Abstract
Objectives
Implementation of guideline-recommended depression screening in oncology presents numerous challenges. Implementation strategies that are responsive to local context may be critical elements of adoption and sustainment. We evaluated barriers and facilitators to implementation of a depression screening program for breast cancer patients in a community medical oncology setting as part of a cluster randomized controlled trial.
Methods
Guided by the Consolidated Framework for Implementation Research, we employed qualitative methods to evaluate clinician, administrator, and patient perceptions of the program using semi-structured interviews. We used a team-coding approach for the data; thematic development focused on barriers and facilitators to implementation using a grounded theory approach. The codebook was refined through open discussions of subjectivity and unintentional bias, coding, and memo applications (including emergent coding), and the hierarchical structure and relationships of themes.
Results
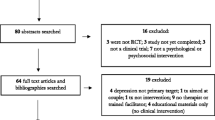
We conducted 20 interviews with 11 clinicians/administrators and 9 patients. Five major themes emerged: (1) gradual acceptance and support of the intervention and workflow; (2) compatibility with system and personal norms and goals; (3) reinforcement of the value of and need for adaptability; (4) self-efficacy within the nursing team; and (5) importance of identifying accountable front-line staff beyond leadership “champions.”
Conclusions
Findings suggest a high degree of acceptability and feasibility due to the selection of appropriate implementation strategies, alignment of norms and goals, and a high degree of workflow adaptability. These findings will be uniquely helpful in generating actionable, real-world knowledge to inform the design, implementation, and sustainment of guideline-recommended depression screening programs in oncology.
Trial Registration
ClinicalTrials.gov #NCT02941614
Similar content being viewed by others
References
Dudgeon D, King S, Howell D, Green E, Gilbert J, Hughes E et al (2012) Cancer Care Ontario's experience with implementation of routine physical and psychological symptom distress screening. Psychooncology 21(4):357–364
Adler N, Page A (2008) Cancer care for the whole patient: meeting psychosocial health needs. National Acadamies Press, Washington, DC
Suppli NP, Johansen C, Christensen J, Kessing LV, Kroman N, Dalton SO (2014) Increased risk for depression after breast cancer: a nationwide population-based cohort study of associated factors in Denmark, 1998-2011. J Clin Oncol 32(34):3831–3839
Pilevarzadeh MAM, Afsargharehbagh R, Rafiemanesh H, Hashemi SM, Balouchi A (2019) Global prevalence of depression among breast cancer patients: a systematic review and meta-analysis. Breast Cancer Res Treat 176(3):519–533
Stanton AL (2006) Psychosocial concerns and interventions for cancer survivors. J Clin Oncol 24(32):5132–5137
Bultz BD, Watson L, Loscalzo M, Kelly B, Zabora J (2021) From foundation to inspiration: implementing screening for distress (6th Vital Sign) for optimal cancer care—international leadership perspectives on program development. J Psychosoc Oncol Res Pract 3(2):e051
Riba MB, Donovan KA, Andersen B, Braun I, Breitbart WS, Brewer BW et al (2019) Distress management, Version 3.2019, NCCN Clinical Practice Guidelines in Oncology. J Natl Compr Canc Netw 17(10):1229–1249
American College of Surgeons Commission on Cancer. Cancer Program Standards: Ensuring Patient-Centered Care Manual (2016 Edition). Chicago, IL. Standard 3.2 - Psychosocial Distress Screening, pp.56–57. Available from: https://www.facs.org/quality-programs/cancer/coc/standards. Accessed 24 Apr 2023
Andersen BL, Rowland JH, Somerfield MR (2014) Guideline summary: screening, assessment, and care of anxiety and depressive symptoms in adults with cancer: an American Society of Clinical Oncology Guideline Adaptation. J Oncol Pract 32(15):1605–1619
Ehlers SL, Davis K, Bluethmann SM, Quintiliani LM, Kendall J, Ratwani RM et al (2019) Screening for psychosocial distress among patients with cancer: implications for clinical practice, healthcare policy, and dissemination to enhance cancer survivorship. Transl Behav Med 9(2):282–291
