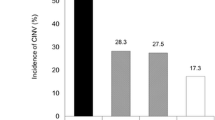
Abstract
Purpose
We assessed the cost-effectiveness of a risk model-guided (RMG) antiemetic prophylaxis strategy compared with the physician’s choice (PC) strategy in patients receiving chemotherapy for early-stage breast cancer.
Methods
We conducted a cost-utility analysis based on a published randomized controlled trial of 324 patients with early-stage breast cancer undergoing chemotherapy at two Canadian cancer centers. Patients were randomized to receive their antiemetic treatments according to either predefined risk scores or the treating physician’s preference. Effectiveness was measured as quality-adjusted life years (QALYs) gained. Cost and utility data were obtained from the Canadian published literature. We used generalized estimating equations to estimate the incremental cost-effectiveness ratios (ICERs) and 95% confidence intervals (CIs) over a range of willingness-to-pay values. The lower and upper bounds of the 95% CIs were used to characterize the statistical uncertainty for the cost-effectiveness estimates and construct cost-effectiveness acceptability curves.
Results
From the health care system’s perspective, the RMG strategy was associated with greater QALYs gained (0.0016, 95% CI 0.0009, 0.0022) and higher cost ($49.19, 95% CI $24.87, $73.08) than the PC strategy, resulting in an ICER of $30,864.28 (95% CI $14,718.98, $62,789.04). At the commonly used threshold of $50,000/QALY, the probability that RMG prophylaxis is cost-effective was >94%; this probability increased with greater willingness-to-pay values.
Conclusion
The risk-guided antiemetic prophylaxis is an economically attractive option for patients receiving chemotherapy for early-stage breast cancer. This information supports the implementation of risk prediction models to guide chemotherapy-induced nausea and vomiting prophylaxis in clinical practices.



Similar content being viewed by others
References
Coates A, Abraham S, Kaye SB, Sowerbutts T, Frewin C, Fox RM, Tattersall MH (1983) On the receiving end—patient perception of the side-effects of cancer chemotherapy. Eur J Cancer 19(2):203–208
Roscoe JA, Morrow GR, Hickok JT, Stern RM (2000) Nausea and vomiting remain a significant clinical problem: trends over time in controlling chemotherapy-induced nausea and vomiting in 1413 patients treated in community clinical practices. J Pain Symptom Manage 20(2):113–121
Kuchuk I, Bouganim N, Beusterien K, Grinspan J, Vandermeer L, Gertler S, Dent SF, Song X, Segal R, Mazzarello S, Crawley F, Dranitsaris G, Clemons M (2013) Preference weights for chemotherapy side effects from the perspective of women with breast cancer. Breast Cancer Res Treat 142(1):101–107. doi:10.1007/s10549-013-2727-3
Jordan K, Sippel C, Schmoll HJ (2007) Guidelines for antiemetic treatment of chemotherapy-induced nausea and vomiting: past, present, and future recommendations. Oncologist 12(9):1143–1150. doi:10.1634/theoncologist.12-9-1143
Ng T, Mazzarello S, Wang Z, Hutton B, Dranitsaris G, Vandermeer L, Smith S, Clemons M (2016) Choice of study endpoint significantly impacts the results of breast cancer trials evaluating chemotherapy-induced nausea and vomiting. Breast Cancer Res Treat 155(2):337–344. doi:10.1007/s10549-015-3669-8
Dranitsaris G, Mazzarello S, Smith S, Vandermeer L, Bouganim N, Clemons M (2016) Measuring the impact of guideline-based antiemetic therapy on nausea and vomiting control in breast cancer patients with multiple risk factors. Support Care Cancer 24(4):1563–1569. doi:10.1007/s00520-015-2944-x
Hernandez Torres C, Mazzarello S, Ng T, Dranitsaris G, Hutton B, Smith S, Munro A, Jacobs C, Clemons M (2015) Defining optimal control of chemotherapy-induced nausea and vomiting-based on patients’ experience. Support Care Cancer 23(11):3341–3359. doi:10.1007/s00520-015-2801-y
Dranitsaris G, Clemons M (2014) Risk prediction models for chemotherapy-induced nausea and vomiting: almost ready for prime time? Support Care Cancer 4:863–864. doi:10.1007/s00520-014-2134-2
