Key message
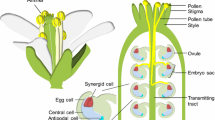
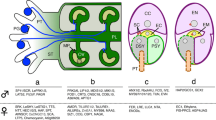
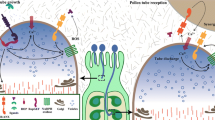
Overview of pollen development.
Abstract
Male gametophyte development of angiosperms is a complex process that requires coordinated activity of different cell types and tissues of both gametophytic and sporophytic origin and the appropriate specific gene expression. Pollen ontogeny is also an excellent model for the dissection of cellular networks that control cell growth, polarity, cellular differentiation and cell signaling. This article describes two sequential phases of angiosperm pollen ontogenesis—developmental phase leading to the formation of mature pollen grains, and a functional or progamic phase, beginning with the impact of the grains on the stigma surface and ending at double fertilization. Here we present an overview of important cellular processes in pollen development and explosive pollen tube growth stressing the importance of reserves accumulation and mobilization and also the mutual activation of pollen tube and pistil tissues, pollen tube guidance and the communication between male and female gametophytes. We further describe the recent advances in regulatory mechanisms involved such as posttranscriptional regulation (including mass transcript storage) and posttranslational modifications to modulate protein function, intracellular metabolic signaling, ionic gradients such as Ca2+ and H+ ions, cell wall synthesis, protein secretion and intercellular signaling within the reproductive tissues.



Similar content being viewed by others
References
Agudelo CG, Sanati Nezhad A, Ghanbari M, Naghavi M, Packirisamy M, Geitmann A (2013) TipChip: a modular, MEMS-based platform for experimentation and phenotyping of tip-growing cells. Plant J 73:1057–1068. doi:10.1111/tpj.12093
Allwood EG, Anthony RG, Smertenko AP, Reichelt S, Drobak BK, Doonan JH, Weeds AG, Hussey PJ (2002) Regulation of the pollen-specific actin-depolymerizing factor LIADF1. Plant Cell 14:2915–2927. doi:10.1105/tpc.005363
Arata H, Higashiyama T (2014) Poly(dimethylsiloxane)-based microdevices for studying plant reproduction. Biochem Soc Trans 42:320–324. doi:10.1042/bst20130258
Ariizumi T, Toriyama K (2011) Genetic regulation of sporopollenin synthesis and pollen exine development. In: Merchant SS, Briggs WR, Ort D (eds) Annual Review of Plant Biology, vol 62, pp 437–460
Barnabas B, Fridvalszky L (1984) Adhesion and germination of differently treated maize pollen grains on the stigma. Acta Bot Hung 30:329–332
Beale KM, Leydon AR, Johnson MA (2012) Gamete fusion is required to block multiple pollen tubes from entering an Arabidopsis ovule. Curr Biol 22:1090–1094. doi:10.1016/j.cub.2012.04.041
Bedinger P (1992) The remarkable biology of pollen. Plant Cell 4:879–887
Bendtsen JD, Jensen LJ, Blom N, von Heijne G, Brunak S (2004) Feature-based prediction of non-classical and leaderless protein secretion. Protein Eng Des Sel 17:349–356. doi:10.1093/protein/gzh037
Berger F, Twell D (2011) Germline specification and function in plants. Annu Rev Plant Biol 62:461–484. doi:10.1146/annurev-arplant-042110-103824
Bishop AL, Hall A (2000) Rho GTPases and their effector proteins. Biochem J 348:241–255. doi:10.1042/0264-6021:3480241
Bleckmann A, Alter S, Dresselhaus T (2014) The beginning of a seed: regulatory mechanisms of double fertilization. Front Plant Sci 5:452. doi:10.3389/fpls.2014.00452
Bodenmiller B, Mueller LN, Mueller M, Domon B, Aebersold R (2007) Reproducible isolation of distinct, overlapping segments of the phosphoproteome. Nat Methods 4:231–237. doi:10.1038/nmeth1005
Bokvaj P, Hafidh S, Honys D (2015) Transcriptome profiling of male gametophyte development Nicotiana tabacum. Genom Data 3:106–111
Borg M, Brownfield L, Twell D (2009) Male gametophyte development: a molecular perspective. J Exp Bot 60:1465–1478. doi:10.1093/jxb/ern355
Borg M, Brownfield L, Khatab H, Sidorova A, Lingaya M, Twell D (2011) The R2R3 MYB transcription factor DUO1 activates a male germline-specific regulon essential for sperm cell differentiation in Arabidopsis. Plant Cell 23:534–549. doi:10.1105/tpc.110.081059
