Abstract
Background
Advanced glycation endproducts (AGEs), final reaction products of protein with sugars, are known to contribute to various disorders, including diabetes, aspects of aging, and neurodegenerative diseases. Recently, we reported elevated levels of serum AGEs in patients with nonalcoholic steatohepatitis (NASH); further, we found that AGEs induced the generation of reactive oxygen species followed by the proliferation and activation of hepatic stellate cells, a major contributor to liver fibrosis. In this study, to explore the clinical usefulness of AGEs as a biomarker for the attenuation of NASH, we investigated whether the treatment of NASH with dyslipidemia could decrease serum levels of AGEs.
Methods
This study included 43 patients with biopsy-proven NASH with dyslipidemia. Serum glyceraldehyde-derived AGE measurements and clinical laboratory tests were performed periodically during an open-label study of atorvastatin (10 mg daily) for 12 months. Standard weight-loss counseling was continued during the treatment period. Oral glucose tolerance tests and liver density assessment by computerized tomography were performed before and after treatment. Follow-up liver biopsy was performed in 22 patients.
Results
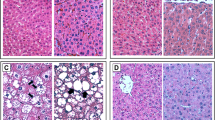
All 43 patients had dyslipidemia. The body mass indexes and serum glucose levels did not change during the treatment. After the treatment, NASH-related metabolic parameters were significantly improved. Serum glyceraldehyde-derived AGE levels were significantly decreased (10.4 ± 3.8 and 2.5 ± 1.1 IU/mL before and after treatment, respectively). The steatosis grade and nonalcoholic fatty liver disease (NAFLD) activity score were significantly improved.
Conclusions
The present data demonstrated that atorvastatin decreased the serum levels of AGEs in NASH patients with dyslipidemia and suggest the usefulness of AGEs as a biomarker for the attenuation of NASH.

Similar content being viewed by others
Abbreviations
- NASH:
-
Nonalcoholic steatohepatitis
- NAFLD:
-
Nonalcoholic fatty liver disease
- IR:
-
Insulin resistance
- glycer-AGE:
-
Glyceraldehyde-derived advanced glycation endproducts
- AGEs:
-
Advanced glycation endproducts
- RAGE:
-
Receptor for AGEs
- ROS:
-
Reactive oxygen species
- TBARS:
-
Thiobarbituric acid reactive substances
- OGTT:
-
Oral glucose tolerance test
- IGT:
-
Impaired glucose tolerance
- DM:
-
Diabetes mellitus
- HOMA-IR:
-
Homeostasis model-IR: NAS
- NAFLD:
-
Activity score
- IGT:
-
Impaired glucose tolerance
- BMI:
-
Body mass index
- TNF-α:
-
Tumor necrosis factor-α
- FFA:
-
Free fatty acid
- P-III-P:
-
Procollagen type III propeptide
- HMG-CoA:
-
3-Hydroxy-3-methylglutaryl-coenzyme A
References
Teli MR, James OF, Burt AD, Bennett MK, Day CP. The natural history of nonalcoholic fatty liver: a follow-up study. Hepatology. 1995;22:1714–9.
Dam-Larsen S, Franzmann M, Andersen IB, Christoffersen P, Jensen LB, Sørensen TI, et al. Long term prognosis of fatty liver: risk of chronic liver disease and death. Gut. 2004;53:750–5.
Matteoni CA, Younossi ZM, Gramlich T, Boparai N, Liu YC, McCullough AJ. Nonalcoholic fatty liver disease: a spectrum of clinical and pathological severity. Gastroenterology. 1999;116:1413–9.
James O, Day C. Non-alcoholic steatohepatitis: another disease of affluence. Lancet. 1999;353:1634–6.
Chitturi S, Abeygunasekera S, Farrell GC, Holmes-Walker J, Hui JM, Fung C, et al. NASH and insulin resistance: Insulin hypersecretion and specific association with the insulin resistance syndrome. Hepatology. 2002;35:373–9.
Marchesini G, Brizi M, Bianchi G, Tomassetti S, Bugianesi E, Lenzi M, et al. Nonalcoholic fatty liver disease: a feature of the metabolic syndrome. Diabetes. 2001;50:1844–50.
Hui JM, Farrell GC. Clear messages from sonographic shadows? Links between metabolic disorders and liver disease, and what to do about them. J Gastroenterol Hepatol. 2003;18:1115–7.
Marchesini G, Bugianesi E, Forlani G, Cerrelli F, Lenzi M, Manini R, et al. Nonalcoholic fatty liver, steatohepatitis, and the metabolic syndrome. Hepatology. 2003;37:917–23.