Mitchell AJ, Lord K, Slattery J, Grainger L, Symonds P (2012) How feasible is implementation of distress screening by cancer clinicians in routine clinical care? Cancer. 118(24):6260–6269
Jacobsen PB, Norton WE (2019) The role of implementation science in improving distress assessment and management in oncology: a commentary on "Screening for psychosocial distress among patients with cancer: implications for clinical practice, healthcare policy, and dissemination to enhance cancer survivorship". Transl Behav Med 9(2):292–295
McCarter K, Fradgley EA, Britton B, Tait J, Paul C (2020) Not seeing the forest for the trees: a systematic review of comprehensive distress management programs and implementation strategies. Curr Opin Support Palliat Care 14(3):220–231
Curran GM, Bauer M, Mittman B, Pyne JM, Stetler C (2012) Effectiveness-implementation hybrid designs: combining elements of clinical effectiveness and implementation research to enhance public health impact. Medical care 50(3):217
Loudon K, Treweek S, Sullivan F, Donnan P, Thorpe KE, Zwarenstein M (2015) The PRECIS-2 tool: designing trials that are fit for purpose. BMJ 350:h2147
Thorpe KE, Zwarenstein M, Oxman AD, Treweek S, Furberg CD, Altman DG et al (2009) A pragmatic-explanatory continuum indicator summary (PRECIS): a tool to help trial designers. J Clin Epidemiol 62(5):464–475
Pirl WF, Fann JR, Greer JA, Braun I, Deshields T, Fulcher C et al (2014) Recommendations for the implementation of distress screening programs in cancer centers: report from the American Psychosocial Oncology Society (APOS), Association of Oncology Social Work (AOSW), and Oncology Nursing Society (ONS) joint task force. Cancer 120(19):2946–2954
Smith AB, Rush R, Wright P, Stark D, Velikova G, Sharpe M (2009) Validation of an item bank for detecting and assessing psychological distress in cancer patients. Psychooncology 18(2):195–199
Mitchell AJ (2010) Short screening tools for cancer-related distress: a review and diagnostic validity meta-analysis. J Natl Compr Cancer Netw 8(4):487–494
Ivers NM, Grimshaw JM, Jamtvedt G, Flottorp S, O'Brien MA, French SD et al (2014) Growing literature, stagnant science? Systematic review, meta-regression and cumulative analysis of audit and feedback interventions in health care. J Gen Intern Med 29(11):1534–1541
Berta W, Cranley L, Dearing JW, Dogherty EJ, Squires JE, Estabrooks CA (2015) Why (we think) facilitation works: insights from organizational learning theory. Implement Sci 10(1):141
Hahn EE, Munoz-Plaza CE, Pounds D, Lyons LJ, Lee JS, Shen E et al (2022) Effect of a community-based medical oncology depression screening program on behavioral health referrals among patients with breast cancer: a randomized clinical trial. JAMA 327(1):41–49
Damschroder LJ, Aron DC, Keith RE, Kirsh SR, Alexander JA, Lowery JC (2009) Fostering implementation of health services research findings into practice: a consolidated framework for advancing implementation science. Implement Sci 4:50
Damschroder LJ, Hagedorn HJ (2011) A guiding framework and approach for implementation research in substance use disorders treatment. Psychol Addict Behav 25(2):194–205
DeJonckheere M, Vaughn LM (2019) Semistructured interviewing in primary care research: a balance of relationship and rigour. Fam Med Community Health 7(2):e000057
Palinkas LA, Horwitz SM, Green CA, Wisdom JP, Duan N, Hoagwood K (2015) Purposeful sampling for qualitative data collection and analysis in mixed method implementation research. Admin Pol Ment Health 42(5):533–544
Hennink MM, Kaiser BN, Weber MB (2019) What influences saturation? Estimating Sample Sizes in Focus Group Research. Qual Health Res 29(10):1483–1496
Saunders B, Sim J, Kingstone T, Baker S, Waterfield J, Bartlam B et al (2018) Saturation in qualitative research: exploring its conceptualization and operationalization. Qual Quant 52(4):1893–1907
Francis JJ, Johnston M, Robertson C, Glidewell L, Entwistle V, Eccles MP et al (2010) What is an adequate sample size? Operationalising data saturation for theory-based interview studies. Psychol Health 25(10):1229–1245