Borges F, Gomes G, Gardner R, Moreno N, McCormick S, Feijó JA, Becker JD (2008) Comparative transcriptomics of Arabidopsis sperm cells. Plant Physiol 148:1168–1181. doi:10.1104/pp.108.125229
Brett C, Waldron K (1990) Physiology and biochemistry of plant cell walls. Unwin Hyman, London
Brewbaker JL (1967) Distribution and phylogenetic significance of binucleate and trinucleate pollen grains in angiosperms. Am J Bot 54:1069–1083. doi:10.2307/2440530
Camacho L, Malho R (2003) Endo/exocytosis in the pollen tube apex is differentially regulated by Ca2+ and GTPases. J Exp Bot 54:83–92. doi:10.1093/jxb/erg043
Cannon MC, Terneus K, Hall Q, Tan L, Wang Y, Wegenhart BL, Chen L, Lamport DT, Chen Y, Kieliszewski MJ (2008) Self-assembly of the plant cell wall requires an extensin scaffold. Proc Natl Acad Sci USA 105:2226–2231. doi:10.1073/pnas.0711980105
Čapková V, Hrabětová E, Tupý J (1988) Protein synthesis in pollen tubes: preferential formation of new species independent of transcription. Sex Plant Reprod 1:150–155
Čapková V, Fidlerová A, van Amstel T, Croes AF, Mata C, Schrauwen JAM, Wullems GJ, Tupý J (1997) Role of N-glycosylation of 66 and 69 kDa glycoproteins in wall formation during pollen tube growth in vitro. Eur J Cell Biol 72:282–285
Capron A, Gourgues M, Neiva LS, Faure JE, Berger F, Pagnussat G, Krishnan A, Alvarez-Mejia C, Vielle-Calzada JP, Lee YR, Liu B, Sundaresan V (2008) Maternal control of male-gamete delivery in Arabidopsis involves a putative GPI-anchored protein encoded by the LORELEI gene. Plant Cell 20:3038–3049. doi:10.1105/tpc.108.061713
Chae K, Lord EM (2011) Pollen tube growth and guidance: roles of small, secreted proteins. Ann Bot 108:627–636. doi:10.1093/aob/mcr015
Chaturvedi P, Ischebeck T, Egelhofer V, Lichtscheidl I, Weckwerth W (2013) Cell-specific analysis of the tomato pollen proteome from pollen mother cell to mature pollen provides evidence for developmental priming. J Proteome Res 12:4892–4903. doi:10.1021/pr400197p
Chen XY, Kim JY (2009) Callose synthesis in higher plants. Plant Signal Behav 4:489–492
Chen CY, Wong EI, Vidali L, Estavillo A, Hepler PK, Wu HM, Cheung AY (2002) The regulation of actin organization by actin-depolymerizing factor in elongating pollen tubes. Plant Cell 14:2175–2190. doi:10.1105/tpc.003038
Chen YH, Li HJ, Shi DQ, Yuan L, Liu J, Sreenivasan R, Baskar R, Grossniklaus U, Yang WC (2007) The central cell plays a critical role in pollen tube guidance in Arabidopsis. Plant Cell 19:3563–3577. doi:10.1105/tpc.107.053967
Chen Y, Liu P, Hoehenwarter W, Lin J (2012) Proteomic and phosphoproteomic analysis of Picea wilsonii pollen development under nutrient limitation. J Proteome Res 11:4180–4190. doi:10.1021/pr300295m
Cheung AY, Boavida LC, Aggarwal M, Wu HM, Feijó JA (2010) The pollen tube journey in the pistil and imaging the in vivo process by two-photon microscopy. J Exp Bot 61:1907–1915. doi:10.1093/jxb/erq062
Choudhary P, Saha P, Ray T, Tang Y, Yang D, Cannon MC (2015) EXTENSIN18 is required for full male fertility as well as normal vegetative growth in Arabidopsis. Front Plant Sci 6:553. doi:10.3389/fpls.2015.00553
de Graaf BHJ, Cheung AY, Andreyeva T, Levasseur K, Kieliszewski M, Wu HM (2005) Rab11 GTPase-regulated membrane trafficking is crucial for tip-focused pollen tube growth in tobacco. Plant Cell 17:2564–2579. doi:10.1105/tpc.105.033183
De Storme N, Geelen D (2013) Cytokinesis in plant male meiosis. Plant Signal Behav 8:e23394. doi:10.4161/psb.23394
Dobritsa AA, Coerper D (2012) The novel plant protein INAPERTURATE POLLEN1 marks distinct cellular domains and controls formation of apertures in the Arabidopsis pollen exine. Plant Cell 24:4452–4464. doi:10.1105/tpc.112.101220
Dobritsa AA, Shrestha J, Morant M, Pinot F, Matsuno M, Swanson R, Moller BL, Preuss D (2009) CYP704B1 is a long-chain fatty acid omega-hydroxylase essential for sporopollenin synthesis in pollen of Arabidopsis. Plant Physiol 151:574–589. doi:10.1104/pp.109.144469