Lee MA, Cho EK, Oh SY, Ahn JB, Lee JY, Thomas B, Jung H, Kim JG (2016) Clinical practices and outcomes on chemotherapy-induced nausea and vomiting management in South Korea: comparison with Asia-Pacific data of the pan Australasian chemotherapy induced emesis burden of illness study. Cancer Res Treat 48(4):1420–1428. doi:10.4143/crt.2015.309
Zong X, Zhang J, Ji X, Gao J, Ji J (2016) Patterns of antiemetic prophylaxis for chemotherapy-induced nausea and vomiting in China. Chin J Cancer Res 28(2):168–179. doi:10.21147/j.issn.1000-9604.2016.02.04
Caracuel F, Munoz N, Banos U, Ramirez G (2015) Adherence to antiemetic guidelines and control of chemotherapy-induced nausea and vomiting (CINV) in a large hospital. J Oncol Pharm Pract 21(3):163–169. doi:10.1177/1078155214524809
Van Laar ES, Desai JM, Jatoi A (2015) Professional educational needs for chemotherapy-induced nausea and vomiting (CINV): multinational survey results from 2388 health care providers. Support Care Cancer 23(1):151–157. doi:10.1007/s00520-014-2325-x
Hutton B, Clemons M, Mazzarello S, Kuchuk I, Skidmore B, Ng T (2015) Identifying an optimal antiemetic regimen for patients receiving anthracycline and cyclophosphamide-based chemotherapy for breast cancer--an inspection of the evidence base informing clinical decision-making. Cancer Treat Rev 41(10):951–959. doi:10.1016/j.ctrv.2015.09.007
Dranitsaris G, Joy A, Young S, Clemons M, Callaghan W, Petrella T (2009) Identifying patients at high risk for nausea and vomiting after chemotherapy: the development of a practical prediction tool I. J Support Oncol 7:W1–W8
Petrella T, Clemons M, Joy A, Young S, Callaghan W, Dranitsaris G (2009) Identifying patients at high risk for nausea and vomiting after chemotherapy: the development of a practical prediction tool II. J Support Oncol 7:W9–16
Bouganim N, Dranitsaris G, Hopkins S, Vandermeer L, Godbout L, Dent S, Wheatley-Price P, Milano C, Clemons M (2012) Prospective validation of risk prediction indexes for acute and delayed chemotherapy-induced nausea and vomiting. Curr Oncol 19(6):e414–e421. doi:10.3747/co.19.1074
Clemons M, Bouganim N, Smith S, Mazzarello S, Vandermeer L, Segal R, Dent S, Gertler S, Song X, Wheatley-Price P, Dranitsaris G (2016) Risk model-guided antiemetic prophylaxis vs physician’s choice in patients receiving chemotherapy for early-stage breast cancer: a randomized clinical trial. JAMA Oncol 2(2):225–231. doi:10.1001/jamaoncol.2015.3730
Grunberg S, WEeks J, Mgnan WF, Herndon J, Naughton M, Blackwell KL, Wood ME, Christian DL, Perry MC, Dees C, Reed E, Marshall E, for the Cancer and Leukemia Group B (2009) Determination of utilities for control of chemotherapy-induced nausea or vomiting–CALGB 309801. J Support Oncol 7:W17–W22
Ontario Ministry of Health and Long-term Care (2016a) Ontario Drug Benefit Formulary/Comparative Drug Index. https://www.formulary.health.gov.on.ca/formulary/. Accessed May 2 2016
Mittmann N, Verma S, Koo M, Alloul K, Trudeau M (2010) Cost effectiveness of TAC versus FAC in adjuvant treatment of node-positive breast cancer. Curr Oncol 17(1):7–16
Younis T, Rayson D, Sellon M, Skedgel C (2008) Adjuvant chemotherapy for breast cancer: a cost-utility analysis of FEC-D vs. FEC 100. Breast Cancer Res Treat 111(2):261–267. doi:10.1007/s10549-007-9770-x
Younis T, Rayson D, Skedgel C (2011) The cost-utility of adjuvant chemotherapy using docetaxel and cyclophosphamide compared with doxorubicin and cyclophosphamide in breast cancer. Curr Oncol 18(6):e288–e296
Ontario Case Costing Initiative (2012) OCCI costing analysis tool. http://www.occp.com/mainPage.htm. Accessed February 14 2015
Ontario Ministry of Health and Long-term Care (2016b) Schedule of benefits for physician services under the Health Insurance Act. http://www.health.gov.on.ca/english/providers/program/ohip/sob/physserv/sob_master20160406.pdf. Accessed May 2 2016
Statistics Canada (2016a) Table 326-0021—Consumer Price Index, annual (2002=100 unless otherwise noted), CANSIM (database). http://www5.statcan.gc.ca/cansim/a26?lang=eng&retrLang=eng&id=3260021&&pattern=&stByVal=1&p1=1&p2=37&tabMode=dataTable&csid=. Accessed May 31 2016
