Abstract
Purpose
Head and neck cancer (HNCa) presents numerous challenges secondary to treatment. While there is substantial clinical awareness to the range of challenges demonstrated in this population, information on the impact of post-treatment fatigue is limited. This study investigated the degree of perceived fatigue in those treated for HNCa.
Methods
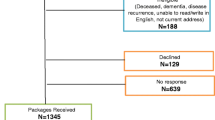
The study was a cross-sectional, self-report, survey design. Adult participants (n = 47) completed a series of three questionnaires; two validated fatigue measures – the Fatigue Screening Inventory (FSI) and the Multidimensional Fatigue Inventory (MFI-20) and a general health-related quality of life measure the European Organisation of Research on the Treatment of Cancer – Quality of Life Questionnaire (EORTC-QLQC30) and the head and neck site specific module (QLQ - H&N 35) were administered.
Results
Of the 47 participants, more than half (55%) were identified as having clinically significant self-reported levels of fatigue. Correlational analysis revealed an inverse relationship between fatigue and overall health-related quality of life (HRQOL) implying that as fatigue increases, one’s perceived HRQOL decreases.
Conclusions
These data suggest that efforts to proactively screen for and index fatigue and seek anticipatory interventions may benefit both short- and long-term HRQOL outcomes in those diagnosed with HNCa.
Level of Evidence: IV.

Similar content being viewed by others
References
Weis J, Horneber M (2014) Cancer-Related Fatigue. Springer, Tarporley. https://doi.org/10.1007/978-1-907673-76-4
Curt GA, Breitbart W, Cella D et al (2000) Impact of cancer-related fatigue on the lives of patients: new findings from the Fatigue Coalition. Oncologist 5(5):353–360
Stone P, Richards M, Hardy J (1998) Fatigue in patients with cancer. Eur J Cancer 34(11):1670–1676
Winningham ML, Nail LM, Burke MB et al (1994) Fatigue and the cancer experience: the state of the knowledge. Oncol Nurs Forum 21(1):23–36
Visser MRM, Smets EMA (1998) Fatigue, depression and quality of life in cancer patients: how are they related? Support Care Cancer 6(2):101–108. https://doi.org/10.1007/s005200050142
Stasi R, Abriani L, Beccaglia P, Terzoli E, Amadori S (2003) Cancer-related fatigue. Cancer 98(9):1786–1801. https://doi.org/10.1002/cncr.11742
Medical Subject Headings - Fatigue. Retrieved January 10, 2023 from https://bioportal.bioontology.org/ontologies/MESH?p=classes&conceptid=D015673
National Comprehensive Cancer Network (2003) National Comprehensive Cancer Network practice guidelines cancer-related fatigue panel 2003 guidelines. National Comprehensive Cancer Network, Rockledge
Mock V, Atkinson A, Barsevick A et al (2000) NCCN Practice Guidelines for Cancer-Related Fatigue. Oncology (Williston Park) 14(11A):151–161
Siegel R, DeSantis C, Virgo K, Stein K, Mariotto A, Smith T et al (2012) Cancer treatment and survivorship statistics, 2012. Cancer J Clin 62(4):220–241. https://doi.org/10.3322/caac.21149
Vogelzang NJ, Breitbart W, Cella D et al (1997) Patient, caregiver, and oncologist perceptions of cancer-related fatigue: results of a tripart assessment survey. The Fatigue Coalition. Semin Hematol 34(3 Suppl 2):4–12
Bornbaum CC, Fung K, Franklin JH, Nichols A, Yoo J, Doyle PC (2012) A descriptive analysis of the relationship between quality of life and distress in individuals with head and neck cancer. Support Care Cancer 20(9):2157–2165. https://doi.org/10.1007/s00520-011-1326-2
Langenberg SM, van Herpen CM, van Opstal C, Wymenga AN, van der Graaf WT, Prins JB (2019) Caregivers’ burden and fatigue during and after patients’ treatment with concomitant chemoradiotherapy for locally advanced head and neck cancer: a prospective, observational pilot study. Support Care Cancer 27(11):4145–4154
Cella D, Peterman A, Passik S, Jacobsen P, Breitbart W (1998) Progress toward guidelines for the management of fatigue. Oncology (Williston Park) 12(11A):369–377
Ackerstaff AH, Hilgers FJ, Aaronson NK, Balm AJ (1994) Communication, functional disorders and lifestyle changes after total laryngectomy. Clin Otolaryngol Allied Sci 19(4):295–300
Rosenthal DI, Mendoza TR, Chambers MS et al (2007) Measuring head and neck cancer symptom burden: The development and validation of the M. D. Anderson symptom inventory, head and neck module. Head Neck 29(10):923–931. https://doi.org/10.1002/hed.20602
Hammermüller C, Hinz A, Dietz A, Wichmann G, Pirlich M, Berger T, Zimmermann K, Neumuth T, Mehnert-Theuerkauf A, Wiegand S, Zebralla V (2021) Depression, anxiety, fatigue, and quality of life in a large sample of patients suffering from head and neck cancer in comparison with the general population. BMC Cancer 21(1):1–1
Milas ZL, Neelands B, Trufan SJ, Benbow J, Carrizosa D, Brickman DS, Frenkel CH, Sumrall AL, Sudhakar SM, Ward MC (2022) Survivorship Care Plans Improve the Identification of Post-radiation Hypothyroidism After Head and Neck Cancer Treatment. Anticancer Res 42(9):4429–4437