Dobritsa AA, Lei Z, Nishikawa S, Urbanczyk-Wochniak E, Huhman DV, Preuss D, Sumner LW (2010) LAP5 and LAP6 encode anther-specific proteins with similarity to chalcone synthase essential for pollen exine development in Arabidopsis. Plant Physiol 153:937–955. doi:10.1104/pp.110.157446
Dobritsa AA, Geanconteri A, Shrestha J, Carlson A, Kooyers N, Coerper D, Urbanczyk-Wochniak E, Bench BJ, Sumner LW, Swanson R, Preuss D (2011) A large-scale genetic screen in Arabidopsis to identify genes involved in pollen exine production. Plant Physiol 157:947–970. doi:10.1104/pp.111.179523
Dresselhaus T, Franklin-Tong N (2013) Male–female crosstalk during pollen germination, tube growth and guidance, and double fertilization. Mol Plant 6:1018–1036. doi:10.1093/mp/sst061
Dresselhaus T, Sprunck S (2012) Plant fertilization: maximizing reproductive success. Curr Biol 22:R487–R489. doi:10.1016/j.cub.2012.04.048
Dunn JD, Reid GE, Bruening ML (2010) Techniques for phosphopeptide enrichment prior to analysis by mass spectrometry. Mass Spectrom Rev 29:29–54. doi:10.1002/mas.20219
Dupľáková N, Reňák D, Hovanec P, Honysová B, Twell D, Honys D (2007) Arabidopsis Gene Family Profiler (aGFP): user-oriented transcriptomic database with easy-to-use graphic interface. BMC Plant Biol 7:39. doi:10.1186/1471-2229-7-39
Durek P, Schmidt R, Heazlewood JL, Jones A, MacLean D, Nagel A, Kersten B, Schulze WX (2010) PhosPhAt: the Arabidopsis thaliana phosphorylation site database: an update. Nucleic Acids Res 38:D828–D834. doi:10.1093/nar/gkp810
Eady C, Lindsey K, Twell D (1995) The significance of microspore division and division symmetry for vegetative cell-specific transcription and generative cell differentiation. Plant Cell 7:65–74. doi:10.1105/tpc.7.1.65
Eisenhaber B, Wildpaner M, Schultz CJ, Borner GH, Dupree P, Eisenhaber F (2003) Glycosylphosphatidylinositol lipid anchoring of plant proteins. Sensitive prediction from sequence- and genome-wide studies for Arabidopsis and rice. Plant Physiol 133:1691–1701. doi:10.1104/pp.103.023580
Elfving F (1879) Studien über die Pollenkörner der Angiospermen. Jenaische Zeitschrift für Naturwissenschaft 13:1–28
Ellis M, Egelund J, Schultz CJ, Bacic A (2010) Arabinogalactan-proteins: key regulators at the cell surface? Plant Physiol 153:403–419. doi:10.1104/pp.110.156000
Feher A, Lajko DB (2015) Signals fly when kinases meet Rho-of-plants (ROP) small G-proteins. Plant Sci 237:93–107. doi:10.1016/j.plantsci.2015.05.007
Feijó JA, Sainhas J, Hackett GR, Kunkel JG, Hepler PK (1999) Growing pollen tubes possess a constitutive alkaline band in the clear zone and a growth-dependent acidic tip. J Cell Biol 144:483–496. doi:10.1083/jcb.144.3.483
Feijó JA, Costa SS, Prado AM, Becker JD, Certal AC (2004) Signalling by tips. Curr Opin Plant Biol 7:589–598. doi:10.1016/j.pbi.2004.07.014
Fellenberg C, Vogt T (2015) Evolutionarily conserved phenylpropanoid pattern on angiosperm pollen. Trends Plant Sci 20:212–218. doi:10.1016/j.tplants.2015.01.011
Ferguson C, Teeri TT, Siika-aho M, Read SM, Bacic A (1998) Location of cellulose and callose in pollen tubes and grains of Nicotiana tabacum. Planta 206:452–460. doi:10.1007/s004250050421
Fidlerová A, Smýkal P, Tupý J, Čapková V (2001) Glycoproteins 66 and 69 kDa of pollen tube wall: properties and distribution in angiosperms. J Plant Physiol 158:1367–1374. doi:10.1078/0176-1617-00562
Fíla J, Honys D (2012) Enrichment techniques employed in phosphoproteomics. Amino Acids 43:1025–1047. doi:10.1007/s00726-011-1111-z
Fíla J, Čapková V, Feciková J, Honys D (2011) Impact of homogenization and protein extraction conditions on the obtained tobacco pollen proteomic patterns. Biol Plant 55:499–506. doi:10.1007/s10535-011-0116-5
Fíla J, Matros A, Radau S, Zahedi RP, Čapková V, Mock H-P, Honys D (2012) Revealing phosphoproteins playing role in tobacco pollen activated in vitro. Proteomics 12:3229–3250. doi:10.1002/pmic.201100318
Fíla J, Čapková V, Honys D (2014) Phosphoproteomic studies in Arabidopsis and tobacco male gametophytes. Biochem Soc Trans 42:383–387. doi:10.1042/bst20130249
Firon N, Nepi M, Pacini E (2012) Water status and associated processes mark critical stages in pollen development and functioning. Ann Bot 109:1201–1213. doi:10.1093/aob/mcs070
