Abstract
Background
In recent years in Japan, the rate of clarithromycin (CAM) resistance in Helicobacter pylori has risen to around 30%, and the eradication rate with triple therapy [proton pump inhibitor + amoxicillin (AMPC) + CAM] has been trending downward to around 70%. In 2007, rabeprazole (RPZ)-based triple therapy (RPZ + AMPC + CAM: RAC therapy) was approved in Japan, and a large-scale nationwide study was therefore initiated to evaluate the efficacy and safety of RAC therapy in clinical practice.
Methods
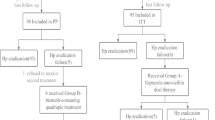
Patients with H. pylori-positive gastric/duodenal ulcer (including ulcer scars) were administered triple therapy comprising RPZ 10 mg, AMPC 750 mg, and CAM 200 mg (or 400 mg), twice daily for 7 days.
Results
The eradication rate was 80.7% (2,551/3,162). The results of multivariate analysis indicated the following as factors affecting the eradication rate: sex, treatment compliance, history of H. pylori treatment, presence of urologic disease, presence of respiratory disease, and year of starting treatment. The incidence of adverse drug reactions (such as diarrhea and dysgeusia) was 4.4% (166/3,789). The results of multivariate analysis indicated the following as factors affecting the incidence of adverse drug reactions: sex, daily CAM dose, and history of allergies.
Conclusion
In a large-scale nationwide study of use in clinical practice, RAC therapy was confirmed to be effective and safe.
Similar content being viewed by others
References
Ford AC, Delaney BC, Forman D, Moayyedi P. Eradication therapy in Helicobacter pylori positive peptic ulcer disease: systematic review and economic analysis. Am J Gastroenterol. 2004;99:1833–55.
Leodolter A, Kulig M, Brasch H, Meyer-Sabellek W, Willich SN, Malfertheiner P. A meta-analysis comparing eradication, healing and relapse rates in patients with Helicobacter pylori-associated gastric or duodenal ulcer. Aliment Pharmacol Ther. 2001;15:1949–58.
Malfertheiner P, Megraud F, O’Morain C, Bazzoli F, El-Omar E, Graham D, et al. Current concepts in the management of Helicobacter pylori infection: the Maastricht III Consensus Report. Gut. 2007;56:772–81.
Chey WD, Wong BC. Practice parameters committee of the American College of Gastroenterology. American College of Gastroenterology guideline on the management of Helicobacter pylori infection. Am J Gastroenterol. 2007;102:1808–25.
Asaka M, Kato M, Takahashi S, Fukuda Y, Sugiyama T, Ota H, et al. Guidelines for the management of Helicobacter pylori infection in Japan: 2009 revised edition. Helicobacter. 2010;15:1–20.
Kobayashi I, Murakami K, Kato M, Kato S, Azuma T, Takahashi S, et al. Changing antimicrobial susceptibility epidemiology of Helicobacter pylori strains in Japan between 2002 and 2005. J Clin Microbiol. 2007;45:4006–10.
Nasu M, Nishino T, Azuma K, Kawakami Y, Kobayashi I, Saito A, et al. A report of the amoxicillin and clarithromycin breakpoints of MIC in Helicobacter pylori eradication therapy. Jpn J Chemother. 2000;48:561–7. (in Japanese).
Kobayashi I, Murakami K, Kato M, Kato S, Azuma T, Takahashi S, et al. Current status of drug resistance in Helicobacter pylori in Japan: annual summary of drug resistance surveillance and 5-year summary report in 2008. Jpn J Helicobacter Res. 2009;10:98–103. (in Japanese).
Horiki N, Omata F, Uemura M, Suzuki S, Ishii N, Iizuka Y, et al. Annual change of primary resistance to clarithromycin among Helicobacter pylori isolates from 1996 through 2008 in Japan. Helicobacter. 2009;14:86–90.
Sasaki M, Ogasawara N, Utsumi K, Kawamura N, Kamiya T, Kataoka H, et al. Changes in 12-year first-line eradication rate of Helicobacter pylori based on triple therapy with PPI, AMPC and CAM. J Clin Biochem Nutr. 2010;47:53–8.
Tokunaga K, Tanaka A, Sugano H, Takahashi S. The present status and problems of Helicobacter pylori first-line eradication therapy. Nippon Rinsho. 2009;67:2291–6. (in Japanese, abstract in English).
Kuwayama H, Asaka M, Sugiyama T, Fukuda Y, Aoyama N, Hirai Y, et al. Rabeprazole-based eradication therapy for Helicobacter pylori: a large-scale study in Japan. Aliment Pharmacol Ther. 2007;25:1105–13.
Asaka M, Sugiyama T, Kato M, Satoh K, Kuwayama H, Fukuda Y, et al. A multicenter, double-blind study on triple therapy with lansoprazole, amoxicillin and clarithromycin for eradication of Helicobacter pylori in Japanese peptic ulcer patients. Helicobacter. 2001;6:254–61.
