Abstract
Background
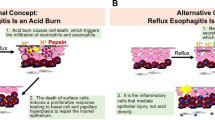
Reflux of the duodenal contents with gastric acid has been reported to contribute to the development of esophageal mucosal damage and inflammation. Recent studies show that pancreatic trypsin can stimulate the production of inflammatory mediators, including chemokines and prostaglandins from human esophageal epithelial cells in vitro. The aim of the present study was to investigate the role of pancreatic trypsin in the pathogenesis of chronic esophageal inflammation induced by gastroduodenal reflux in rats.
Methods
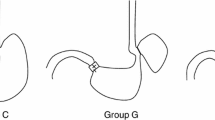
Esophagogastroduodenal anastomosis was carried out in male Wistar rats by anastomosing the jejunum to the gastroesophageal junction under diethyl ether inhalation anesthesia. The animals undergoing surgery were treated with the control diet, rabeprazole sodium, nizatidine, ecabet sodium, camostat mesilate (CMM), ONO-1714, a specific inducible nitric oxide synthase (iNOS) inhibitor, or meloxicam, a selective cyclooxygenase-2 (COX-2) inhibitor. Esophageal injury was evaluated by macroscopic and microscopic findings, and mRNA expression for CINC-1, COX-2, and iNOS was determined by real-time polymerase chain reaction (PCR). Trypsin activity within the esophageal lumen was measured 2 weeks after surgery, and the expression of protease-activated receptor (PAR)-1 and -2 was confirmed by reverse transcription (RT)-PCR.
Results
At 8 weeks after surgery, gastroduodenal reflux induced esophageal erosions and ulcer formation as well as marked thickening of the esophageal wall. Histological study showed an increase of thickness of the esopha-geal mucosa, hyperplasia of the epidermis and basal cells, ulcer formation, and marked infiltration of inflammatory cells. The macroscopic ulcer score and histological ulcer length were significantly reduced by treatment with rabeprazole or CMM but not by nizatidine or ecabet sodium, compared with each control. Rabeprazole, nizatidine, or ecabet sodium did not affect the severity of mucosal hyperplastic scores or histological parameters in esophagitis. In contrast, the CMM group showed a significant decrease in the mucosal hyperplastic and inflammatory scores. The enhanced expression of CINC-1, COX-2, and iNOS mRNA in the control group was also markedly inhibited in the CMM-treated group. ONO-1714 or meloxicam treatment significantly reduced the macroscopic scores of ulcer and hyperplasia. The trypsin activity in the esophageal lumen was significantly increased in the control diet group, and this increase was significantly inhibited in the CMM-treated group. The expression of PAR-1 and -2 mRNA was confirmed in rat esophageal epithelium.
Conclusions
With this model, we have demonstrated that CMM significantly reduces inflammation and hyperplasia in the esophageal mucosa. These results indicate that trypsin, which is primarily inhibited by CMM, plays an important role in the mucosal damage induced by gastroduodenal reflux and that it can be a therapeutic target in patients with gastroduodenal reflux esophagitis.