Fu Y, Wu G, Yang ZB (2001) Rop GTPase-dependent dynamics of tip-localized F-actin controls tip growth in pollen tubes. J Cell Biol 152:1019–1032. doi:10.1083/jcb.152.5.1019
Furness CA, Rudall PJ (2004) Pollen aperture evolution: a crucial factor for eudicot success? Trends Plant Sci 9:154–158. doi:10.1016/j.tplants.2004.01.001
Gaillard A, Vergne P, Beckert M (1991) Optimization of maize microspore isolation and culture conditions for reliable plant regeneration. Plant Cell Rep 10:55–58. doi:10.1007/BF00236456
Grobei MA, Qeli E, Brunner E, Rehrauer H, Zhang R, Roschitzki B, Basler K, Ahrens CH, Grossniklaus U (2009) Deterministic protein inference for shotgun proteomics data provides new insights into Arabidopsis pollen development and function. Genome Res 19:1786–1800. doi:10.1101/gr.089060.108
Guan Y, Guo J, Li H, Yang Z (2013) Signaling in pollen tube growth: crosstalk, feedback, and missing links. Mol Plant 6:1053–1064. doi:10.1093/mp/sst070
Hafidh S, Čapková V, Honys D (2011) Safe keeping the message: mRNP complexes tweaking after transcription. Adv Exp Med Biol 722:118–136. doi:10.1007/978-1-4614-0332-6_8
Hafidh S, Breznenová K, Honys D (2012a) De novo post-pollen mitosis II tobacco pollen tube transcriptome. Plant Signal Behav 7:918–921. doi:10.4161/psb.20745
Hafidh S, Breznenová K, Růžička P, Feciková J, Čapková V, Honys D (2012b) Comprehensive analysis of tobacco pollen transcriptome unveils common pathways in polar cell expansion and underlying heterochronic shift during spermatogenesis. BMC Plant Biol 12:24. doi:10.1186/1471-2229-12-24
Hafidh S, Potěšil D, Fíla J, Feciková J, Čapková V, Zdráhal Z, Honys D (2014) In search of ligands and receptors of the pollen tube: the missing link in pollen tube perception. Biochem Soc Trans 42:388–394. doi:10.1042/BST20130204
Hamamura Y, Saito C, Awai C, Kurihara D, Miyawaki A, Nakagawa T, Kanaoka MM, Sasaki N, Nakano A, Berger F, Higashiyama T (2011) Live-cell imaging reveals the dynamics of two sperm cells during double fertilization in Arabidopsis thaliana. Curr Biol 21:497–502. doi:10.1016/j.cub.2011.02.013
Hamamura Y, Nagahara S, Higashiyama T (2012) Double fertilization on the move. Curr Opin Plant Biol 15:70–77. doi:10.1016/j.pbi.2011.11.001
Hanisch FA (2001) O-glycosylation of the mucin type. Biol Chem 382:143–149. doi:10.1515/bc.2001.022
Heazlewood JL, Durek P, Hummel J, Selbig J, Weckwerth W, Walther D, Schulze WX (2008) PhosPhAt: a database of phosphorylation sites in Arabidopsis thaliana and a plant-specific phosphorylation site predictor. Nucleic Acids Res 36:D1015–D1021. doi:10.1093/nar/gkm812
Hepler PK, Winship LJ (2015) The pollen tube clear zone: clues to the mechanism of polarized growth. J Integr Plant Biol 57:79–92. doi:10.1111/jipb.12315
Heslop-Harrison J (1987) Pollen germination and pollen-tube growth. In: Giles KL, Prakash J (eds) Pollen: cytology and development. Academic Press, London, pp 1–78
Heslop-Harrison J, Heslop-Harrison Y (1986) Pollen-tube chemotropism: fact or delusion? In: Cresti M, Romano D (eds) In biology of reproduction and cell motility in plants and animals. University of Sienna Press, Siena, pp 169–174
Heslop-Harrison J, Heslop-Harrison Y, Cresti M, Tiezzi A, Moscatelli A (1988) Cytoskeletal elements, cell shaping and movement in the angiosperm pollen tube. J Cell Sci 91:49–60
Higashiyama T (2010) Peptide signaling in pollen–pistil interactions. Plant Cell Physiol 51:177–189. doi:10.1093/pcp/pcq008
Higashiyama T (2015) The mechanism and key molecules involved in pollen tube guidance. Annu Rev Plant Biol 2015:393–413
Higashiyama T, Kuroiwa H, Kawano S, Kuroiwa T (1998) Guidance in vitro of the pollen tube to the naked embryo sac of Torenia fournieri. Plant Cell 10:2019–2032
Holdaway-Clarke TL, Feijó JA, Hackett GR, Kunkel JG, Hepler PK (1997) Pollen tube growth and the intracellular cytosolic calcium gradient oscillate in phase while extracellular calcium influx is delayed. Plant Cell 9:1999–2010
Holmes-Davis R, Tanaka CK, Vensel WH, Hurkman WJ, McCormick S (2005) Proteome mapping of mature pollen of Arabidopsis thaliana. Proteomics 5:4864–4884. doi:10.1002/pmic.200402011