MacQueen KM, McLellan E, Kay K, Milstein B (1998) Codebook development for team-based qualitative analysis. CAM J 10(2):31–36
Floersch J (2010) Integrating thematic, grounded theory. Qualitative Social Work
Glaser BG (2008) The constant comparative method of qualitative analysis. Grounded Theory Rev 7(3)
Glaser BG, Strauss A (1967) The discovery of grounded theory: strategies for qualitative research. Aldine Publishing Company, Chicago
Strauss A, Corbin J (1997) Grounded theory in practice. Sage Publications, Thousand Oaks, CA, p 280
Knies AK, Jutagir DR, Ercolano E, Pasacreta N, Lazenby M, McCorkle R (2019) Barriers and facilitators to implementing the commission on cancer's distress screening program standard. Palliat Support Care 17(3):253–261
Kayser K, Brydon DM, Moon H, Zebrack B (2020) Institutional capacity to provide psychosocial care in cancer programs: addressing barriers to delivering quality cancer care. Psychooncology 29(12):1995–2002
Rivest J, Desbeaumes Jodoin V, Fitch M, Martineau JT, Levenson JA (2022) Screening for distress in cancer care: how to overcome barriers after unsuccessful implementation? Palliat Support Care 20(1):1–3
Azizoddin DR, Lakin JR, Hauser J, Rynar LZ, Weldon C, Molokie R et al (2020) Meeting the guidelines: implementing a distress screening intervention for veterans with cancer. Psychooncology 29(12):2067–2074
Fradgley EA, Byrnes E, McCarter K, Rankin N, Britton B, Clover K et al (2020) A cross-sectional audit of current practices and areas for improvement of distress screening and management in Australian cancer services: is there a will and a way to improve? Support Care Cancer 28(1):249–259
Donovan KA, Grassi L, Deshields TL, Corbett C, Riba MB (2020) Advancing the science of distress screening and management in cancer care. Epidemiol Psychiatr Sci 29:e85
Smith TG, Beckwitt AE, van de Poll-Franse LV, Bontemps-Jones JE, James TA, McCabe RM et al (2022) Oncology team perspectives on distress screening: a multisite study of a well-established use of patient-reported outcomes for clinical assessment. Supportive Care in Cancer 30(2):1261–1271
Funding
This project was supported by funds provided by The Regents of the University of California, Research Grants Program Office, California Breast Cancer Research Program, Grant Number 22OB-0029. The opinions, findings, and conclusions herein are those of the author and do not necessarily represent those of The Regents of the University of California or any of its programs.
Author information
Authors and Affiliations
Contributions
EEH, CMP, and MKG generated the idea for the study and designed the study. CMP and EEH led the drafting of the manuscript. CMP, DP, and LJL prepared approval applications, provided project management, contributed to the interview guide, and coordinated recruitment. DP and JSL identified eligible patients, administrators, and clinicians for recruitment. SLC, FMB, LND, KWK, DBB, and AF contributed clinical expertise to the interview guide. CMP and EEH oversaw development of the interview guide, and CMP and LJL conducted interviews. CMP, EEH, and LJL conducted the analyses. All authors contributed to the drafting of the manuscript and read and approved the final manuscript. We would like to acknowledge our interview participants for giving of their time for our study.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
This study was performed in line with the principles of the Declaration of Helsinki and all study activities were approved by the Kaiser Permanente Southern California Institutional Review Board (IRB) #11103. Verbal consent was obtained for all participants.
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Hahn, E.E., Munoz-Plaza, C.E., Lyons, L.J. et al. Barriers and facilitators to implementation and sustainment of guideline-recommended depression screening for patients with breast cancer in medical oncology: a qualitative study. Support Care Cancer 31, 461 (2023). https://doi.org/10.1007/s00520-023-07922-0
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00520-023-07922-0