Abstract
Purpose
To evaluate the effectiveness of photobiomodulation (PBMT) in preventing dysgeusia in breast cancer patients treated with doxorubicin-cyclophosphamide (AC).
Methods
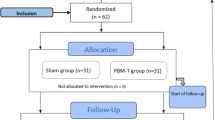
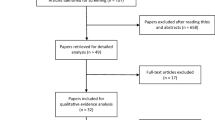
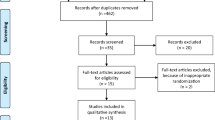
This is a phase II, randomized, triple-blind, placebo-controlled clinical trial involving 112 breast cancer patients treated with AC. The patients were divided equally into two groups: a test group treated with 2 J red laser and 3 J infrared laser on 21 points that were symmetrically distributed on the tongue on day 0 of four cycles of AC, and an equal placebo group treated with simulated PBMT to blind the patient, evaluator, and statistician. The clinicopathological and sociodemographic data, results of taste test, and subjective taste analysis, and the QoL, ECOG performance status, body mass index, and other side effects were recorded. The data were analyzed using ANOVA-RM/Bonferroni, Friedman/Dunn, and chi-square/Fisher’s exact tests.
Results
PBMT patients showed less objective and subjective taste loss (p<0.05). On the other hand, the placebo group showed a higher ECOG status (p=0.037) and more significant weight loss (p<0.001) after four cycles of AC. The QoL was significantly higher in the PBMT group (p<0.05) at all assessment periods, and PBMT treatment also reduced the incidence of cachexia (p=0.020), anorexia (p<0.001), diarrhea (p=0.040), oral mucositis (p=0.020), and vomiting (p=0.008).
Conclusion
PBMT reduced the taste loss and improved the overall health status and QoL of patients with breast cancer treated with AC.
Trial registration
Brazilian Clinical Trials Registry (www.ensaiosclinicos.gov.br) approval number RBR-9qnm34y, registered on 01/05/2021



Similar content being viewed by others
Availability of data
Not applicable.
References
Gadisa DA, Assefa M, Wang SH, Yimer G (2020) Toxicity profile of doxorubicin-cyclophosphamide and doxorubicin-cyclophosphamide followed by paclitaxel regimen and its associated factors among women with breast cancer in Ethiopia: a prospective cohort study. J Oncol Pharm Pract 26(8):1912–1920. https://doi.org/10.1177/1078155220907658
Berruti A, Generali D, Kaufmann M, Puztai L, Curigliano G et al (2011) International expert consensus on primary systemic therapy in the management of early breast cancer: highlights of the fourth symposium on primary systemic therapy in the management of operable breast cancer, Cremona, Italy. J Natl Cancer Inst Monogr 43(1):147–151. https://doi.org/10.1093/jncimonographs/lgr037
Harahap Y, Ardiningsih P, Winarti AC, Purwanto DJ (2020) Analysis of the doxorubicin and doxorubicinol in the plasma of breast cancer patients for monitoring the toxicity of doxorubicin. Drug Des Devel Ther 25(14):3469–3475. https://doi.org/10.2147/DDDT.S251144
Ongnok B, Chattipakorn N, Chattipakon SC (2020) Doxorubicin and cisplatin induced cognitive impairment: the possible mechanisms and interventions. Exp Neurol 32(4):113–118. https://doi.org/10.1016/j.expneurol.2019.113118
Pugnaloni S, Vignini A, Borroni F, Sabbatinelli J, Alia S et al (2020) Modifications of taste sensitivity in cancer patients: a method for the evaluations of dysgeusia. Support Care Cancer 28(3):1173–1181
De Vries YC, Boesveldt S, Kelfkens CS, Posthuma EE (2018) Taste and smell perception and quality of life during and after systemic therapy for breast cancer. Breast Cancer Res Treat 170(1):27–34. https://doi.org/10.1007/s10549-018-4720-3
