Abstract
Background
Initial treatment with morphine followed by fentanyl transdermal patch is the standard in Japan, since even the smallest patch (2.5 mg) could deliver too high an initial dose for Japanese patients. We evaluated the analgesic effect and safety of using the fentanyl transdermal patch as a first-line strong opioid for cancer pain that is resistant to nonsteroidal anti-inflammatory drugs (NSAIDs).
Patients and methods
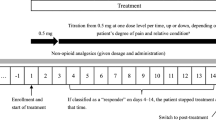
For 20 hospitalized patients with cancer pain that could not be controlled by NSAIDs, the fentanyl transdermal patch (1.25 mg; half of a 2.5-mg patch) was administered as a first-line strong opioid. We used rescue medications depending on the degree of pain, and the dose of fentanyl transdermal patch was adjusted every 3 days. To evaluate analgesic efficacy of the patch, the degree of pain was assessed twice a day, in the morning and at night, using a face rating scale. The formulation and dose of morphine used during observation period were recorded. The safety of treatment was evaluated by measuring vital signs once a day, and the severity of side effects were evaluated. Any abnormal findings in blood and urine test were recorded.
Results
The median pain score before administration of fentanyl transdermal patch was 3 ± 0.58 and was decreased to 2 ± 0.71 on day 9 of administration. The mean dose of fentanyl transdermal patch on day 9 of administration was 2.31 ± 1.34 mg, and the mean dose of morphine as rescue therapy was 4.62 ± 7.76 mg. No clinically significant changes in vital signs were observed. No severe adverse events were present when the dose of the fentanyl transdermal patch was 1.25 mg, but two patients experienced dizziness when the dose was increased from 2.5 to 5 mg. No abnormal laboratory data appeared during the administration.
Conclusion
The use of 1.25-mg fentanyl transdermal patch (50% of a 2.5-mg patch) seems to be safe and efficient as a first-line strong opioid. The use of 3.75-mg fentanyl transdermal patch may be necessary since adverse events including nausea and sleepiness are likely to occur by increasing from 2.5 to 5 mg.




Similar content being viewed by others
References
Kongsgaard UE, Poulain P (1998) Transdermal fentanyl for pain control in adults with chronic cancer pain. Eur J Pain 2:53–62
Ahmedzai S, Brooks D (1994) Transdermal fentanyl in cancer pain. The TTS-Fentanyl Multicentre Study Group. J Drug Dev 6:93–97
Ahmedzai S, Brooks D (1997) Transdermal fentanyl versus sustained-release oral morphine in cancer pain: preference, efficacy, and quality of life. The TTS-Fentanyl Comparative Trial Group. J Pain Symptom Manage 13:254–261
Sloan PA, Moulin DE, Hays H (1998) A clinical evalution of transdermal therapeutic system fentanyl for the treatment of cancer pain. J Pain Symptom Manage 16:102–111
Tan M, Xu R, Seth R (2004) Granisetron vs dolasetron for acute chemotherapy-induced nausea and vomiting (CINV) in high and moderately high emetogenic chemotherapy: an open-label pilot study. Curr Med Res Opin 20:879–882
Takeda F (translation). World Health Organization (volume). Opening from pain in cancer: WHO method cancer pain treatment. Kanahara publication 1996
Division of Pharmaceuticals and Chemicals, The Ministry of Health and Welfare. Outline of drug and the stimulant administration in 1991. 1993
Hiraga K (1999) Current state and view in the future of cancerous pain management in Japan. Pain Clin 20:479–484
Hiraga K, Takeda F (1999) The current state and the view in the future of the cancer pain treatment in Japan: the transition of the cancer pain treatment in the university hospital chiefly. Palliat Care 1:134–142
Andrews CJH, Prys-Roberts C (1983) Fentanyl—a review. Clin Anaesthesiol 1:97–122
Mather LE (1983) Clinical pharmacokinetics of fentanyl and its newer derivatives. Clin Pharmacokinet 8:422–446
Gardocki JF, Yelnosky J (1964) A study of some of the pharmacologic actions of fentanyl citrate. Toxicol Appl Pharmacol 6:48–62
Roy SD, Flynn GL (1990) Transdermal delivery of narcotic analgesics: pH, anatomical, and subject influences on cutaneous permeability of fentanyl and sufentanil. Pharm Res 7:842–847
Roy SD, Flynn GL (1989) Transdermal delivery of narcotic analgesics: comparative permeabilities of narcotic analgesics through human cadaver skin. Pharm Res 6:825–832
Trescot AM, Datta S, Lee M, Hansen H (2008) Opioid pharmacology. Pain Physician 11:S133–53
Mystakidou K, Tsilika E, Parpa E, Kouloulias V, Kouvaris I, Georgaki S et al (2003) Long-term cancer pain management in morphine pre-treated and opioid naive patients with transdermal fentanyl. Int J Cancer 107:486–492
Mystakidou K, Parpa E, Tsilika E, Katsouda E, Kouloulias V, Kouvaris J et al (2004) Pain management of cancer patients with transdermal fentanyl: a study of 1828 step I, II, & III transfers. J Pain 5:119–132
van Seventer R, Smit JM, Schipper RM, Wicks MA, Zuurmond WW (2003) Comparison of TTS-fentanyl with sustained-release oral morphine in the treatment of patients not using opioids for mild-to-moderate pain. Curr Med Res Opin 19:457–469
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Hoya, Y., Okamoto, T. & Yanaga, K. Evaluation of analgesic effect and safety of fentanyl transdermal patch for cancer pain as the first line. Support Care Cancer 18, 761–764 (2010). https://doi.org/10.1007/s00520-010-0869-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00520-010-0869-y