immunostaining and Western blotting were used to analyze the expression of galectin-3 in vitro and in the clinical samples. Short hairpin RNA and overexpression of galectin-3 were used to study loss- and gain-of-function in a wound-healing assay and a Transwell migration assay, and Western blotting was used to study the Ras–Raf signaling pathway.
Results
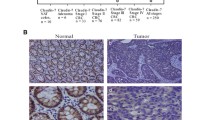
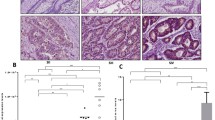
Galectin-3 was expressed at lower levels in DLD-1 than in Caco2 cells. The lower galectin-3 level in DLD-1 cells was associated with decreased cell migration, in comparison with that of Caco2 cells. Overexpression of galectin-3 increased the migration rate of DLD-1, while knockdown of galectin-3 decreased the migration. Overexpression of galectin-3 was correlated with increased lamellipodia formation and distal lung localization in a mouse model. The galectin-3 enhancement of DLD-1 cell migration was mediated by K-Ras, Raf and Erk1/2 pathway activation, but not the H-Ras, p38, or JNK activation.
Conclusions
Galectin-3 plays an important role in regulating colon cancer cell migration and potential distal localization. The galectin-3 enhancement of cell migration is mediated through the K-Ras–Raf–Erk1/2 pathway. Specific targeting of the K-Ras–Raf–Erk1/2 pathway may be useful for treating colon cancers associated with increased galectin-3 expression.







Similar content being viewed by others
References
Schoeppner HL, Raz A, Ho SB, Bresalier RS. Expression of an endogenous galactose-binding lectin correlates with neoplastic progression in the colon. Cancer. 1995;75:2818–26.
Sanjuán X, Fernández PL, Castells A, Castronovo V, van der Brule F, Liu FT, et al. Differential expression of galectin 3 and galectin 1 in colorectal cancer progression. Gastroenterology. 1997;113:1906–15.
Nagy N, Legendre H, Engels O, André S, Kaltner H, Wasano K, et al. Refined prognostic evaluation in colon carcinoma using immunohistochemical galectin finger-printing. Cancer. 2003;97:1849–58.
Glinsky VV, Glinsky GV, Glinskii OV, Huxley VH, Turk JR, Mossine VV, et al. Intravascular metastatic cancer cell homotypic aggregation at the sites of primary attachment to the endothelium. Cancer Res. 2003;63:3805–11.
Ellerhorst J, Nguyen T, Cooper DN, Lotan D, Lotan R. Differential expression of endogenous galectin-1 and galectin-3 in human prostate cancer cell lines and effects of overexpressing galectin-1 on cell phenotype. Int J Oncol. 1999;14:217–24.
Van den Brule F, Califice S, Garnier F, Fernandez PL, Berchuck A, Castronovo V. Galectin-1 accumulation in the ovary carcinoma peritumoral stroma is induced by ovary carcinoma cells and affects both cancer cell proliferation and adhesion to laminin-1 and fibronectin. Lab Invest. 2003;83:377–86.
Su WH, Chen HI, Jen CJ. Polymorphonuclear leukocyte transverse migration induces rapid alterations in endothelial focal contacts. J Leukoc Biol. 2007;82:542–50.
Chuang PC, Sun HS, Chen TM, Tsai SJ. Prostaglandin E2 induces fibroblast growth factor 9 via EP3-dependent protein kinase C delta and Elk-1 signaling. Mol Cell Biol. 2006;26:8281–92.
Raz A, Lotan R. Endogenous galactoside-binding lectins: a new class of functional tumor cell surface molecules related to metastasis. Cancer Metastasis Rev. 1987;6:433–52.
Kim S, Takahashi H, Lin WW, Descargues P, Grivennikov S, Kim Y, et al. Carcinoma-produced factors activate myeloid cells through TLR2 to stimulate metastasis. Nature. 2009;457:102–6.