Hyogo H, Iwamoto K, Arihiro K, Takeuchi M, Sato T, Ochi H, et al. Elevated levels of serum advanced glycation end products in patients with nonalcoholic steatohepatitis. J Gastroenterol Hepatol. 2007;22:1114–9.
Yamagishi S, Takeuchi M, Inagaki Y, Nakamura K, Imaizumi T. Role of advanced glycation end products (AGEs) and their receptor (RAGE) in the pathogenesis of diabetic microangiopathy. Int J Clin Pharmacol Res. 2003;23:129–34.
Inagaki Y, Yamagishi S, Okamoto T, Takeuchi M, Amano S. Pigment epithelium-derived factor prevents advanced glycation end products-induced monocyte chemoattractant protein-1 production in microvascular endothelial cells by suppressing intracellular reactive oxygen species generation. Diabetologia. 2003;46:284–7.
Yamagishi S, Inagaki Y, Amano S, Okamoto T, Takeuchi M, Makita Z. Pigment epithelium-derived factor protects cultured retinal pericytes from advanced glycation end product-induced injury through its antioxidative properties. Biochem Biophys Res Commun. 2002;296:877–82.
Miele C, Riboulet A, Maitan MA, Oriente F, Romano C, Formisano P, et al. Human glycated albumin affects glucose metabolism in L6 skeletal muscle cells by impairing insulin-induced insulin receptor substrate (IRS) signaling through a protein kinase C alpha-mediated mechanism. J Biol Chem. 2003;278:47376–87.
Yamagishi S, Imaizumi T. Diabetic vascular complications: pathophysiology, biochemical basis and potential therapeutic strategy. Curr Pharm Des. 2005;11:2279–99.
Iwamoto K, Kanno K, Hyogo H, Yamagishi S, Takeuchi M, Tazuma S. Advanced glycation end products enhance the proliferation and activation of hepatic stellate cells. J Gastroenterol. 2008;43:298–304.
Hyogo H, Yamagishi S. Advanced glycation end products (AGEs) and their involvement in liver disease. Curr Pharm Des. 2008;14:969–72.
Whalan DJ. The ethics and morality of clinical trials in man. Med J Aust. 1975;1:491–4.
Hyogo H, Tazuma S, Arihiro K, Iwamoto K, Nabeshima Y, Inoue M, et al. Efficacy of atorvastatin for the treatment of nonalcoholic steatohepatitis with dyslipidemia. Metabolism. 2008;57:1711–8.
Report of the Expert Committee on the Diagnosis and Classification of Diabetes Mellitus. Diabetes Care. 2003;26(Suppl 1):S5–20.
Matthews DR, Hosker JP, Rudenski AS, Naylor BA, Treacher DF, Turner RC. Homeostasis model assessment: insulin resistance and beta-cell function from fasting plasma glucose and insulin concentrations in man. Diabetologia. 1985;28:412–9.
Takeuchi M, Makita Z, Bucala R, Suzuki T, Koike T, Kameda Y. Immunological evidence that non-carboxymethyllysine advanced glycation end-products are produced from short chain sugars and dicarbonyl compounds in vivo. Mol Med. 2000;6:114–25.
Kleiner DE, Brunt EM, Van Natta M, Behling C, Contos MJ, Cummings OW, et al. Nonalcoholic Steatohepatitis Clinical Research Network. Design and validation of a histological scoring system for nonalcoholic fatty liver disease. Hepatology. 2005;41:1313–21.
Japanese Society for the Study of Obesity. New criteria of obesity (in Japanese). J Jpn Soc Study Obes. 2000;6:18–28.
Kiyici M, Gulten M, Gurel S, Nak SG, Dolar E, Savci G, et al. Ursodeoxycholic acid and atorvastatin in the treatment of nonalcoholic steatohepatitis. Can J Gastroenterol. 2003;17:713–8.
Hatzitolios A, Savopoulos C, Lazaraki G, Sidiropoulos I, Haritanti P, Lefkopoulos A, et al. Efficacy of omega-3 acids, atorvastatin and orlistat in non-alcoholic fatty liver disease with dyslipidemia. Indian J Gastroenterol. 2004;23:127–8.
Gómez-Domínguez E, Gisbert JP, Moreno-Monteagudo JA, García-Buey L, Moreno-Otero R. A pilot study of atorvastatin treatment in dyslipemid, non-alcoholic fatty liver patients. Aliment Pharmacol Ther. 2006;23:1643–7.