Kuwayama H, Luk G, Yoshida S, Nakamura T, Kubo M, Uemura N, et al. Efficacy of a low-dose omeprazole-based triple-therapy regimen for Helicobacter pylori eradication independent of cytochrome P450 genotype: the Japanese MACH Study. Clin Drug Investig. 2005;25:293–305.
Higuchi K, Maekawa T, Nakagawa K, Chouno S, Hayakumo T, Tomono N, et al. Efficacy and safety of Helicobacter pylori eradication therapy with omeprazole, amoxicillin and high- and low-dose clarithromycin in Japanese patients: a randomised, double-blind, multicentre study. Clin Drug Investig. 2006;26:403–14.
Sakamoto S, Chino F, Okubo H, Takahashi Y, Aoki H, Morita K, et al. Results of the drug use result survey of triple therapy with lansoprazole, amoxicillin, and clarithromycin for eradication of Helicobacter pylori in patients with peptic ulcer. J Clin Ther Med. 2006;22:21–30. (in Japanese, abstract in English).
Saitoh T, Fukushima Y, Otsuka H, Hirakawa J, Mori H, Asano T, et al. Effect of rabeprazole, lansoprazole and omeprazole on intragastric pH in CYP2C19 extensive metabolizers. Aliment Pharmacol Ther. 2002;16:1811–7.
Ohara T, Goshi S, Taneike I, Tamura Y, Zhang H, Yamamoto T. Inhibitory action of a novel proton pump inhibitor, rabeprazole, and its thioether derivative against the growth and motility of clarithromycin resistant Helicobacter pylori. Helicobacter. 2001;6:125–9.
Graham DY, Lew GM, Malaty HM, Evans DG, Evans DJ Jr, Klein PD, et al. Factors influencing the eradication of Helicobacter pylori with triple therapy. Gastroenterology. 1992;102:493–6.
Wermeille J, Cunningham M, Dederding JP, Girard L, Baumann R, Zelger G, et al. Failure of Helicobacter pylori eradication: is poor compliance the main cause? Gastroenterol Clin Biol. 2002;26:216–9.
Suzuki T, Matsuo K, Sawaki A, Wakai K, Hirose K, Ito H, et al. Influence of smoking and CYP2C19 genotypes on H. pylori eradication success. Epidemiol Infect. 2007;135:171–6.
Suzuki T, Matsuo K, Ito H, Sawai A, Hirose K, Wakai K, et al. Smoking increases the treatment failure for Helicobacter pylori eradication. Am J Med. 2006;119:217–24.
Namiot DB, Leszczynska K, Namiot Z, Kurylonek AJ, Kemona A. Smoking and drinking habits are important predictors of Helicobacter pylori eradication. Adv Med Sci. 2008;53:310–5.
Murakami K, Fujioka T, Okimoto T, Sato R, Kodama M, Nasu M. Drug combinations with amoxycillin reduce selection of clarithromycin resistance during Helicobacter pylori eradication therapy. Int J Antimicrob Agents. 2002;19:67–70.
Murakami K, Fujioka T. Treatment for Helicobacter pylori eradication and drug resistance in Japan. Helicobacter Res. 1998;2:423–8. (in Japanese).
Hoshiya S, Watanabe K, Tokunaga K, Tanaka A, Ninomiya H, Shingaki M, et al. Relationship between eradication therapy and clarithromycin-resistant Helicobacter pylori in Japan. J Gastroenterol. 2000;35:10–4.
Wurzer H, Rodrigo L, Stamler D, Archambault A, Rokkas T, Skandalis N, et al. Short-course therapy with amoxycillin-clarithromycin triple therapy for 10 days (ACT-10) eradicated Helicobacter pylori and heals duodenal ulcer. Aliment Pharmacol Ther. 1997;11:943–52.
Kihira K, Satoh K, Saifuku K, Kawakami S, Fukazawa K, Ishino Y, et al. Rabeprazole, amoxycillin and low- or high-dose clarithromycin for cure of Helicobacter pylori infection. Aliment Pharmacol Ther. 2000;14:1083–7.
Fukase K, Kato M, Kikuchi S, Inoue K, Uemura N, Okamoto S, et al. Effect of eradication of Helicobacter pylori on incidence of metachronous gastric carcinoma after endoscopic resection of early gastric cancer: an open-label, randomised controlled trial. Lancet. 2008;372:392–7.
Acknowledgments
This study was conducted by Eisai Co., Ltd., based on instructions from the Ministry of Health, Labour and Welfare of Japan at the time of RAC therapy approval. We would like to express our sincere gratitude to all the investigators and sub-investigators at the 754 facilities nationwide who cooperated with this study and provided crucial data. Toshio Fujioka, M.D., Ph.D., serves as a consultant to Eisai Co., Ltd.
Conflict of interest
None.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Fujioka, T., Aoyama, N., Sakai, K. et al. A large-scale nationwide multicenter prospective observational study of triple therapy using rabeprazole, amoxicillin, and clarithromycin for Helicobacter pylori eradication in Japan. J Gastroenterol 47, 276–283 (2012). https://doi.org/10.1007/s00535-011-0487-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00535-011-0487-6