Endo K, Kohnoe S, Tsujita E, Watanabe A, Nakashima H, Baba H, et al. Galectin-3 expression is a potent prognostic marker in colorectal cancer. Anticancer Res. 2005;25:3117–21.
Bresalier RS, Mazurek N, Sternberg LR, Byrd JC, Yunker CK, Nangia-Makker P, et al. Metastasis of human colon cancer is altered by modifying expression of the beta-galactoside-binding protein galectin 3. Gastroenterology. 1998;115:287–96.
Dudas SP, Yunker CK, Sternberg LR, Byrd JC, Bresalier RS. Expression of human intestinal mucin is modulated by the β-galactoside binding protein galectin-3 in colon cancer. Gastroenterology. 2002;123:817–26.
Saravanan C, Liu FT, Gipson IK, Panjwani N. Galectin-3 promotes lamellipodia formation in epithelial cells by interacting with complex N-glycans on alpha3beta1 integrin. J Cell Sci. 2009;122:3684–93.
Hsu DK, Chernyavsky AI, Chen HY, Yu L, Grando SA, Liu FT. Endogenous galectin-3 is localized in membrane lipid rafts and regulates migration of dendritic cells. J Invest Dermatol. 2009;129:573–83.
Wang Y, Nangia-Makker P, Tait L, Balan V, Hogan V, Pienta KJ, et al. Regulation of prostate cancer progression by galectin-3. Am J Pathol. 2009;174:1515–23.
Hittelet A, Legendre H, Nagy N, Bronckart Y, Pector JC, Salmon I, et al. Upregulation of galectins-1 and -3 in human colon cancer and their role in regulating cell migration. Int J Cancer. 2003;103:370–9.
Inder KL, Lau C, Loo D, Chaudhary N, Goodall A, Martin S, et al. Nucleophosmin and nucleolin regulate K-Ras plasma membrane interactions and MAPK signal transduction. J Biol Chem. 2009;284:28410–9.
Hoogwater FJ, Nijkamp MW, Smakman N, Steller EJ, Emmink BL, Westendorp BF, et al. Oncogenic K-Ras turns death receptors into metastasis-promoting receptors in human and mouse colorectal cancer cells. Gastroenterology. 2010;138:2357–67.
Dreissigacker U, Mueller MS, Unger M, Siegert P, Genze F, Gierschik P, et al. Oncogenic K-Ras down-regulates Rac1 and RhoA activity and enhances migration and invasion of pancreatic carcinoma cells through activation of p38. Cell Signal. 2006;18:1156–68.
Woods D, Cherwinski H, Venetsanakos E, Bhat A, Gysin S, Humbert M, et al. Induction of beta3-integrin gene expression by sustained activation of the Ras-regulated Raf-MEK-extracellular signal-regulated kinase signaling pathway. Mol Cell Biol. 2001;21:3192–205.
Shalom-Feuerstein R, Plowman SJ, Rotblat B, Ariotti N, Tian T, Hancock JF, et al. K-ras nanoclustering is subverted by overexpression of the scaffold protein galectin-3. Cancer Res. 2008;68(16):6608–16.
Acknowledgments
The study was supported by a Grant, CMRPG880621, CMRPG880622, from Chang Gung Memorial Hospital, Taiwan, and by Grant, NSC 100-2325-B-182A and 100-2325-B-182A-001 from the National Science Council, Taiwan, ROC. We thank Chang Gung Medical Foundation Kaohsiung Chang Gung Memorial Hospital, Tissue Bank (CLRPG 870463) for providing the study materials.
Conflict of interest
None.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Wu, KL., Huang, EY., Jhu, EW. et al. Overexpression of galectin-3 enhances migration of colon cancer cells related to activation of the K-Ras–Raf–Erk1/2 pathway. J Gastroenterol 48, 350–359 (2013). https://doi.org/10.1007/s00535-012-0663-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00535-012-0663-3