Abstract
Purpose
This phase II trial assessed the clinical benefit of beta-hydroxy-beta-methylbutyrate, arginine, and glutamine (HMB/Arg/Gln) for preventing chemoradiotherapy (CRT)-induced oral mucositis (OM) in patients with head and neck cancer (HNC).
Methods
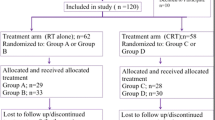
Patients with HNC receiving definitive or postoperative cisplatin-based CRT were enrolled. HMB/Arg/Gln was administered orally or per percutaneous endoscopic gastrostomy from the first day of CRT up to its completion. All patients received opioid-based pain control and oral care programs that we previously reported. The primary endpoint was the incidence of grade ≥ 3 OM (functional/symptomatic) according to the Common Terminology Criteria of Adverse Events version 3.0. Quality of life (EORTC QLQ-C30/PROMS) at baseline and upon radiotherapy at a dosage of 50 Gy were assessed.
Results
Thirty-five patients with HNC were enrolled. Sixteen of them (45.7%) developed grade ≥ 3 OM (i.e., functional/symptomatic). The incidence of grade ≤ 1 OM (functional/symptomatic) was 51.5% at 2 weeks and 82.9% at 4 weeks after radiotherapy completion. Clinical examination revealed that 10 patients (28.6%) developed grade ≥ 3 OM. The incidence of grade ≤ 1 OM (clinical exam) was 80.0% at 2 weeks and 100% at 4 weeks after radiotherapy completion. Adverse events related to HMB/Arg/Gln were an increase in blood urea nitrogen and diarrhea, but were easily managed.
Conclusions
The addition of HMB/Arg/Gln to opioid-based pain control and oral care programs was feasible but still insufficient at reducing the incidence of CRT-induced severe OM. However, the benefit of HMB/Arg/Gln should not be neglected given the findings of clinical examinations and the rapid recovery from severe OM.
Trial registration


Similar content being viewed by others
References
Marur S, Forastiere AA (2008) Head and neck cancer: changing epidemiology, diagnosis, and treatment. Mayo Clin Proc 83:489–501
Forastiere AA, Goepfert H, Maor M, Pajak TF, Weber R, Morrison W, Glisson B, Trotti A, Ridge JA, Chao C, Peters G, Lee DJ, Leaf A, Ensley J, Cooper J (2003) Concurrent chemotherapy and radiotherapy for organ preservation in advanced laryngeal cancer. N Engl J Med 349:2091–2098
Adelstein DJ, Li Y, Adams GL, Wagner H Jr, Kish JA, Ensley JF, Schuller DE, Forastiere AA (2003) An intergroup phase III comparison of standard radiation therapy and two schedules of concurrent chemoradiotherapy in patients with unresectable squamous cell head and neck cancer. J Clin Oncol 21:92–98
Cooper JS, Pajak TF, Forastiere A, Radiation Therapy Oncology Group 9501/Intergroup et al (2004) Postoperative concurrent radiotherapy and chemotherapy for high-risk squamous-cell carcinoma of the head and neck. N Engl J Med 350:1937–1944
Bernier J, Domenge C, Ozsahin M, European Organization for Research and Treatment of Cancer Trial 22931 et al (2004) Postoperative irradiation with or without concomitant chemotherapy for locally advanced head and neck. N Engl J Med 350:1945–1952
Naidu MU, Ramana GV, Rani PU, Mohan IK, Suman A, Roy P (2004) Chemotherapy induced and/or radiation therapy-induced oral mucositis—complicating the treatment of cancer. Neoplasia 6:423–431
Vera-Llonch M, Oster G, Hagiwara M, Sonis S (2006) Oral mucositis in patients undergoing radiation treatment for head and neck carcinoma. Cancer 106:329–336
Hansen O, Overgaard J, Hansen HS, Overgaard M, Höyer M, Jörgensen KE (1997) Importance of overall treatment time for the outcome of radiotherapy of advanced head and neck carcinoma: dependency on tumor differentiation. Radiother Oncol 43:47–51
Russo G, Haddad R, Posner M, Machtay M (2008) Radiation treatment breaks and ulcerative mucositis in head and neck cancer. Oncologist 13:886–898
Bese NS, Hendry J, Jeremic B (2007) Effects of prolongation of overall treatment time due to unplanned interruptions during radiotherapy of different tumor sites and practical methods for compensation. Int J Radiat Oncol Biol Phys 68:654–661
Zenda S, Matsuura K, Tachibana H, Homma A, Kirita T, Monden N, Iwae S, Ota Y, Akimoto T, Otsuru H, Tahara M, Kato K, Asai M (2011) Multicenter phase II study of an opioid-based pain control program for head and neck cancer patients receiving chemoradiotherapy. Radiother Oncol 101:410–414
Yokota T, Tachibana H, Konishi T, Yurikusa T, Hamauchi S, Sakai K, Nishikawa M, Suzuki M, Naganawa Y, Hagihara T, Tsumaki H, Kubo T, Sato M, Taguri M, Morita S, Eguchi T, Kubota K, Zenda S (2016) Multicenter phase II study of an oral care program for patients with head and neck cancer receiving chemoradiotherapy. Support Care Cancer 24:3029–3036
