Abstract
Purpose
To describe the management and outcome of critically-ill patients with Cyclophosphamide (CY)-associated cardiac toxicity.
Methods
All patients admitted to the intensive care units (ICUs) of the Nantes and Rennes University Hospitals for a CY-associated cardiac toxicity between January 2015 and December 2020 were included.
Results
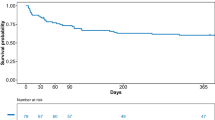
Of the thirty-four patients included in the study, twenty-four (70%) underwent allogeneic hematopoietic stem cell transplantation (HSCT), four (12%) autologous HSCT, and six (18%) chemotherapy for hematological malignancies. Acute pulmonary edema (65%), cardiac arrest (9%), and cardiac arrhythmia (6%) were the most common reasons for ICU admission. Patients were admitted to the ICU 6.5 (4-12) days after the intravenous administration of a median dose of CY of 100 [60-101] mg/Kg. Echocardiographic findings showed moderate to severe left ventricular systolic dysfunction (69%) and pericardial effusion (52%). Eighteen (53%) patients ultimately developed cardiogenic shock and required vasopressors (47%) and/or inotropes (18%). Invasive mechanical ventilation and renal replacement therapy were required in twenty (59%) and five (14%) patients, respectively. Sixteen (47%) patients died of whom 12 (35.3%) died from refractory cardiogenic shock. The left ventricular ejection fraction improved over time in most survivors with a median time until full recovery of 33 (12-62) days. Two (11%) patients had a persistent left ventricular dysfunction at 6 months.
Conclusion
Refractory cardiogenic shock is the primary cause of death of patients with severe CY-related cardiotoxicity. Nonetheless, the cardiac function of most survivors recovered within a month.



Similar content being viewed by others
References
Jones DS, Podolsky SH, Greene JA (2012) The Burden of Disease and the Changing Task of Medicine. N Engl J Med 366:2333–2338. https://doi.org/10.1056/NEJMp1113569
Ferlay J, Shin H-R, Bray F et al (2010) Estimates of worldwide burden of cancer in 2008: GLOBOCAN 2008. International Journal of Cancer 127:2893–2917. https://doi.org/10.1002/ijc.25516
Dizon DS, Krilov L, Cohen E et al (2016) Clinical Cancer Advances 2016: Annual Report on Progress Against Cancer From the American Society of Clinical Oncology. J Clin Oncol 34:987–1011. https://doi.org/10.1200/JCO.2015.65.8427
Récher C, Coiffier B, Haioun C et al (2011) Intensified chemotherapy with ACVBP plus rituximab versus standard CHOP plus rituximab for the treatment of diffuse large B-cell lymphoma (LNH03-2B): an open-label randomised phase 3 trial. Lancet 378:1858–1867. https://doi.org/10.1016/S0140-6736(11)61040-4
Baili P, Di Salvo F, Marcos-Gragera R et al (2015) Age and case mix-standardised survival for all cancer patients in Europe 1999–2007: Results of EUROCARE-5, a population-based study. Eur J Cancer 51:2120–2129. https://doi.org/10.1016/j.ejca.2015.07.025
Zampieri FG, Romano TG, Salluh JIF et al (2021) Trends in clinical profiles, organ support use and outcomes of patients with cancer requiring unplanned ICU admission: a multicenter cohort study. Intensive Care Med 47:170–179. https://doi.org/10.1007/s00134-020-06184-2
Ferreyro BL, Scales DC, Wunsch H et al (2021) Critical illness in patients with hematologic malignancy: a population-based cohort study. Intensive Care Med 47:1104–1114. https://doi.org/10.1007/s00134-021-06502-2
Zamorano JL, Lancellotti P, Rodriguez Muñoz D et al (2016) 2016 ESC Position Paper on cancer treatments and cardiovascular toxicity developed under the auspices of the ESC Committee for Practice Guidelines: The Task Force for cancer treatments and cardiovascular toxicity of the European Society of Cardiology (ESC). Eur Heart J 37:2768–2801. https://doi.org/10.1093/eurheartj/ehw211
Santos GW, Sensenbrenner LL, Burke PJ et al (1972) The use of cyclophosphamide for clinical marrow transplantation. Transplant Proc 4:559–564
Kurauchi K, Nishikawa T, Miyahara E et al (2017) Role of metabolites of cyclophosphamide in cardiotoxicity. BMC Res Notes 10:406. https://doi.org/10.1186/s13104-017-2726-2
Moghe A, Ghare S, Lamoreau B et al (2015) Molecular Mechanisms of Acrolein Toxicity: Relevance to Human Disease. Toxicol Sci 143:242–255. https://doi.org/10.1093/toxsci/kfu233
Nishikawa T, Miyahara E, Kurauchi K et al (2015) Mechanisms of Fatal Cardiotoxicity following High-Dose Cyclophosphamide Therapy and a Method for Its Prevention. PLoS One 10:e0131394. https://doi.org/10.1371/journal.pone.0131394
Gottdiener JS, Appelbaum FR, Ferrans VJ et al (1981) Cardiotoxicity associated with high-dose cyclophosphamide therapy. Arch Intern Med 141:758–763
Goldberg MA, Antin JH, Guinan EC, Rappeport JM (1986) Cyclophosphamide cardiotoxicity: an analysis of dosing as a risk factor. Blood 68:1114–1118
Katayama M, Imai Y, Hashimoto H et al (2009) Fulminant fatal cardiotoxicity following cyclophosphamide therapy. J Cardiol 54:330–334. https://doi.org/10.1016/j.jjcc.2009.01.006