Honys D, Twell D (2003) Comparative analysis of the Arabidopsis pollen transcriptome. Plant Physiol 132:640–652. doi:10.1104/pp.103.020925
Honys D, Twell D (2004) Transcriptome analysis of haploid male gametophyte development in Arabidopsis. Genome Biol. doi:10.1186/gb-2004-5-11-r85
Honys D, Combe JP, Twell D, Čapková V (2000) The translationally repressed pollen-specific ntp303 mRNA is stored in non-polysomal mRNPs during pollen maturation. Sex Plant Reprod 13:135–144
Honys D, Reňák D, Twell D (2006) Male gametyphyte development and function. In: da Silva JT (ed) Floriculture, ornamental and plant biotechnology: advances and topical issues, vol 1. Global Science Books, London, pp 76–87
Honys D, Reňák D, Feciková J, Jedelský PL, Nebesářová J, Dobrev P, Čapková V (2009) Cytoskeleton-associated large RNP complexes in tobacco male gametophyte (EPPs) are associated with ribosomes and are involved in protein synthesis, processing, and localization. J Proteome Res 8:2015–2031. doi:10.1021/pr8009897
Horade M, Kanaoka M, Kuzuya M, Higashiyama T, Kaji N (2013) A microfluidic device for quantitative analysis of chemoattraction in plants. RSC Adv 3:22301–22307
Hruz T, Laule O, Szabo G, Wessendorp F, Bleuler S, Oertle L, Widmayer P, Gruissem W, Zimmermann P (2008) Genevestigator v3: a reference expression database for the meta-analysis of transcriptomes. Adv Bioinform 2008:420747. doi:10.1155/2008/420747
Hsieh K, Huang AH (2005) Lipid-rich tapetosomes in Brassica tapetum are composed of oleosin-coated oil droplets and vesicles, both assembled in and then detached from the endoplasmic reticulum. Plant J 43:889–899. doi:10.1111/j.1365-313X.2005.02502.x
Huang MD, Hsing YI, Huang AH (2011) Transcriptomes of the anther sporophyte: availability and uses. Plant Cell Physiol 52:1459–1466. doi:10.1093/pcp/pcr088
Huck N, Moore JM, Federer M, Grossniklaus U (2003) The Arabidopsis mutant feronia disrupts the female gametophytic control of pollen tube reception. Development 130:2149–2159
Hulskamp M, Schneitz K, Pruitt RE (1995) Genetic evidence for a long-range activity that directs pollen tube guidance in Arabidopsis. Plant Cell 7:57–64. doi:10.1105/tpc.7.1.57
Ischebeck T, Valledor L, Lyon D, Gingl S, Nagler M, Meijon M, Egelhofer V, Weckwerth W (2014) Comprehensive cell-specific protein analysis in early and late pollen development from diploid microsporocytes to pollen tube growth. Mol Cell Proteomics 13:295–310. doi:10.1074/mcp.M113.028100
Jia Q-S, Zhu J, Xu XF, Lou Y, Zhang ZL, Zhang ZP, Yang ZN (2014) Arabidopsis AT-hook protein TEK positively regulates the expression of arabinogalactan proteins for Nexine formation. Mol Plant 8:251–260
Kanaoka MM, Kawano N, Matsubara Y, Susaki D, Okuda S, Sasaki N, Higashiyama T (2011) Identification and characterization of TcCRP1, a pollen tube attractant from Torenia concolor. Ann Bot 108:739–747. doi:10.1093/aob/mcr111
Kasahara RD, Portereiko MF, Sandaklie-Nikolova L, Rabiger DS, Drews GN (2005) MYB98 is required for pollen tube guidance and synergid cell differentiation in Arabidopsis. Plant Cell 17:2981–2992. doi:10.1105/tpc.105.034603
Kasahara RD, Maruyama D, Hamamura Y, Sakakibara T, Twell D, Higashiyama T (2012) Fertilization recovery after defective sperm cell release in Arabidopsis. Curr Biol 22:1084–1089. doi:10.1016/j.cub.2012.03.069
Kessler SA, Grossniklaus U (2011) She’s the boss: signaling in pollen tube reception. Curr Opin Plant Biol 14:622–627. doi:10.1016/j.pbi.2011.07.012
Klahre U, Becker C, Schmitt AC, Kost B (2006) Nt-RhoGDI2 regulates Rac/Rop signaling and polar cell growth in tobacco pollen tubes. Plant J 46:1018–1031. doi:10.1111/j.1365-313X.2006.02757.x
Knorre DG, Kudryashova NV, Godovikova TS (2009) Chemical and functional aspects of posttranslational modification of proteins. Acta Nat 1:29–51
Kovar DR, Drobak BK, Staiger CJ (2000) Maize profilin isoforms are functionally distinct. Plant Cell 12:583–598
Kultz D (1998) Phylogenetic and functional classification of mitogen- and stress-activated protein kinases. J Mol Evol 46:571–588. doi:10.1007/pl00006338