Reyes-Gibby CC, Anderson KO, Merriman KW, Todd KH, Shete SS, Hanna EY (2014) Survival patterns in squamous cell carcinoma of the head and neck: pain as an independent prognostic factor for survival. J Pain 15(10):1015–1022
Dolgoy ND, O’Krafka P, McNeely ML (2020) Cancer-related fatigue in head and neck cancer survivors: Energy and functional impacts. Cancer Treat Res Commun 25:100244
Hann DM, Jacobsen PB, Azzarello LM et al (1998) Measurement of fatigue in cancer patients: development and validation of the Fatigue Symptom Inventory. Qual Life Res 7(4):301–310
Smets EM, Garssen B, Bonke B, de Haes JC (1995) The Multidimensional Fatigue Inventory (MFI) psychometric qualities of an instrument to assess fatigue. J Psychosom Res 39(3):315–325
Aaronson NK, Ahmedzai S, Bergman B et al (1993) The European Organization for Research and Treatment of Cancer QLQ-C30: a quality-of-life instrument for use in international clinical trials in oncology. J Natl Cancer Inst 85(5):365–376
Donovan KA, Jacobsen PB, Small BJ, Munster PN, Andrykowski MA (2008) Identifying clinically meaningful fatigue with the Fatigue Symptom Inventory. J Pain Symptom Manage 36(5):480–487. https://doi.org/10.1016/j.jpainsymman.2007.11.013
Bjordal K, Kaasa S (2009) Psychometric Validation of the EORTC Core Quality of Life Questionnaire, 30-Item Version and A iagnosis-Specific Module for Head and Neck Cancer Patients. Acta Oncol 31(3):311–320. https://doi.org/10.3109/02841869209108178
King MT, Dobson AJ, Harnett PR (1996) A comparison of two quality-of-life questionnaires for cancer clinical trials: the functional living index–cancer (FLIC) and the quality of life questionnaire core module (QLQ-C30). J Clin Epidemiol 49(1):21–29
Meier CA, Braverman LE, Ebner SA, Veronikis IRINI, Daniels GH, Ross DS, DeGroot LJ (1994) Diagnostic use of recombinant human thyrotropin in patients with thyroid carcinoma (phase I/II study). J Clin Endocrinol Metabol 78(1):188–196
Canadian Cancer Society (2014) Canadian Cancer Society Statistics 2014. Author, Toronto
Hofman M, Ryan JL, Figueroa-Moseley CD, Jean-Pierre P, Morrow GR (2007) Cancer-related fatigue: The scale of the problem. Oncologist 12(Supplement 1):4–10
Petruson KM, Silander EM, Hammerlid EB (2005) Quality of life as predictor of weight loss in patients with head and neck cancer. Head Neck 27(4):302–310. https://doi.org/10.1002/hed.20172
Rogers LQ, Courneya KS, Robbins KT, Rao K, Malone J, Seiz A, Burra V (2008) Factors associated with fatigue, sleep, and cognitive function among patients with head and neck cancer. Head Neck 30(10):1310–1317
Bossi P, Di Pede P, Guglielmo M, Granata R, Alfieri S, Iacovelli NA, Orlandi E, Guzzo M, Bianchi R, Ferella L, Infante G (2019) Prevalence of fatigue in head and neck cancer survivors. Ann Otol Rhinol Laryngol 128(5):413–419
Portney LG, Watkins MP (2000) Foundations of clinical research: applications to practice. Pearson/Prentice Hall, Upper Saddle River, NJ
Acknowledgements
Portions of this work were completed while the senior authors (G.M.S. and P.C.D.) were affiliated with the Laboratory for Well-Being and Quality of Life at the University of Western Ontario.
Author information
Authors and Affiliations
Contributions
Conceptualization was undertaken by G.M.S. and P.C.D.; all authors contributed to the design of the study, as well as data collection and its analysis. The first draft of the manuscript was written by G.M.S. and P.C.D. and all authors read and provided comments on earlier drafts of the manuscript. All authors have read and approved of the final manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethics approval
This study was reviewed and approved by the Research Ethics Board of the University of Western Ontario.
Consent to participate
Informed consent was obtained from all study participants prior to their agreement to participate in this research. All participants were informed about the purpose of this research project and that their agreement for participation was fully voluntary.
Consent for publication
All authors confirm that ethical approval confirmed that all participants provided informed consent relative to the publication of the data obtained as part of this research.
Conflict of interest
None of the authors have any relevant financial or non-financial interests to disclose.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Grace M. Spiro and Philip C. Doyle - first authorship is shared equally
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Spiro, G.M., Doyle, P.C., Jovanovic, N. et al. Self-perception of fatigue in individuals diagnosed with head and neck cancer. Support Care Cancer 31, 465 (2023). https://doi.org/10.1007/s00520-023-07936-8
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00520-023-07936-8