Abstract
Background
The luminal surface of the gastrointestinal tract is covered by a viscoelastic gel layer that acts as a protective barrier against the intraluminal environment. Because the situation of the small intestine has not been elucidated to the same degree as other sections, in this study, we investigated the effects of indomethacin on the rat small intestinal mucosa.
Methods
Male Wistar rats were given indomethacin 10 mg/kg s-c and sacrificed 1, 3, 7, or 14 days later. The small intestine was opened along the anti-mesenteric side, and examined macroscopically. Total mucin content in the small intestinal epithelium was measured and immunoreactivity was examined using anti-mucin monoclonal antibodies HCM31 and PGM34.
Results
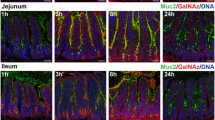
Indomethacin caused punched out and linear ulcers located mostly along the mesenteric margin of the distal jejunum with sparing of the ileum. Histological examination showed sialomucin recognized by HCM31 increased on day 3 especially in the regenerating epithelium around the ulcer edge. Furthermore, the surface mucous gel layer displayed a multilaminated pattern, consisting of non-sulfated sialomucin-rich layers and sulfated mucin-rich layers, where both mucins had the common core protein, MUC2. Biochemical measurements also showed the total mucin content of the jejunum increased transiently and HCM31-positive mucin increased approximately 4 times greater than baseline on day 3, but no marked changes were observed in the ileum, with few ulcers observed.
Conclusions
Indomethacin administration causes quantitative and qualitative change in jejunal mucin. In particular, sialomucin plays an important role in regenerating epithelium during the healing process following indomethacin-induced mucosal damage.







Similar content being viewed by others
References
Walker WA. Development of the intestinal mucosal barrier. J Pediatr Gastroenterol Nutr. 2002;34(Suppl 1):S33–9.
Corfield AP, Myerscough N, Longman R, Sylvester P, Arul S, Pignatelli M. Mucins and mucosal protection in the gastrointestinal tract: new prospects for mucins in the pathology of gastrointestinal disease. Gut. 2000;47:589–94.
Takeuchi K, Miyazawa T, Tanaka A, Kato S, Kunikata T. Pathogenic importance of intestinal hypermotility in NSAID-induced small intestinal damage in rats. Digestion. 2002;66:30–41.
Tanaka A, Matsumoto M, Hayashi Y, Takeuchi K. Functional mechanism underlying cyclooxygenase-2 expression in rat small intestine following administration of indomethacin: relation to intestinal hypermotility. J Gastroenterol Hepatol. 2005;20:38–45.
Satoh H, Guth PH, Grossman MI. Role of food in gastrointestinal ulceration produced by indomethacin in the rat. Gastroenterology. 1982;83:210–5.
Evans SM, Whittle BJ. Role of bacteria and inducible nitric oxide synthase activity in the systemic inflammatory microvascular response provoked by indomethacin in the rat. Eur J Pharmacol. 2003;461:63–71.
Hagiwara M, Kataoka K, Arimochi H, Kuwahara T, Ohnishi Y. Role of unbalanced growth of gram-negative bacteria in ileal ulcer formation in rats treated with a nonsteroidal anti-inflammatory drug. J Med Invest. 2004;51:43–51.
Reuter BK, Davies NM, Wallace JL. Nonsteroidal anti-inflammatory drug enteropathy in rats: role of permeability, bacteria, and enterohepatic circulation. Gastroenterology. 1997;112:109–17.
Hollander F. The two-component mucous barrier; its activity in protecting the gastroduodenal mucosa against peptic ulceration. AMA Arch Intern Med. 1954;93:107–20.
Menguy R. Gastric mucus and the gastric mucous barrier. A review. Am J Surg. 1969;117:806–12.
Allen A. Structure of gastrointestinal mucus glycoproteins and the viscous and gel-forming properties of mucus. Br Med Bull. 1978;34:28–33.
Keshav S, Lawson L, Chung LP, Stein M, Perry VH, Gordon S. Tumor necrosis factor mRNA localized to Paneth cells of normal murine intestinal epithelium by in situ hybridization. J Exp Med. 1990;171:327–32.
Molmenti EP, Perlmutter DH, Rubin DC. Cell-specific expression of alpha 1-antitrypsin in human intestinal epithelium. J Clin Invest. 1993;92:2022–34.
Peeters T, Vantrappen G. The Paneth cell: a source of intestinal lysozyme. Gut. 1975;16:553–8.
Meyer-Hoffert U, Hornef MW, Henriques-Normark B, Axelsson LG, Midtvedt T, Putsep K, Andersson M. Secreted enteric antimicrobial activity localizes to the mucus surface layer. Gut. 2008;57:764–71.
Azuumi Y, Ohara S, Ishihara K, Okabe H, Hotta K. Correlation of quantitative changes of gastric mucosal glycoproteins with aspirin-induced gastric damage in rats. Gut. 1980;21:533–6.