Hoch JS, Briggs AH, Willan AR (2002) Something old, something new, something borrowed, something blue: a framework for the marriage of health econometrics and cost-effectiveness analysis. Health Econ 11(5):415–430. doi:10.1002/hec.678
Diggle P, Heagerty P, Liang K, Zeger S (2013) Analysis of longitudinal data, 2nd edn. Oxford University Press, Oxford
Drummond MF, Stoddart GL, Torrance GW (2005) Methods for the economic evaluation for health care program. University Press, Oxford
Statistics Canada (2016b) Average hourly wages of employees by selected characteristics and occupation, unadjusted data, by province (monthly) (Canada). http://www.statcan.gc.ca/tables-tableaux/sum-som/l01/cst01/labr69a-eng.htm. Accessed May 31 2016
O’Brien BJ, Rusthoven J, Rocchi A, Latreille J, Fine S, Vandenberg T, Laberge F (1993) Impact of chemotherapy-associated nausea and vomiting on patients’ functional status and on costs: survey of five Canadian centres. CMAJ 149(3):296–302
Lachaine J, Yelle L, Kaizer L, Dufour A, Hopkins S, Deuson R (2005) Chemotherapy-induced emesis: quality of life and economic impact in the context of current practice in Canada. Support Cancer Ther 2(3):181–187. doi:10.3816/SCT.2005.n.011
Annemans L, Strens D, Lox E, Petit C, Malonne H (2008) Cost-effectiveness analysis of aprepitant in the prevention of chemotherapy-induced nausea and vomiting in Belgium. Support Care Cancer 16(8):905–915. doi:10.1007/s00520-007-0349-1
Lordick F, Ehlken B, Ihbe-Heffinger A, Berger K, Krobot KJ, Pellissier J, Davies G, Deuson R (2007) Health outcomes and cost-effectiveness of aprepitant in outpatients receiving antiemetic prophylaxis for highly emetogenic chemotherapy in Germany. Eur J Cancer 43(2):299–307. doi:10.1016/j.ejca.2006.09.019
Chan SL, Jen J, Burke T, Pellissier J (2014) Economic analysis of aprepitant-containing regimen to prevent chemotherapy-induced nausea and vomiting in patients receiving highly emetogenic chemotherapy in Hong Kong. Asia Pac J Clin Oncol 10(1):80–91. doi:10.1111/ajco.12170
Moore S, Tumeh J, Wojtanowski S, Flowers C (2007) Cost-effectiveness of aprepitant for the prevention of chemotherapy-induced nausea and vomiting associated with highly emetogenic chemotherapy. Value Health 10(1):23–31. doi:10.1111/j.1524-4733.2006.00141.x
Dranitsaris G, Leung P (2004) Using decision modeling to determine pricing of new pharmaceuticals: the case of neurokinin-1 receptor antagonist antiemetics for cancer chemotherapy. Int J Technol Assess Health Care 20(3):289–295
Aapro M, Molassiotis A, Dicato M, Pelaez I, Rodriguez-Lescure A, Pastorelli D, Ma L, Burke T, Gu A, Gascon P, Roila F (2012) The effect of guideline-consistent antiemetic therapy on chemotherapy-induced nausea and vomiting (CINV): the Pan European Emesis Registry (PEER). Ann Oncol 23(8):1986–1992. doi:10.1093/annonc/mds021
Authors’ contributions
KT, LV, SM, DF, and MC contributed to the conception of the study question and design. DC, JH, and GD guided the analyses. LV and ZW helped with data management. KT performed the analyses and prepared the first draft of the manuscript. All authors reviewed and approved the manuscript.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
The study protocol has been approved by local institutional review boards.
Conflict of interest
The authors declare that they have no conflicts of interest.
Funding
This study was conducted without funding.
Electronic supplementary material
ESM 1
(DOCX 174 kb)
Rights and permissions
About this article
Cite this article
Thavorn, K., Coyle, D., Hoch, J.S. et al. A cost-utility analysis of risk model-guided versus physician’s choice antiemetic prophylaxis in patients receiving chemotherapy for early-stage breast cancer: a net benefit regression approach. Support Care Cancer 25, 2505–2513 (2017). https://doi.org/10.1007/s00520-017-3658-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00520-017-3658-z