De Vries YC, Van Den Berg MMGA, De Vries JHM, Boesveldt S (2017) Differences in dietary in take during chemotherapy in breast cancer patients compared to women without cancer. Support Care Cancer 25(8):2581–2591. https://doi.org/10.1007/s00520-017-3668-x
Ponticelli EM, Claris M, Frigerio M, Bergese I, Scavino E, Bernardini A (2017) Dysgeusia and health-related quality of life of cancer patients receiving chemotherapy: a cross-sectional study. Eur J Cancer Care 26(2). https://doi.org/10.1111/ecc.12633
Freitas PM, Simões A (2015) Lasers in dentistry: guideline for clinical practice. Willey Beckwell 49(1):27–33
Paglioni MP, Araújo ALD, Arboleda LPA, Palmier NR, Fonsêca JM et al (2019) Tumor safety and side effects of photobiomodulation therapy used for prevention and management of cancer treatment toxicities: a systematic review. Oral Oncol 93(1):21–28. https://doi.org/10.1016/j.oraloncology.2019.04.004
Zanin T, Zanin F, Carvalhosa AA, Souza PHC, Pacheco MT et al (2010) Use of 660 nm diode laser in the prevention and treatment of human oral mucositis induced by radiotherapy and chemotherapy. Photomed Laser Surg 28(2):233–237. https://doi.org/10.1089/pho.2008.2242
Mobadder ME, Farhat F, Mobadder WEM, Nammour S (2019) Photobiomodulation therapy in the treatment of oral mucositis, dysphagia, oral dryness, taste alteration, and burning mouth sensation due to cancer therapy: a case series. Int J Environ Res Public Health 16(22):4505. https://doi.org/10.3390/ijerph16224505
Mobadder ME, Nammour S (2021) Photobiomodulation for taste alteration. Encyclopedia. 2021(1):240–248
Bigotte L, Olsson Y (1987) Degeneration of trigeminal ganglion neurons caused by retrograde axonal transport of doxorubicin. Neurology. 37(6):985–992. https://doi.org/10.1212/wnl.37.6.985
De Oliveira RF, Silva AC, Simões A, Youssef MN, De Freitas PM (2015) Laser therapy in the treatment of paresthesia: a retrospective study of 125 clinical cases. Photomed Laser Surg 33(8):415–423. https://doi.org/10.1089/pho.2015.3888
Zecha JAEM, Durlacher JER, Nair RG, Epstein JB, Sonis ST et al (2016) Low level laser therapy/photobiomodulation in the management of side effects of chemoradiation therapy in head and neck cancer: part 1: mechanisms of action, dosimetric, and safety considerations. Support Care Cancer 24(6):2781–2792
Löe H, Silness J (1963) Periodontal disease in pregnancy I. Prevalence and severity. Acta Odontol Scand 21(6):533–551
Navazesh M (1993) Methods for collecting saliva. Ann N Y Acad Sci 69(4):72–77
Albuquerque AFM, Soares ECS, de Barros Silva PG et al (2019) Clinical investigation of gustatory and neurosensory alterations following mandibular third molar surgery: an observational prospective study. Clin Oral Investig 23(1):2941–2949
National Cancer Institute (2017) Common Toxicity Criteria, version 5.0 http://safetyprofiler-ctep.nci.nih.gov/CTC/CTC.aspx
Epstein JB, Barasch A (2010) Taste disorders in cancer patients: pathogenesis and approach to evaluation and treatment. Oral Oncol 46(2):77–81
Silveira MF, Marôco JP, Martins AMEBL, Marcopinto LF (2014) Impact of oral health on the physical and psychosocial dimensions: an analysis using structural equation modeling. Cad Saúde Pública 30(6):1–15
Oken M, Richard CH, Douglass CT, John MDH, Thomas ED, Eleanor ET, Paul PC (1982) Toxicity and response criteria of the Eastern Cooperative Oncology Group. Am J Clin Oncol 5(6):649–656
Sánchez-Lara K, Sosa-Sánchez R, Green-Renner D, Rodríguez C, Laviano A, Motola-Kuba D, Arrieta O (2010) Influence of taste disorders on dietary behaviors in cancer patients under chemotherapy. Nutr J 24(1):9–15
