Abstract
Background
The use of granulocyte colony-stimulating factor (G-CSF) as a prophylaxis against febrile neutropenia (FN) is well documented in the literature; however, the therapeutic use of G-CSF in the treatment of FN remains controversial. This study assessed the efficacy of adjunctive G-CSF in the treatment of FN by evaluating clinical outcomes.
Methods
This was a single-center, prospective cohort study conducted at the National Cancer Center in Singapore. Adult patients who had received chemotherapy and developed FN between January 2009 and January 2012 were included in the analysis. The clinical efficacy of adjunctive G-CSF was evaluated by investigating the duration of hospitalization, duration to absolute neutrophil count (ANC) recovery, duration of grade IV neutropenia, duration to fever resolution, duration of antibiotic therapy, and incidence of documented infections. A multivariate analysis was performed to identify patients who could potentially benefit from adjunctive G-CSF.
Results
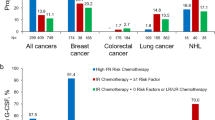
Four hundred and thirty patients were analyzed. Majority manifested low-risk FN (81.2 %) based on the Multinational Association of Supportive Care in Cancer (MASCC) scoring. Compared to patients who did not receive adjunctive G-CSF, patients receiving adjunctive G-CSF had a nonsignificant reduction in the duration of hospitalization (3.5 vs. 3.7 days, p = 0.41) and in ANC recovery time (3.4 vs. 3.5 days, p = 0.76). Neutropenia-related mortality was lower among those who have received adjunctive G-CSF (2.4 vs. 8.4 %, p = 0.006). Patients of Indian ethnicity and those who underwent gemcitabine-containing chemotherapy were less likely to receive adjunctive G-CSF treatment.
Conclusions
This observational study suggested that adjunctive G-CSF may confer clinical benefits among solid tumor and lymphoma patients with established febrile neutropenia. Further research should be conducted to validate the findings.
Similar content being viewed by others
References
Aapro MS, Bohlius J, Cameron DA et al (2010) Update of EORTC guidelines for the use of granulocyte-colony stimulating factor to reduce the incidence of chemotherapy-induced febrile neutropenia in adult patients with lymphoproliferative disorders and solid tumours. Eur J Cancer (Oxford, England: 1990) 47:8–32, 2011
American Society of Clinical Oncology (2006) Update of ASCO practice guideline recommendations for the use of white blood cell growth factors: guideline summary. J Oncol Pract 2:196–201
Pizzo PA (1993) Management of fever in patients with cancer and treatment-induced neutropenia. N Engl J Med 328:1323–1332
Pizzo PA (1999) Fever in immunocompromised patients. N Engl J Med 341:893–900
Pizzo PA, Hathorn JW, Hiemenz J et al (1986) A randomized trial comparing ceftazidime alone with combination antibiotic therapy in cancer patients with fever and neutropenia. N Engl J Med 315:552–558
Souza LM, Boone TC, Gabrilove J et al (1986) Recombinant human granulocyte colony-stimulating factor: effects on normal and leukemic myeloid cells. Science 232:61–65
Crawford J, Ozer H, Stoller R et al (1991) Reduction by granulocyte colony-stimulating factor of fever and neutropenia induced by chemotherapy in patients with small-cell lung cancer. N Engl J Med 325:164–170
National Comprehensive Cancer Network (2010) NCCN: clinical practice guidelines in oncology. Myeloid growth factors. http://www.nccn.org/professionals/physician_gls/PDF/myeloid_growth.pdf. Accessed Jan 2013
Maher DW, Lieschke GJ, Green M et al (1994) Filgrastim in patients with chemotherapy-induced febrile neutropenia: a double-blind, placebo-controlled trial. Ann Intern Med 121:492–501
Riikonen P, Saarinen UM, Makipernaa A et al (1994) Recombinant human granulocyte-macrophage colony-stimulating factor in the treatment of febrile neutropenia: a double blind placebo-controlled study in children. Pediatr Infect Dis J 13:197–202