Cazin B, Gorin NC, Laporte JP et al (1986) Cardiac complications after bone marrow transplantation. A report on a series of 63 consecutive transplantations. Cancer 57:2061–2069. https://doi.org/10.1002/1097-0142(19860515)57:10<2061::AID-CNCR2820571031>3.0.CO;2-H
Duléry R, Mohty R, Labopin M et al (2021) Early Cardiac Toxicity Associated With Post-Transplant Cyclophosphamide in Allogeneic Stem Cell Transplantation. JACC: CardioOncology 3:250–259. https://doi.org/10.1016/j.jaccao.2021.02.011
Ishida S, Doki N, Shingai N et al (2016) The clinical features of fatal cyclophosphamide-induced cardiotoxicity in a conditioning regimen for allogeneic hematopoietic stem cell transplantation (allo-HSCT). Ann Hematol 95:1145–1150. https://doi.org/10.1007/s00277-016-2654-6
Appelbaum F, Strauchen JA, Graw RG et al (1976) Acute lethal carditis caused by high-dose combination chemotherapy. A unique clinical and pathological entity. Lancet 1:58–62. https://doi.org/10.1016/s0140-6736(76)90151-3
Pai VB, Nahata MC (2000) Cardiotoxicity of Chemotherapeutic Agents: Incidence, Treatment and Prevention. Drug Safety 22:263–302. https://doi.org/10.2165/00002018-200022040-00002
Yeh ETH, Bickford CL (2009) Cardiovascular Complications of Cancer Therapy. J Am Coll Cardiol 53:2231–2247. https://doi.org/10.1016/j.jacc.2009.02.050
Mills BA, Roberts RW (1979) Cyclophosphamide-induced cardiomyopathy. A report of two cases and review of the english literature. Cancer 43:2223–2226. https://doi.org/10.1002/1097-0142(197906)43:6<2223::AID-CNCR2820430610>3.0.CO;2-Y
Luznik L, O’Donnell PV, Symons HJ et al (2008) HLA-haploidentical bone marrow transplantation for hematologic malignancies using nonmyeloablative conditioning and high-dose, posttransplantation cyclophosphamide. Biol Blood Marrow Transplant 14:641–650. https://doi.org/10.1016/j.bbmt.2008.03.005
Kamphuis JAM, Linschoten M, Cramer MJ et al (2019) Cancer Therapy-Related Cardiac Dysfunction of Nonanthracycline Chemotherapeutics. JACC: CardioOncology 1:280–290. https://doi.org/10.1016/j.jaccao.2019.09.007
Morandi P, Ruffini PA, Benvenuto GM et al (2005) Cardiac toxicity of high-dose chemotherapy. Bone Marrow Transplant 35:323–334. https://doi.org/10.1038/sj.bmt.1704763
Blazoski C, Kirupaharan P, Hilton R et al (2023) Outcomes of extracorporeal membrane oxygenation in immunosuppressed vs Immunocompetent patients. Heart Lung 58:179–184. https://doi.org/10.1016/j.hrtlng.2022.12.003
Schmidt M, Schellongowski P, Patroniti N et al (2018) Six-Month Outcome of Immunocompromised Patients with Severe Acute Respiratory Distress Syndrome Rescued by Extracorporeal Membrane Oxygenation. An International Multicenter Retrospective Study. Am J Respir Crit Care Med 197:1297–1307. https://doi.org/10.1164/rccm.201708-1761OC
Wohlfarth P, Ullrich R, Staudinger T et al (2014) Extracorporeal membrane oxygenation in adult patients with hematologic malignancies and severe acute respiratory failure. Crit Care 18:R20. https://doi.org/10.1186/cc13701
Acknowledgments
We thank Julien Cadiet for helping in data collection.
Financial support
None.
Funding
The authors declare that no funds, grants, or other support were received during the preparation of this manuscript.
Author information
Authors and Affiliations
Contributions
AV was the principal investigator and takes primary responsibility for the paper. JMT, TLT, JNT, CT, FR, RH, AS, JR, JBL, recruited the patients. AV, VG and JL did the statistical analysis. AV, JL and EC co-ordinated the research and wrote the manuscript.
Corresponding author
Ethics declarations
Ethics approval
This study was approved by the ethics committee of the French Intensive Care Society (CE SRLF 22-058) on December 18, 2022.
Consent to participate
In accordance with French law on retrospective studies of anonymized healthcare data, informed consent was not required.
Competing interests
EC has received lecturer and conference-speaker fees, as well as reimbursements of travel and accommodation expenses related to attending scientific meetings, from Gilead, Shionogi, and Sanofi-Genzyme. JBL has received lecturer and conference-speaker fees from BD and Zoll. RH reports honoraria from Kite/Gilead, Novartis, Incyte, Janssen, MSD, Takeda and Roche; and consultancy at Kite/Gilead, Novartis, Bristol-Myers Squibb/Celgene, ADC Therapeutics, Incyte, Miltenyi. None of the other authors have relevant financial or non-financial interests to disclose.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Vennier, A., Canet, E., Guardiolle, V. et al. Clinical features and outcomes of patients admitted to the ICU for Cyclophosphamide-associated cardiac toxicity: a retrospective cohort. Support Care Cancer 31, 474 (2023). https://doi.org/10.1007/s00520-023-07951-9
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00520-023-07951-9