Similar content being viewed by others
References
N Chiba (1997) ArticleTitleProton pump inhibitors in acute healing and maintenance of erosive or worse esophagitis: a systematic overview Can J Gastroenterol 11 Suppl B 66B–73B Occurrence Handle1:STN:280:DyaK1c%2Fgs1Ogsg%3D%3D Occurrence Handle9347181
M Robinson F Lanza D Avner M Haber (1996) ArticleTitleEffective maintenance treatment of reflux esophagitis with low-dose lansoprazole. A randomized, double-blind, placebo-controlled trial Ann Intern Med 124 859–67 Occurrence Handle1:CAS:528:DyaK28XjtFKisrs%3D Occurrence Handle8610914
J Escourrou P Deprez A Saggioro H Geldof R Fischer C Maier (1999) ArticleTitleMaintenance therapy with pantoprazole 20 mg prevents relapse of reflux oesophagitis Aliment Pharmacol Ther 13 1481–91 Occurrence Handle10.1046/j.1365-2036.1999.00651.x Occurrence Handle1:CAS:528:DyaK1MXnvVKmsLw%3D Occurrence Handle10571605
R Fitzgerald B Onwuegbusi M Bajaj-Elliott I Saeed (2002) ArticleTitleDiversity in the oesophageal phenotypic response to gastro-oesophageal reflux: immunological determinants Gut 50 451–9 Occurrence Handle10.1136/gut.50.4.451 Occurrence Handle1:STN:280:DC%2BD387ot1yrsQ%3D%3D Occurrence Handle11889061
H Isomoto A Wang Y Mizuta Y Akazawa (2003) ArticleTitleElevated levels of chemokines in esophageal mucosa of patients with reflux esophagitis Am J Gastroenterol 98 551–6 Occurrence Handle10.1111/j.1572-0241.2003.07303.x Occurrence Handle1:CAS:528:DC%2BD3sXjtVyltbo%3D Occurrence Handle12650786
N Yoshida K Uchiyama M Kuroda K Sakuma S Kokura H Ichikawa et al. (2004) ArticleTitleInterleukin-8 expression in the esophageal mucosa of patients with gastroesophageal reflux disease Scand J Gastroenterol 39 816–22 Occurrence Handle10.1080/00365520410006729 Occurrence Handle1:CAS:528:DC%2BD2cXnvV2qtLo%3D Occurrence Handle15513378
WK Kauer JH Peters TR DeMeester AP Ireland CG Bremner JA Hagen (1995) ArticleTitleMixed reflux of gastric and duodenal juices is more harmful to the esophagus than gastric juice alone. The need for surgical therapy re-emphasized Ann Surg 222 525–31 Occurrence Handle1:STN:280:DyaK28%2FisFWhsQ%3D%3D Occurrence Handle7574932 Occurrence Handle10.1097/00000658-199522240-00010
P Gillen P Keeling PJ Byrne M Healy RR O’Moore TP Hennessy (1988) ArticleTitleImplication of duodenogastric reflux in the pathogenesis of Barrett's oesophagus Br J Surg 75 540–3 Occurrence Handle1:STN:280:DyaL1c3otFyksA%3D%3D Occurrence Handle3395818
HJ Mud SE Kranendonk H Obertop H Van Houten DL Westbroek (1982) ArticleTitleActive trypsin and reflux oesophagitis: an experimental study in rats Br J Surg 69 269–72 Occurrence Handle1:STN:280:DyaL387otlCgsQ%3D%3D Occurrence Handle6803865
JA Salo E Kivilaakso (1984) ArticleTitleContribution of trypsin and cholate to the pathogenesis of experimental alkaline reflux esophagitis Scand J Gastroenterol 19 875–81 Occurrence Handle1:CAS:528:DyaL2MXit1eisw%3D%3D Occurrence Handle6531656
LF Johnson JW Harmon (1986) ArticleTitleExperimental esophagitis in a rabbit model. Clinical relevance J Clin Gastroenterol 8 Suppl 1 26–44 Occurrence Handle1:STN:280:DyaL283nvFCltA%3D%3D Occurrence Handle3090135 Occurrence Handle10.1097/00004836-198606001-00006