Athyros VG, Mikhailidis DP, Didangelos TP, Giouleme OI, Liberopoulos EN, Karagiannis A, et al. Effect of multifactorial treatment on non-alcoholic fatty liver disease in metabolic syndrome: a randomised study. Curr Med Res Opin. 2006;22:873–83.
Funatsu T, Goto M, Kakuta H, Suzuki M, Ida M, Nishijima S, et al. Reduction in hepatic nonesterified fatty acid concentration after long-term treatment with atorvastatin lowers hepatic triglyceride synthesis and its secretion in sucrose-fed rats. Biochim Biophys Acta. 2002;1580:161–70.
Isusi E, Aspichueta P, Liza M, Hernández ML, Díaz C, Hernández G, et al. Short- and long-term effects of atorvastatin, lovastatin and simvastatin on the cellular metabolism of cholesteryl esters and VLDL secretion in rat hepatocytes. Atherosclerosis. 2000;153:283–94.
Kainuma M, Fujimoto M, Sekiya N, Tsuneyama K, Cheng C, Takano Y, et al. Cholesterol-fed rabbit as a unique model of nonalcoholic, nonobese, non–insulin-resistant fatty liver disease with characteristic fibrosis. J Gastroenterol. 2006;41:971–80.
Nakamura K, Yamagishi SI, Matsui T, Adachi H, Takeuchi M, Imaizumi T, et al. Serum levels of soluble form of receptor for advanced glycation end products (sRAGE) are correlated with AGEs in both diabetic and non-diabetic subjects. Clin Exp Med. 2007;7:188–90.
Yamagishi SI, Matsui T, Adachi H, Takeuchi M. Positive association of circulating levels of advanced glycation end products (AGEs) with pigment epithelium-derived factor (PEDF) in a general population. Pharmacol Res. 2009; doi:10.1016/j.phrs.2009.07.003.
Nakamura K, Yamagishi S, Adachi H, Matsui T, Kurita-Nakamura Y, Takeuchi M, et al. Circulating advanced glycation end products (AGEs) and soluble form of receptor for AGEs (sRAGE) are independent determinants of serum monocyte chemoattractant protein-1 (MCP-1) levels in patients with type 2 diabetes. Diabetes Metab Res Rev. 2008;24:109–14.
Yamagishi S, Amano S, Inagaki Y, Okamoto T, Koga K, Sasaki N, et al. Advanced glycation end products-induced apoptosis and overexpression of vascular endothelial growth factor in bovine retinal pericytes. Biochem Biophys Res Commun. 2002;290:973–8.
Yamamoto Y, Kato I, Doi T, Yonekura H, Ohashi S, Takeuchi M, et al. Development and prevention of advanced diabetic nephropathy in RAGE-overexpressing mice. J Clin Invest. 2001;108:261–8.
Yamamoto Y, Yamagishi S, Yonekura H, Doi T, Tsuji H, Kato I, et al. Roles of the AGE–RAGE system in vascular injury in diabetes. Ann N Y Acad Sci. 2000;902:163–70. discussion 170–2.
Ishitobi T, Hyogo H, Yamagishi S, Kimura Y, Nabeshimai Y, Tazuma S, et al. Effects of Advanced Glycation End-products and High Glucose on Hepatocytes. Jpn Pharmacol Ther. 2008;36:189–91.
Jinnouchi Y, Yamagishi S, Takeuchi M, Ishida S, Jinnouchi Y, Jinnouchi J, et al. Atorvastatin decreases serum levels of advanced glycation end products (AGEs) in patients with type 2 diabetes. Clin Exp Med. 2006;6:191–3.
Nishikawa T, Edelstein D, Du XL, Yamagishi S, Matsumura T, Kaneda Y, et al. Normalizing mitochondrial superoxide production blocks three pathways of hyperglycaemic damage. Nature. 2000;404:787–90.
Aviram M, Rosenblat M, Bisgaier CL, Newton RS. Atorvastatin and gemfibrozil metabolites, but not the parent drugs, are potent antioxidants against lipoprotein oxidation. Atherosclerosis. 1998;138:271–80.
Yoshiike N, Lwin H. Epidemiological aspects of obesity and NASH/NAFLD in Japan. Hepatol Res. 2005;33:77–82.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kimura, Y., Hyogo, H., Yamagishi, Si. et al. Atorvastatin decreases serum levels of advanced glycation endproducts (AGEs) in nonalcoholic steatohepatitis (NASH) patients with dyslipidemia: clinical usefulness of AGEs as a biomarker for the attenuation of NASH. J Gastroenterol 45, 750–757 (2010). https://doi.org/10.1007/s00535-010-0203-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00535-010-0203-y