Lalanne E, Honys D, Johnson A, Borner GH, Lilley KS, Dupree P, Grossniklaus U, Twell D (2004) SETH1 and SETH2, two components of the glycosylphosphatidylinositol anchor biosynthetic pathway, are required for pollen germination and tube growth in Arabidopsis. Plant Cell 16:229–240. doi:10.1105/tpc.014407
Lamport DT, Varnai P (2013) Periplasmic arabinogalactan glycoproteins act as a calcium capacitor that regulates plant growth and development. N Phytol 197:58–64. doi:10.1111/nph.12005
Lamport DT, Kieliszewski MJ, Chen Y, Cannon MC (2011) Role of the extensin superfamily in primary cell wall architecture. Plant Physiol 156:11–19. doi:10.1104/pp.110.169011
Lancelle SA, Hepler PK (1992) Ultrastructure of freeze-substituted pollen tubes of Lilium longiferum. Protoplasma 167:215–230. doi:10.1007/bf01403385
Lee TY, Bretana NA, Lu CT (2011) PlantPhos: using maximal dependence decomposition to identify plant phosphorylation sites with substrate site specificity. BMC Bioinform 12:13. doi:10.1186/1471-2105-12-261
Lee JS, Hnilova M, Maes M, Lin YC, Putarjunan A, Han SK, Avila J, Torii KU (2015) Competitive binding of antagonistic peptides fine-tunes stomatal patterning. Nature 522:439–443. doi:10.1038/nature14561
Lenartowska M, Michalska A (2008) Actin filament organization and polarity in pollen tubes revealed by myosin II subfragment 1 decoration. Planta 228:891–896. doi:10.1007/s00425-008-0802-5
Lerouge P, Cabanes-Macheteau M, Rayon C, Fischette-Laine AC, Gomord V, Faye L (1998) N-glycoprotein biosynthesis in plants: recent developments and future trends. Plant Mol Biol 38:31–48. doi:10.1023/a:1006012005654
Leydon AR, Beale KM, Woroniecka K, Castner E, Chen J, Horgan C, Palanivelu R, Johnson MA (2013) Three MYB transcription factors control pollen tube differentiation required for sperm release. Curr Biol 23:1209–1214. doi:10.1016/j.cub.2013.05.021
Li HJ, Xue Y, Jia DJ, Wang T, Hi DQ, Liu J, Cui F, Xie Q, Ye D, Yang WC (2011) POD1 regulates pollen tube guidance in response to micropylar female signaling and acts in early embryo patterning in Arabidopsis. Plant Cell 23:3288–3302. doi:10.1105/tpc.111.088914
Li S, Ge FR, Xu M, Zhao XY, Huang GQ, Zhou LZ, Wang JG, Kombrink A, McCormick S, Zhang XS, Zhang Y (2013) Arabidopsis COBRA-LIKE 10, a GPI-anchored protein, mediates directional growth of pollen tubes. Plant J 74:486–497. doi:10.1111/tpj.12139
Li Y, Ye Z, Nie Y, Zhang J, Wang G-L, Wang Z (2015) Comparative phosphoproteome analysis of Magnaporthe oryzae-responsive proteins in susceptible and resistant rice cultivars. J Proteomics 115:66–80. doi:10.1016/j.jprot.2014.12.007
Lim ES, Gumpil JS (1984) The flowering, pollination and hybridization of groundnuts (Arachis hypogaea L.). Pertanika 7:61–66
Lindner H, Kessler SA, Muller LM, Shimosato-Asano H, Boisson-Dernier A, Grossniklaus U (2015) TURAN and EVAN mediate pollen tube reception in Arabidopsis synergids through protein glycosylation. PLoS Biol. doi:10.1371/journal.pbio.1002139
Ling Y, Chen T, Jing Y, Fan L, Wan Y, Lin J (2013) γ-Aminobutyric acid (GABA) homeostasis regulates pollen germination and polarized growth in Picea wilsonii. Planta 238:831–843. doi:10.1007/s00425-013-1938-5
Liu J, Zhong S, Guo X, Hao L, Wei X, Huang Q, Hou Y, Shi J, Wang C, Gu H, Qu LJ (2013) Membrane-bound RLCKs LIP1 and LIP2 are essential male factors controlling male–female attraction in Arabidopsis. Curr Biol 23:993–998. doi:10.1016/j.cub.2013.04.043
Lora J, Herrero M, Hormaza JI (2009) The coexistence of bicellular and tricellular pollen in Annona cherimola (Annonaceae): implications for pollen evolution. Am J Bot 96:802–808. doi:10.3732/ajb.0800167
Lou Y, Xu XF, Zhu J, Gu JN, Blackmore S, Yang ZN (2014) The tapetal AHL family protein TEK determines nexine formation in the pollen wall. Nat Commun 5:3855. doi:10.1038/ncomms4855
Lovy-Wheeler A, Kunkel JG, Allwood EG, Hussey PJ, Hepler PK (2006) Oscillatory increases in alkalinity anticipate growth and may regulate actin dynamics in pollen tubes of lily. Plant Cell 18:2182–2193. doi:10.1105/tpc.106.044867