N Asokananthan PT Graham J Fink DA Knight AJ Bakker AS McWilliam et al. (2002) ArticleTitleActivation of protease-activated receptor (PAR)-1, PAR-2, and PAR-4 stimulates IL-6, IL-8, and prostaglandin E2 release from human respiratory epithelial cells J Immunol 168 3577–85 Occurrence Handle1:CAS:528:DC%2BD38Xis1ajtbo%3D Occurrence Handle11907122
A Kawabe Y Shimada T Soma M Maeda A Itami J Kaganoi et al. (2004) ArticleTitleProduction of prostaglandin E2 via bile acid is enhanced by trypsin and acid in normal human esophageal epithelial cells Life Sci 75 21–34 Occurrence Handle10.1016/j.lfs.2003.11.022 Occurrence Handle1:CAS:528:DC%2BD2cXjsVWrtLk%3D Occurrence Handle15102519
Y Tamura M Hirado K Okamura Y Minato S Fujii (1977) ArticleTitleSynthetic inhibitors of trypsin, plasmin, kallikrein, thrombin, C1r-, and C1 esterase Biochim Biophys Acta 484 417–22 Occurrence Handle1:CAS:528:DyaE2sXlvFKiu7g%3D Occurrence Handle143965
T Imada C Chen S Hatori M Shiozawa Y Rino (1999) ArticleTitleEffect of trypsin inhibitor on reflux oesophagitis after total gastrectomy in rats Eur J Surg 165 1045–50 Occurrence Handle10.1080/110241599750007874 Occurrence Handle1:STN:280:DC%2BD3c%2FmtFSmtQ%3D%3D Occurrence Handle10595608
I Sasaki (1989) ArticleTitleEffect of camostat in the treatment of reflux esophagitis after gastrectomy: an experimental study in rats and a pilot clinical study Biomed Res 10 167–73 Occurrence Handle1:CAS:528:DyaK3cXkvVGktg%3D%3D
TY Oh JS Lee BO Ahn H Cho WB Kim YB Kim et al. (2001) ArticleTitleOxidative damages are critical in pathogenesis of reflux esophagitis: implication of antioxidants in its treatment Free Radic Biol Med 30 905–15 Occurrence Handle10.1016/S0891-5849(01)00472-5 Occurrence Handle1:CAS:528:DC%2BD3MXisF2qu7w%3D Occurrence Handle11295533
TY Oh JS Lee BO Ahn H Cho WB Kim YB Kim et al. (2001) ArticleTitleOxidative stress is more important than acid in the pathogenesis of reflux oesophagitis in rats Gut 49 364–71 Occurrence Handle10.1136/gut.49.3.364 Occurrence Handle1:CAS:528:DC%2BD3MXnt1Cqur0%3D Occurrence Handle11511558
F Ismail-Beigi PF Horton CE Pope Suffix2nd (1970) ArticleTitleHistological consequences of gastroesophageal reflux in man Gastroenterology 58 163–74 Occurrence Handle1:STN:280:DyaE3c7gsFWnug%3D%3D Occurrence Handle5413015
P Chomczynski N Sacchi (1987) ArticleTitleSingle-step method of RNA isolation by acid guanidinium thiocyanate–phenol–chloroform extraction Anal Biochem 162 156–9 Occurrence Handle10.1016/0003-2697(87)90021-2 Occurrence Handle1:CAS:528:DyaL2sXitFSns7Y%3D Occurrence Handle2440339
S Kawabata T Miura T Morita H Kato K Fujikawa S Iwanaga et al. (1988) ArticleTitleHighly sensitive peptide-4-methylcoumaryl-7-amide substrates for blood-clotting proteases and trypsin Eur J Biochem 172 17–25 Occurrence Handle10.1111/j.1432-1033.1988.tb13849.x Occurrence Handle1:CAS:528:DyaL1cXhs1ertbs%3D Occurrence Handle3278905
F Zhang NK Altorki YC Wu RA Soslow K Subbaramaiah AJ Dannenberg (2001) ArticleTitleDuodenal reflux induces cyclooxygenase-2 in the esophageal mucosa of rats: evidence for involvement of bile acids Gastroenterology 121 1391–9 Occurrence Handle10.1053/gast.2001.29781 Occurrence Handle1:CAS:528:DC%2BD38Xhs12huw%3D%3D Occurrence Handle11729118
T Kawai H Ikeda Y Harada T Saitou (1992) ArticleTitleChanges in the rat stomach after long-term administration of proton pump inhibitors (AG-1749 and E-3810) Nippon Rinsho 50 188–93 Occurrence Handle1:STN:280:DyaK387nslGntA%3D%3D Occurrence Handle1311785