Mayordomo JI, Rivera F, Díaz-Puente MT et al (1995) Improving treatment of chemotherapy-induced neutropenic fever by administration of colony-stimulating factors. J Natl Cancer Inst 87:803–808
Anaissie EJ, Vartivarian S, Bodey GP et al (1996) Randomized comparison between antibiotics alone and antibiotics plus granulocyte-macrophage colony-stimulating factor (Escherichia coli-derived) in cancer patients with fever and neutropenia. Am J Med 100:17–23
Clark OAC, Lyman GH, Castro AA et al (2005) Colony-stimulating factors for chemotherapy-induced febrile neutropenia: a meta-analysis of randomized controlled trials. J Clin Oncol 23:4198–4214
Berghmans T, Paesmans M, Lafitte J et al (2002) Therapeutic use of granulocyte and granulocyte-macrophage colony-stimulating factors in febrile neutropenic cancer patients. Support Care Cancer 10:181–188
Siddiqui T, Burney IA, Kakepoto GN et al (2002) Lack of benefit of granulocyte macrophage or granulocyte colony stimulating factor in patients with febrile neutropenia. J Pak Med Assoc 52:206–10
García-Carbonero R, Mayordomo JI, Tornamira MV et al (2001) Granulocyte colony-stimulating factor in the treatment of high-risk febrile neutropenia: a multicenter randomized trial. J Natl Cancer Inst 93:31–38
Freifeld AG, Bow EJ, Sepkowitz KA et al (2011) Clinical practice guideline for the use of antimicrobial agents in neutropenic patients with cancer: 2010 update by the Infectious Diseases Society of America. Clin Infect Dis 52:e56–93
National Cancer Institute and Department of Health and Human Services (2003) Common terminology criteria for adverse events v3.0 (CTCAE). http://ctep.cancer.gov/protocolDevelopment/electronic_applications/docs/ctcaev3.pdf. Accessed Jan 2013
Holmes FA, O’Shaughnessy JA, Vukelja S et al (2002) Blinded, randomized, multicenter study to evaluate single administration pegfilgrastim once per cycle versus daily filgrastim as an adjunct to chemotherapy in patients with high-risk stage II or stage III/IV breast cancer. J Clin Oncol 20:727–31
Basu SK, Fernandez ID, Fisher SG et al (2005) Length of stay and mortality associated with febrile neutropenia among children with cancer. J Clin Oncol 23:7958–7966
Lopez-Hernandez MA, Jimenez-Alvarado R, Borbolla-Escoboza R et al (2000) Granulocyte colony-stimulating factor in the treatment of febrile neutropenia. Gac Med Mex 136:99–105
Bodey GP, Buckley M, Sathe YS et al (1966) Quantitative relationships between circulating leukocytes and infection in patients with acute leukemia. Ann Intern Med 64:328–340
Kuderer NM, Dale DC, Crawford J et al (2006) Mortality, morbidity, and cost associated with febrile neutropenia in adult cancer patients. Cancer 106:2258–2266
Hor SY, Lee SC, Wong CI et al (2008) PXR, CAR and HNF4alpha genotypes and their association with pharmacokinetics and pharmacodynamics of docetaxel and doxorubicin in Asian patients. Pharmacogenomics J 8:139–46
Forrest GN, Schimpff SC, Cross A (2002) Febrile neutropenia, colony-stimulating factors and therapy: time for a new methodology? Support Care Cancer 10:177–80
Acknowledgments
We would like to thank Mrs. Ten Eng Lee and Mrs. Betty Yong for their assistance in the data collection. This study was partially funded by a grant received in 2010 from the Healthcare Quality Improvement and Innovation Fund established by the Ministry of Health, Singapore. The sponsor had no role in the design, implementation, analysis, or write-up of this audit.
Conflict of interest
The authors have no conflicts of interest that are directly relevant to the content of this study.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Chan, A., Wong, Q.X., Ali, M.K. et al. Clinical efficacy of adjunctive G-CSF on solid tumor and lymphoma patients with established febrile neutropenia. Support Care Cancer 22, 1105–1112 (2014). https://doi.org/10.1007/s00520-013-2067-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00520-013-2067-1