Abstract
Background
This prospective study was to assess the safety and feasibility of daily granulocyte and monocyte adsorptive apheresis (GMA) therapy in patients with active ulcerative colitis (UC).
Methods
Thirty consecutive patients with moderately or severely active UC received daily GMA treatment (5 sessions over 5 consecutive days) with the Adacolumn. Adverse events (AE), patient tolerability, and clinical symptoms were monitored daily.
Results
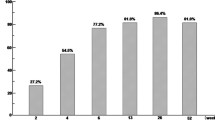
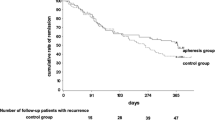
Sixteen patients (53%) experienced AE during at least one GMA session. The most frequent AE was mild headache followed by fatigue and fever. None of the AE was serious, and all patients completed the 5 consecutive GMA sessions. Clinical symptoms (stool frequency and/or rectal bleeding) were improved in 21 patients (70%) during the course of GMA therapy. Clinical remission defined as normal stool frequency and no rectal bleeding was achieved in 7 patients (23%) after 5 GMA sessions. Seven of 20 patients (35%) with moderately active disease achieved clinical remission, whereas none of the 10 patients with severely active disease achieved clinical remission. Total and differential leukocyte counts, platelet count, and hemoglobin level did not significantly change, but C-reactive protein level significantly decreased during the course of GMA therapy.
Conclusions
This is the first report on daily GMA in the treatment of patients with UC. Daily GMA was safe and well tolerated without serious AE. Furthermore, daily GMA was associated with rapid improvement of clinical symptoms in patients with moderately active UC. However, controlled trials are warranted to assess a definite efficacy for daily GMA therapy.


Similar content being viewed by others
References
Shimoyama T, Sawada K, Hiwatashi N, Sawada T, Matsueda K, Munakata A, et al. Safety and efficacy of granulocyte and monocyte adsorption apheresis in patients with active ulcerative colitis: a multicenter study. J Clin Apher. 2001;16:1–9.
Saniabadi AR, Hanai H, Takeuchi K, Umemura K, Nakashima M, Adachi T, et al. Adacolumn, an adsorptive carrier based granulocyte and monocyte apheresis device for the treatment of inflammatory and refractory diseases associated with leukocytes. Therap Apher Dial. 2003;7:48–59.
Hanai H, Watanabe F, Takeuchi K, Iida T, Yamada M, Iwaoka Y, et al. Leukocyte adsorptive apheresis for the treatment of active ulcerative colitis: a prospective, uncontrolled, pilot study. Clin Gastroenterol Hepatol. 2003;1:28–35.
Suzuki Y, Yoshimura N, Saniabadi AR, Saito Y. Selective granulocyte and monocyte adsorptive apheresis as a first-line treatment for steroid naïve patients with active ulcerative colitis: a prospective uncontrolled study. Dig Dis Sci. 2004;49:565–71.
Naganuma M, Funakoshi S, Sakuraba A, Takagi H, Inoue N, Ogata H, et al. Granulocytapheresis is useful as an alternative therapy in patients with steroid-refractory or -dependent ulcerative colitis. Inflamm Bowel Dis. 2004;10:251–7.
Hanai H, Watanabe F, Yamada M, Sato Y, Takeuchi K, Iida T, et al. Adsorptive granulocyte and monocyte apheresis versus prednisolone in patients with corticosteroid-dependent moderately severe ulcerative colitis. Digestion. 2004;70:36–44.
Yamamoto T, Umegae S, Kitagawa T, Yasuda Y, Yamada Y, Takahashi D, et al. Granulocyte and monocyte adsorptive apheresis in the treatment of active distal ulcerative colitis: a prospective, pilot study. Aliment Pharmacol Ther. 2004;20:783–92.
Yamamoto T, Saniabadi AR, Maruyama Y, Umegae S, Matsumoto K. Factors affecting clinical and endoscopic efficacies of selective leucocytapheresis for ulcerative colitis. Dig Liver Dis. 2007;39:626–33.
Habermalz B, Sauerland S. Clinical effectiveness of selective granulocyte, monocyte adsorptive apheresis with the Adacolumn device in ulcerative colitis. Dig Dis Sci. 2010;55:1421–8.
Sakuraba A, Motoya S, Watanabe K, Nishishita M, Kanke K, Matsui T, et al. An open-label prospective randomized multicenter study shows very rapid remission of ulcerative colitis by intensive granulocyte and monocyte adsorptive apheresis as compared with routine weekly treatment. Am J Gastroenterol. 2009;104:2990–5.
Sakuraba A, Sato T, Naganuma M, Morohoshi Y, Matsuoka K, Inoue N, et al. A pilot open-labeled prospective randomized study between weekly and intensive treatment of granulocyte and monocyte adsorption apheresis for active ulcerative colitis. J Gastroenterol. 2008;43:51–6.
Sutherland LR, Martin F, Greer S, Robinson M, Greenberger N, Saibil F, et al. 5-Aminosalicylic acid enema in the treatment of distal ulcerative colitis, proctosigmoiditis, and proctitis. Gastroenterology. 1987;92:1984–8.
Yamamoto T, Umegae S, Matsumoto K. Mucosal healing in patients with ulcerative colitis during a course of selective leukocytapheresis therapy: a prospective cohort study. Inflamm Bowel Dis. 2010;16:1905–11.
Norda R, Berséus O, Stegmayr B. Adverse events and problems in therapeutic hemapheresis. A report from the Swedish registry. Transfus Apher Sci. 2001;25:33–41.
Kanai T, Hibi T, Watanabe M. The logics of leukocytapheresis as a natural biological therapy for inflammatory bowel disease. Expert Opin Biol Ther. 2006;6:453–66.
Sakata Y, Iwakiri R, Amemori S, Yamaguchi K, Fujise T, Otani H, et al. Comparison of the efficacy of granulocyte and monocyte/macrophage adsorptive apheresis and leucocytapheresis in active ulcerative colitis patients: a prospective randomized study. Eur J Gastroenterol Hepatol. 2008;20:629–33.
Tsujikawa T, Andoh A, Ogaẃa A, Sonoda A, Yagi Y, Hata K, et al. Feasibility of five days of consecutive leukocytapheresis for the treatment of ulcerative colitis: a preliminary study. Ther Apher Dial. 2009;13:14–8.
Saniabadi AR, Hanai H, Fukunaga K, Sawada K, Shima C, Bjarnason I, et al. Therapeutic leukocytapheresis for inflammatory bowel disease. Transfus Apher Sci. 2007;37:191–200.
Conflict of interest
The authors declare that they have no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Yamamoto, T., Umegae, S. & Matsumoto, K. Daily granulocyte and monocyte adsorptive apheresis in patients with active ulcerative colitis: a prospective safety and feasibility study. J Gastroenterol 46, 1003–1009 (2011). https://doi.org/10.1007/s00535-011-0428-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00535-011-0428-4