TM Lin DC Evans MW Warrick RP Pioch (1986) ArticleTitleActions of nizatidine, a selective histamine H2-receptor antagonist, on gastric acid secretion in dogs, rats and frogs J Pharmacol Exp Ther 239 406–10 Occurrence Handle1:CAS:528:DyaL2sXislKnsw%3D%3D Occurrence Handle2877081
Y Ito S Nakamura Y Onoda Y Sugawara O Takaiti (1993) ArticleTitleEffects of the new anti-ulcer drug ecabet sodium (TA-2711) on pepsin activity. I. Inactivation of enzyme protein Jpn J Pharmacol 62 169–74 Occurrence Handle1:CAS:528:DyaK3sXltlWhtbw%3D Occurrence Handle8371515
M Hamaguchi Y Fujiwara T Takashima T Hayakawa E Sasaki M Shiba et al. (2003) ArticleTitleIncreased expression of cytokines and adhesion molecules in rat chronic esophagitis Digestion 68 189–97 Occurrence Handle10.1159/000075698 Occurrence Handle1:CAS:528:DC%2BD2cXhtlygu7o%3D Occurrence Handle14691346
N Omura H Kashiwagi G Chen F Yano Y Suzuki T Aoki (2000) ArticleTitleEffects of ecabet sodium on experimentally induced reflux esophagitis J Gastroenterol 35 504–9 Occurrence Handle10.1007/s005350070072 Occurrence Handle1:CAS:528:DC%2BD3cXltlygt7s%3D Occurrence Handle10905357
SK Bohm LM Khitin EF Grady G Aponte DG Payan NW Bunnett (1996) ArticleTitleMechanisms of desensitization and resensitization of proteinase-activated receptor-2 J Biol Chem 271 22003–16 Occurrence Handle10.1074/jbc.271.36.22003 Occurrence Handle1:CAS:528:DyaK28XlsFKns7k%3D Occurrence Handle8703006
N Cenac AM Coelho C Nguyen S Compton P Andrade-Gordon WK MacNaughton et al. (2002) ArticleTitleInduction of intestinal inflammation in mouse by activation of proteinase-activated receptor-2 Am J Pathol 161 1903–15 Occurrence Handle1:CAS:528:DC%2BD38XptV2gtbc%3D Occurrence Handle12414536
TD Nguyen MW Moody M Steinhoff C Okolo DS Koh NW Bunnett (1999) ArticleTitleTrypsin activates pancreatic duct epithelial cell ion channels through proteinase-activated receptor-2 J Clin Invest 103 261–9 Occurrence Handle1:CAS:528:DyaK1MXltFCjsg%3D%3D Occurrence Handle9916138 Occurrence Handle10.1172/JCI2539
T Olejar R Matej M Zadinova P Pouckova (2001) ArticleTitleExpression of proteinase-activated receptor 2 during taurocholate-induced acute pancreatic lesion development in Wistar rats Int J Gastrointest Cancer 30 113–21 Occurrence Handle10.1385/IJGC:30:3:113 Occurrence Handle1:CAS:528:DC%2BD3sXhtVSqtro%3D Occurrence Handle12540023
N Yoshida K Katada M Kuroda M Shimozawa K Uchiyama Y Naito et al. (2004) ArticleTitleTrypsin activates human esophageal epithelial cells to produce inflammatory cytokines through protease-activated receptor 2 Gastroenterology 126 A-501
K Watanabe M Iida K Takaishi T Suzuki Y Hamada Y Iizuka et al. (1993) ArticleTitleChemoattractants for neutrophils in lipopolysaccharide-induced inflammatory exudate from rats are not interleukin-8 counterparts but gro-gene-product/melanoma-growth-stimulating-activity-related factors Eur J Biochem 214 267–70 Occurrence Handle10.1111/j.1432-1033.1993.tb17920.x Occurrence Handle1:CAS:528:DyaK3sXlslyqtLs%3D Occurrence Handle8508797
H Suzuki M Suematsu S Miura YY Liu K Watanabe M Miyasaka et al. (1994) ArticleTitleRat CINC/gro: a novel mediator for locomotive and secretagogue activation of neutrophils in vivo J Leukoc Biol 55 652–7 Occurrence Handle1:CAS:528:DyaK2cXktlarsb0%3D Occurrence Handle8182343
E Imamoto N Yoshida K Uchiyama H Higashihara N Tomatsuri M Ueda et al. (2003) ArticleTitleEffects of bile acids and acidic exposure on IL-8 expression in human esophageal epithelial cells Gastroenterology 124 A-409