Ikezawa T, Ichikawa T, Adachi K, Sugano S, Ojima T, Nakamura Y, et al. Analysis of mucin composition in gastric juices of chronic rheumatic patients with upper gastrointestinal damage. Biomed Res. 2005;26:147–51.
Kojima Y, Ishihara K, Komuro Y, Saigenji K, Hotta K. Effects of the muscarinic receptor agonist carbachol and/or antagonist pirenzepine on gastric mucus secretion in rats. Scand J Gastroenterol. 1993;28:647–51.
Ikezawa T, Goso Y, Ichikawa T, Hayashida H, Kurihara M, Okayasu I, et al. Appearance of specific mucins recognized by monoclonal antibodies in rat gastric mucosa healing from HCl-induced gastric mucosal damage. J Gastroenterol. 2004;39:113–9.
Ishihara K, Kurihara M, Goso Y, Ota H, Katsuyama T, Hotta K. Establishment of monoclonal antibodies against carbohydrate moiety of gastric mucins distributed in the different sites and layers of rat gastric mucosa. Glycoconj J. 1996;13:857–64.
Ikezawa T, Goso Y, Ichikawa T, Hayashida H, Nakamura T, Kurihara M, et al. Immunohistochemical localization in rat gastrointestinal tract of a sialomucin species recognized by HCM31, a new anti-mucin monoclonal antibody. Biomed Res. 2002;23:63–8.
Tsubokawa D, Goso Y, Sawaguchi A, Kurihara M, Ichikawa T, Sato N, et al. A monoclonal antibody, PGM34, against 6-sulfated blood-group H type 2 antigen, on the carbohydrate moiety of mucin. FEBS J. 2007;274:1833–48.
Anthony A, Dhillon AP, Thrasivoulou C, Pounder RE, Wakefield AJ. Pre-ulcerative villous contraction and microvascular occlusion induced by indomethacin in the rat jejunum: a detailed morphological study. Aliment Pharmacol Ther. 1995;9:605–13.
Wax J, Clinger WA, Varner P, Bass P, Winder CV. Relationship of the enterohepatic cycle to ulcerogenesis in the rat small bowel with flufenamic acid. Gastroenterology. 1970;58:772–80.
Anthony A, Pounder RE, Dhillon AP, Wakefield AJ. Vascular anatomy defines sites of indomethacin induced jejunal ulceration along the mesenteric margin. Gut. 1997;41:763–70.
Saegusa Y, Ichikawa T, Iwai T, Goso Y, Okayasu I, Ikezawa T, Shikama N, Saigenji K, Ishihara K. Changes in the mucus barrier of the rat during 5-fluorouracil-induced gastrointestinal mucositis. Scand J Gastroenterol. 2008;43:59–65.
Kato S, Tanaka A, Kunikata T, Umeda M, Takeuchi K. Protective effect of lafutidine against indomethacin-induced intestinal ulceration in rats: relation to capsaicin-sensitive sensory neurons. Digestion. 2000;61:39–46.
Hatazawa R, Ohno R, Tanigami M, Tanaka A, Takeuchi K. Roles of endogenous prostaglandins and cyclooxygenase isozymes in healing of indomethacin-induced small intestinal lesions in rats. J Pharmacol Exp Ther. 2006;318:691–9.
Azuumi Y, Ishihara K, Ohara S, Okabe H, Hotta K. Efficacy of anti-ulcer drugs on the recovery of gastric mucosal glycoproteins with aspirin-induced gastric damage in rat. Gastroenterol Jpn. 1981;16:331–4.
Kojima Y, Ishihara K, Ohara S, Saigenji K, Hotta K. Effects of the M1 muscarinic receptor antagonist pirenzepine on gastric mucus glycoprotein in rats with or without ethanol-induced gastric damage. Scand J Gastroenterol. 1992;27:764–8.
Yusuf S, Nok AJ, Ameh DA, Adelaiye AB, Balogun EO. Correlation of gastric mucosal damage with sialic acid profile in rats: effect of hydrochloric acid, pepsin and hypertonic saline. Cell Biochem Funct. 2005;23:339–45.
Ho SB, Takamura K, Anway R, Shekels LL, Toribara NW, Ota H. The adherent gastric mucous layer is composed of alternating layers of MUC5AC and MUC6 mucin proteins. Dig Dis Sci. 2004;49:1598–606.
Matsuo K, Ota H, Akamatsu T, Sugiyama A, Katsuyama T. Histochemistry of the surface mucous gel layer of the human colon. Gut. 1997;40:782–9.
Acknowledgments
We express our sincere thanks to S. Sugawara, M. Hashimura, and Y. Numata for technical assistance. Part of this work was supported by Grants from the Shin-Caterpillar Mitsubishi Co., Grants from the SRL Co., and Grants from the Integrative Research Project of the Graduate School of Medical Sciences, Kitasato University.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Iwai, T., Ichikawa, T., Goso, Y. et al. Effects of indomethacin on the rat small intestinal mucosa: immunohistochemical and biochemical studies using anti-mucin monoclonal antibodies. J Gastroenterol 44, 277–284 (2009). https://doi.org/10.1007/s00535-009-0007-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00535-009-0007-0