Yoshimoto NI, Inagaki M, Sekiguchi Y, Tomishima Y, Masuko K (2020) Chemotherapy alters subjective senses of taste and smell but not dietary patterns in Japanese lung cancer patients. Support Care Cancer 28(4):1667–1674. https://doi.org/10.1007/s00520-019-04958-z
Veronese P, Heachul DT, Scanavacca MI, Hajjar LA, Wu TC et al (2018) Effects of anthracycline, cyclophosphamide and taxane chemotherapy on CTc measurements in patients with breast cancer. PLoS One 13(5):763–784
Kuba S, Fujiyama R, Yamanouchi K, Morita M, Sakimura C et al (2018) Awareness of dysgeusia and gustatory tests in patients undergoing chemotherapy for breast cancer. Support Care Cancer 26(11):3883–3889. https://doi.org/10.1007/s00520-018-4256-4
Turcott JG, Hernández EJ, Lara KS, Estrada DF, Barrón ZLZB (2020) Baseline dysgeusia in chemotherapy-naïve non-small cell lung cancer patients: association with nutrition and QoL. Nutr Cancer 72(2):194–201
Bressan V, Stevanin S, Bianchi M, Aleo G, Bagnasco A, Sasso L (2016) The effects of swallowing disorders, dysgeusia, oral mucositis and xerostomia on nutritional status, oral intake and weight loss in head and neck cancer patients: a systematic review. Cancer Treat Rev 45(1):105–119
Álvaro SE, Abilés J, Garrido MS, Ruíz EP, García JA, Domínguez R (2021) Impact of weight loss on cancer patients’ quality of life at the beginning of the chemotherapy. Support Care Cancer 29(2):627–634
Amézaga J, Alfaro B, Ríos Y, Larraioz A, Ugartemendia G (2018) Assessing taste and smell alterations in cancer patients undergoing chemotherapy according to treatment. Support Care Cancer 6(12):4077–4086
Boltong A, Keast R, Boltong A, Reed RD (2015) Chemosensory science in the context of cancer treatment: implications for patient care. Chemosens Percept 8(3):117–125. https://doi.org/10.3390/nu11102285
Tam SY, Tam VCW, Ramkumar S, Khaw ML, Law HKW, Lee SWY (2020) Review on the cellular mechanisms of low-level laser therapy use in oncology. Front Oncol 10:1255
Hovan AJ, Williams PM, Stevenson-Moore P, Wahlin YB (2010) A systematic review of dysgeusia induced by cancer therapies. Support Care Cancer 18(8):1081–1087
Martini S, Iorio GC, Arcadipane F, Olivero F, Silvetti P et al (2019) Prospective assessment of taste impairment and nausea during radiotherapy for head and neck cancer. Med Oncol 36(5):44
Yan R, Chen X, Gong X, Wang J, Yu J (2018) The association of tooth loss, tooth brushing, and QoL among cancer survivors. Cancer Med 7(12):6374–6384
Hong JH, Ozbek PO, Stanek BT, Dietrich AM, Duncan SE, Lee YW, Lesser G (2009) Taste and odor abnormalities in cancer patients. J Support Oncol 7(2):58–65
Zecha JAEM, Raber-Durlacher JE, Nair RG, Epstein JB, Elad S et al (2018) Low-level laser therapy/photobiomodulation in the management of side effects of chemoradiation therapy in head and neck cancer: part 2: proposed applications and treatment protocols. Support Care Cancer 24(6):2793–2805. https://doi.org/10.1007/s00520-016-3153-y
Elad S, Cheng KKF, Lalla RV, Yarom N, Hong C et al (2020) MASCC/ISOO clinical practice guidelines for the management of mucositis secondary to cancer therapy. Cancer 126(19):4423–4431
Bensadoun RJ, Epstein JB, Nair RJ, Barasch A, Durlacher JER, Migliorati C (2020) Safety and efficacy of photobiomodulation therapy in oncology: a systematic review. Cancer Med 9(22):8279–8300