Y Naito T Yoshikawa (2002) ArticleTitleMolecular and cellular mechanisms involved in Helicobacter pylori-induced inflammation and oxidative stress Free Radic Biol Med 33 323–36 Occurrence Handle10.1016/S0891-5849(02)00868-7 Occurrence Handle1:CAS:528:DC%2BD38Xlt12kur4%3D Occurrence Handle12126754
KT Wilson S Fu KS Ramanujam SJ Meltzer (1998) ArticleTitleIncreased expression of inducible nitric oxide synthase and cyclooxygenase-2 in Barrett's esophagus and associated adenocarcinomas Cancer Res 58 2929–34 Occurrence Handle1:CAS:528:DyaK1cXkvVCntLg%3D Occurrence Handle9679948
MJ Thun (2003) ArticleTitleNSAIDs and esophageal cancer: ready for trials but not yet broad clinical application Gastroenterology 124 246–8 Occurrence Handle10.1053/gast.2003.50025 Occurrence Handle12512047
BS Kaur N Khamnehei M Iravani SS Namburu O Lin G Triadafilopoulos (2002) ArticleTitleRofecoxib inhibits cyclooxygenase 2 expression and activity and reduces cell proliferation in Barrett's esophagus Gastroenterology 123 60–7 Occurrence Handle10.1053/gast.2002.34244 Occurrence Handle1:CAS:528:DC%2BD38Xmt1aiu7w%3D Occurrence Handle12105834
RA Houliston RJ Keogh D Sugden J Dudhia TD Carter CP Wheeler-Jones (2002) ArticleTitleProtease-activated receptors upregulate cyclooxygenase-2 expression in human endothelial cells Thromb Haemost 88 321–8 Occurrence Handle1:CAS:528:DC%2BD38XmsVSmsL0%3D Occurrence Handle12195707
MB Frungieri S Weidinger V Meineke FM Kohn A Mayerhofer (2002) ArticleTitleProliferative action of mast-cell tryptase is mediated by PAR2, COX2, prostaglandins, and PPARgamma: possible relevance to human fibrotic disorders Proc Natl Acad Sci U S A 99 15072–7 Occurrence Handle10.1073/pnas.232422999 Occurrence Handle1:CAS:528:DC%2BD38Xpt1yru78%3D Occurrence Handle12397176
K Yada K Shibata T Matsumoto M Ohta S Yokoyama S Kitano (2005) ArticleTitleProtease-activated receptor-2 regulates cell proliferation and enhances cyclooxygenase-2 mRNA expression in human pancreatic cancer cells J Surg Oncol 89 79–85 Occurrence Handle10.1002/jso.20197 Occurrence Handle1:CAS:528:DC%2BD2MXitVKgurc%3D Occurrence Handle15660373
D Nehra P Howell CP Williams JK Pye J Beynon (1999) ArticleTitleToxic bile acids in gastro-oesophageal reflux disease: influence of gastric acidity Gut 44 598–602 Occurrence Handle1:STN:280:DyaK1M3itFKksQ%3D%3D Occurrence Handle10205192 Occurrence Handle10.1136/gut.44.5.598
DC Gotley AP Morgan D Ball RW Owen MJ Cooper (1991) ArticleTitleComposition of gastro-oesophageal refluxate Gut 32 1093–9 Occurrence Handle1:STN:280:DyaK38%2FmsVansA%3D%3D Occurrence Handle1955160
JA Salmo VP Lehto HS Myllarniemi EO Kivilaakso (1990) ArticleTitleMorphological alterations in tryptic esophagitis: an experimental light microscopic and scanning and transmission electron microscopic study in rabbits J Surg Res 49 14–7 Occurrence Handle10.1016/0022-4804(90)90103-9 Occurrence Handle1:STN:280:DyaK3c3psVKitA%3D%3D Occurrence Handle2359288
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Naito, Y., Uchiyama, K., Kuroda, M. et al. Role of pancreatic trypsin in chronic esophagitis induced by gastroduodenal reflux in rats. J Gastroenterol 41, 198–208 (2006). https://doi.org/10.1007/s00535-005-1742-5
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/s00535-005-1742-5