Lu Y, Chanroj S, Zulkifli L, Johnson MA, Uozumi N, Cheung A, Sze H (2011) Pollen tubes lacking a pair of K+ transporters fail to target ovules in Arabidopsis. Plant Cell 23:81–93. doi:10.1105/tpc.110.080499
Lu P, Chai M, Yang J, Ning G, Wang G, Ma H (2014) The Arabidopsis CALLOSE DEFECTIVE MICROSPORE1 gene is required for male fertility through regulating callose metabolism during microsporogenesis. Plant Physiol 164:1893–1904. doi:10.1104/pp.113.233387
Ma H (2005) Molecular genetic analyses of microsporogenesis and micro grametogenesis in flowering plants. Annu Rev Plant Biol 2005:393–434
Maheshwari P (1950) An introduction to embryology of angiosperms. McGraw-Hill, New York
Malpighi M (1675, 1679) Die Anatomie der Pflanzen. I und II Theil. London 1675 und 1679. Engelmann, Leipzig, 1901
Márton ML, Cordts S, Broadhvest J, Dresselhaus T (2005) Micropylar pollen tube guidance by egg apparatus 1 of maize. Science 307:573–576. doi:10.1126/science.1104954
Márton ML, Fastner A, Uebler S, Dresselhaus T (2012) Overcoming hybridization barriers by the secretion of the maize pollen tube attractant ZmEA1 from Arabidopsis ovules. Curr Biol 22:1194–1198. doi:10.1016/j.cub.2012.04.061
Maruyama D, Hamamura Y, Takeuchi H, Susaki D, Nishimaki M, Kurihara D, Kasahara RD, Higashiyama T (2013) Independent control by each female gamete prevents the attraction of multiple pollen tubes. Dev Cell 25:317–323. doi:10.1016/j.devcel.2013.03.013
Mascarenhas JP (1993) Molecular mechanisms of pollen tube growth and differentiation. Plant Cell 5:1303–1314. doi:10.1105/tpc.5.10.1303
Mascarenhas JP, Machlis L (1962) The hormonal control of the directional growth of pollen tubes. Vitam Horm 20:347–372
Mayank P, Grossman J, Wuest S, Boisson-Dernier A, Roschitzki B, Nanni P, Nühse T, Grossniklaus U (2012) Characterization of the phosphoproteome of mature Arabidopsis pollen. Plant J 72:89–101. doi:10.1111/j.1365-313X.2012.05061.x
McCue AD, Cresti M, Feijó JA, Slotkin RK (2011) Cytoplasmic connection of sperm cells to the pollen vegetative cell nucleus: potential roles of the male germ unit revisited. J Exp Bot 62:1621–1631. doi:10.1093/jxb/err032
Meyer LJ, Gao J, Xu D, Thelen JJ (2012) Phosphoproteomic analysis of seed maturation in Arabidopsis, rapeseed, and soybean. Plant Physiol 159:517–528. doi:10.1104/pp.111.191700
Michard E, Dias P, Feijó JA (2008) Tobacco pollen tubes as cellular models for ion dynamics: improved spatial and temporal resolution of extracellular flux and free cytosolic concentration of calcium and protons using pHluorin and YC3.1 CaMeleon. Sex Plant Reprod 21:169–181. doi:10.1007/s00497-008-0076-x
Michard E, Alves F, Feijó JA (2009) The role of ion fluxes in polarized cell growth and morphogenesis: the pollen tube as an experimental paradigm. Intern J Dev Biol 53:1609–1622. doi:10.1387/ijdb.072296em
Mogami N, Miyamoto M, Onozuka M, Nakamura N (2006) Comparison of callose plug structure between dicotyledon and monocotyledon pollen germinated in vitro. Grana 45:249–256. doi:10.1080/00173130600726687
Mori T, Kuroiwa H, Higashiyama T, Kuroiwa T (2005) GENERATIVE CELL SPECIFIC 1 is essential for angiosperm fertilization. Nat cell biol 8:64–71
Mulcahy DL (1979) The rise of the angiosperms: a genecological factor. Science 206:20–23. doi:10.1126/science.206.4414.20
Mulcahy GB, Mulcahy DL (1988) The effect of supplemented media on the growth in vitro of binucleate and trinucleate pollen. Plant Sci 55:213–216
Mulcahy DL, Sari Gorla M, Mulcahy GB (1996) Pollen selection: past, present and future. Sex Plant Reprod 9:353–356
Nawaschin S (1898) Resultate einer Revision der Befruchtungsvorgänge bei Lilium martagon und Fritillaria tenella. Bulletin de l’Académie Impériale des Sciences 9:377–382
Nguema-Ona E, Coimbra S, Vicre-Gibouin M, Mollet J-C, Driouich A (2012) Arabinogalactan proteins in root and pollen-tube cells: distribution and functional aspects. Ann Bot 110:383–404. doi:10.1093/aob/mcs143
Noir S, Bräutigam A, Colby T, Schmidt J, Panstruga R (2005) A reference map of the Arabidopsis thaliana mature pollen proteome. Biochem Biophys Res Commun 337:1257–1266. doi:10.1016/j.bbrc.2005.09.185