Yokota T, Ogawa T, Takahashi S, Okami K, Fujii T, Tanaka K, Iwae S, Ota I, Ueda T, Monden N, Matsuura K, Kojima H, Ueda S, Sasaki K, Fujimoto Y, Hasegawa Y, Beppu T, Nishimori H, Hirano S, Naka Y, Matsushima Y, Fujii M, Tahara M (2017) Efficacy and safety of rebamipide liquid for chemoradiotherapy-induced oral mucositis in patients with head and neck cancer: a multicenter, randomized, double-blind, placebo-controlled, parallel-group phase II study. BMC Cancer 17:314
Whitney J, Phillips L, Aslam R, Barbul A, Gottrup F, Gould L, Robson MC, Rodeheaver G, Thomas D, Stotts N (2006) Guidelines for the treatment of pressure ulcers. Wound Repair Regen 14:663–679
Williams JZ, Abumrad N, Barbul A (2002) Effect of a specialized amino acid mixture on human collagen deposition. Ann Surg 236:369–374 discussion 74–75
List MA, Ritter-Sterr C, Lansky SB (1990) A performance status scale for head and neck cancer patients. Cancer 66:564–569
Kushner JA, Lawrence HP, Shoval I, Kiss TL, Devins GM et al (2008) Development and validation of a Patient-Reported Oral Mucositis Symptom (PROMS) scale. J Can Dent Assoc 74:59
Ziegler TR, Benfell K, Smith RJ, Young LS, Brown E, Ferrari-Baliviera E, Lowe DK, Wilmore DW (1990) Safety and metabolic effects of L-glutamine administration in humans. JPEN 14(4 Suppl):137S–146S
Hurson M, Regan MC, Kirk SJ, Wasserkurg HL, Barbul A (1995) Metabolic effects of arginine in a healthy elderly population. JPEN 19:227–230
Garg S, Yoo J, Winquist E (2010) Nutritional support for head and neck cancer patients receiving radiotherapy: a systematic review. Support Care Cancer 18:667–677
Paccagnella A, Morello M, Da Mosto MC et al (2010) Early nutritional intervention improves treatment tolerance and outcomes in head and neck cancer patients undergoing concurrent chemoradiotherapy. Support Care Cancer 18:837–845
Karna E, Miltyk W, Wolczynski S, Palka JA (2001) The potential mechanism for glutamine-induced collagen biosynthesis in cultured human skin fibroblasts. Comp Biochem Physiol B Biochem Mol Biol 130:23–32
Barbul A, Lazarou SA, Efron DT, Wasserkrug HL, Efron G (1990) Arginine enhances wound healing and lymphocyte immune responses in humans. Surgery 108:331–336 discussion 336–337
Kirk SJ, Hurson M, Regan MC et al (1993) Arginine stimulates wound healing and immune function in elderly human beings. Surgery 114:155–159 discussion 160
Huang EY, Leung SW, Wang CJ, Chen HC, Sun LM, Fang FM, Yeh SA, Hsu HC, Hsiung CY (2000) Oral glutamine to alleviate radiation-induced oral mucositis: a pilot randomized trial. Int J Radiat Oncol Biol Phys 46:535–539
Smith HJ, Wyke SM, Tisdale MJ (2004) Mechanism of the attenuation of proteolysis-inducing factor stimulated protein degradation in muscle by beta-hydroxy-beta-methylbutyrate. Cancer Res 64:8731–8735
Imai T, Matsuura K, Asada Y, Sagai S, Katagiri K, Ishida E, Saito D, Sadayasu R, Wada H, Saijo S (2014) Effect of HMB/Arg/Gln on the prevention of radiation dermatitis in head and neck cancer patients treated with concurrent chemoradiotherapy. Jpn J Clin Oncol 44(5):422–427
Acknowledgements
The authors thank Ms. Marina Kobayashi for data collection.
Funding
This study was supported by the Public Interest Incorporated Foundation - Shizuoka Industrial Foundation - Pharma Valley Center. The funders had no role regarding study design, data analysis, and the decision to publish or preparation of the manuscript.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
Tomoya Yokota serves in an advisory role for AstraZeneca, Merck Serono, Bayer, Ono Pharma CO., Ltd., and Bristol-Myers Squibb, and has received lecture fees from Merck Serono, Ono Pharma CO., Ltd., Eisai, Bayer, and Bristol-Myers Squibb.
Electronic supplementary material
Supplementary Fig. 1
The effect of HMB/Arg/Gln supplementation on blood urea nitrogen (mg/dL). *, p<0.01. Bar: standard deviation. (GIF 98 kb)
Supplementary Fig. 2
Quality of life and symptom burden. The mean values of functional and symptom scales in QLQ-C30 are shown at baseline, at the time of radiotherapy administration of a dosage of 50 Gy, and 4 weeks after radiotherapy completion. All scales range from 0 to 100 and were scored such that higher values indicate better functioning or higher symptom burden. (GIF 118 kb)
Rights and permissions
About this article
Cite this article
Yokota, T., Hamauchi, S., Yoshida, Y. et al. A phase II study of HMB/Arg/Gln against oral mucositis induced by chemoradiotherapy for patients with head and neck cancer. Support Care Cancer 26, 3241–3248 (2018). https://doi.org/10.1007/s00520-018-4175-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00520-018-4175-4