Nóbrega-Malta CE, Rodrigues MIQ, Lemos JVM, Duarte FB, Silva-Barros PG, Alves APNN (2020) Incidence and risk factors for oral mucositis after bone marrow transplantation patients under low intensity laser therapy: a longitudinal study. J Bone Marrow Transplant Cell Ther 2(1):1–11
Oberoi S, Netto GZ, Beyene J, Treister N, Sung L (2014) Effect of prophylactic low level laser therapy on oral mucositis: a systematic review and meta-analysis. PLoS One 9(9):e107418
Loncar B, Stipetic MM, Evic MB, Risovic D (2020) The effect of low-level laser therapy on salivary glands in patients with xerostomia. Photom. Laser Surg 1:1–9
Feng P, Zhao H, Chai J, Huang L, Wang H (2012) Expression and secretion of TNF-α in mouse taste buds: a novel function of a specific subset of type II taste cells. PLoS One 7(8):e43140
Bitencourt GB, Motta LJ, Teixeira da Silva DF, Turcio KHL, Sfalcin RA et al (2021) Evaluation of the preventive effect of photobiomodulation on orofacial discomfort in dental procedures: a randomized-controlled, crossover study and clinical trial. Photobiomodul Photomed Laser Surg 39(1):38–45
Acknowledgements
We thank the Haroldo Juaçaba Hospital (HHJ) - Ceará Cancer Institute for their contributions to this research, especially the SUS Chemotherapy service, for providing the infrastructure for the execution of this study.
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. Material preparation, data collection, and analysis were performed by Cássia Emanuella Nóbrega Malta, Anna Clara Aragão Matos Carlos, and Paulo Goberlânio de Barros Silva. The first draft of the manuscript was written by Cássia Emanuella Nóbrega Malta and Paulo Goberlânio de Barros Silva, and all authors Manuele Carine Maciel de Alencar, Eveline Fernandes Alves e Silva, Victor Bruno Caitano Nogueira, Ana Paula Negreiros Nunes Alves, Fábio Figueiredo Chaves, and José Fernando Bastos de Moura included patients, performed systemic analysis, and commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Ethics approval
This study was approved by the Ethics Committee of the Haroldo Juaçaba Hospital (HHJ) (Instituto do Câncer do Ceará — ICC) under protocol number 3.286.363. All ethical aspects expressed in Resolution No. 466 of 2012 of the National Health Council/Ministry of Health, which brings the guidelines and regulatory standards for research with human beings and in accordance with the National Research Ethics Committee (CONEP) standard, were respected.
Consent to participate
Informed consent was obtained from all individual participants included in the study
Consent for publication
The authors affirm that human research participants provided informed consent for publication
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information

Supplementary File 1
Schematic drawing of the low intensity red and infrared laser application points, based on the model of ZECHA et al., 2016. (PNG 381 kb)

Supplementary File 2
Individual taste function after four administrations of doxorubicin and cyclophosphamide in breast cancer patients submitted to the PBMT protocol for the prevention of dysgeusia. (PNG 52 kb)
Supplementary File 3
CheckList – Consort 2010 (DOCX 36 kb)
Rights and permissions
About this article
Cite this article
Malta, C.E.N., Carlos, A.C.A.M., de Alencar, M.C.M. et al. Photobiomodulation therapy prevents dysgeusia chemotherapy induced in breast cancer women treated with doxorubicin plus cyclophosphamide: a triple-blinded, randomized, placebo-controlled clinical trial. Support Care Cancer 30, 2569–2580 (2022). https://doi.org/10.1007/s00520-021-06642-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00520-021-06642-7