Oh SA, Pal MD, Park SK, Johnson JA, Twell D (2010) The tobacco MAP215/Dis1-family protein TMBP200 is required for the functional organization of microtubule arrays during male germline establishment. J Exp Bot 61:969–981. doi:10.1093/jxb/erp367
Okuda S, Tsutsui H, Shiina K, Sprunck S, Takeuchi H, Yui R, Kasahara RD, Hamamura Y, Mizukami A, Susaki D, Kawano N, Sakakibara T, Namiki S, Itoh K, Otsuka K, Matsuzaki M, Nozaki H, Kuroiwa T, Nakano A, Kanaoka MM, Dresselhaus T, Sasaki N, Higashiyama T (2009) Defensin-like polypeptide LUREs are pollen tube attractants secreted from synergid cells. Nature 458:357–361. doi:10.1038/nature07882
Onelli E, Idilli AI, Moscatelli A (2015) Emerging roles for microtubules in angiosperm pollen tube growth highlight new research cues. Front Plant Sci. doi:10.3389/fpls.2015.00051
Pacini E (1990) Tapetum and microspore function. In: Blackmore S, Knox RB (eds) Microspores: evolution and ontogeny. Academic Press, London, pp 213–237
Pacini E (1996) Types and meaning of pollen carbohydrate reserves. Sex Plant Reprod 9:362–366
Pacini E, Guarnieri M, Nepi M (2006) Pollen carbohydrates and water content during development, presentation, and dispersal: a short review. Protoplasma 228:73–77. doi:10.1007/s00709-006-0169-z
Palanivelu R, Preuss D (2000) Pollen tube targeting and axon guidance: parallels in tip growth mechanisms. Trends Cell Biol 10:517–524. doi:10.1016/s0962-8924(00)01849-3
Palanivelu R, Preuss D (2006) Distinct short-range ovule signals attract or repel Arabidopsis thaliana pollen tubes in vitro. BMC Plant Biol 6:7. doi:10.1186/1471-2229-6-7
Palanivelu R, Brass L, Edlund AF, Preuss D (2003) Pollen tube growth and guidance is regulated by POP2, an Arabidopsis gene that controls GABA levels. Cell 114:47–59
Park SK, Howden R, Twell D (1998) The Arabidopsis thaliana gametophytic mutation gemini pollen1 disrupts microspore polarity, division asymmetry and pollen cell fate. Development 125:3789–3799
Pereira AM, Masiero S, Nobre MS, Costa ML, Solis MT, Testillano PS, Sprunck S, Coimbra S (2014) Differential expression patterns of arabinogalactan proteins in Arabidopsis thaliana reproductive tissues. J Exp Bot 65:5459–5471
Petersen TN, Brunak S, von Heijne G, Nielsen H (2011) SignalP 4.0: discriminating signal peptides from transmembrane regions. Nat Methods 8:785–786. doi:10.1038/nmeth.1701
Phan HA, Iacuone S, Li SF, Parish RW (2011) The MYB80 transcription factor is required for pollen development and the regulation of tapetal programmed cell death in Arabidopsis thaliana. Plant Cell 23:2209–2224. doi:10.1105/tpc.110.082651
Pierloni A, Martelli PL, Casadio R (2008) PredGPI: a GPI-anchor predictor. BMC Bioinform 9:392. doi:10.1186/1471-2105-9-392
Pierson ES, Miller DD, Callaham DA, van Aken J, Hackett G, Hepler PK (1996) Tip-localized calcium entry fluctuates during pollen tube growth. Dev Biol 174:160–173. doi:10.1006/dbio.1996.0060
Prado N, de Dios Alche J, Casado-Vela J, Mas S, Villalba M, Rodriguez R, Batanero E (2014) Nanovesicles are secreted during pollen germination and pollen tube growth: a possible role in fertilization. Mol Plant 7:573–577. doi:10.1093/mp/sst153
Puc M (2003) Characterisation of pollen allergens. Ann Agric Environ Med 10:143–149
Purkyně JE (1830) De cellulis antherarum fibrosis nec non de granorum pollinarium formis: commentatio phytomica. Sumtibus J. D. Gruesonii, Breslau, Vratislaviae
Qin Y, Yang ZBA (2011) Rapid tip growth: insights from pollen tubes. Semin Cell Dev Biol 22:816–824. doi:10.1016/j.semcdb.2011.06.004
Qin Y, Leydon AR, Manziello A, Pandey R, Mount D, Denic S, Vasic B, Johnson MA, Palanivelu R (2009) Penetration of the stigma and style elicits a novel transcriptome in pollen tubes, pointing to genes critical for growth in a pistil. PLoS Genet. doi:10.1371/journal.pgen.1000621
Qin Y, Wysocki RJ, Somogyi A, Feinstein Y, Franco JY, Tsukamoto T, Dunatunga D, Levy C, Smith S, Simpson R, Gang D, Johnson MA, Palanivelu R (2011) Sulfinylated azadecalins act as functional mimics of a pollen germination stimulant in Arabidopsis pistils. Plant J 68:800–815. doi:10.1111/j.1365-313X.2011.04729.x
Qin T, Liu X, Li J, Sun J, Song L, Mao T (2014) Arabidopsis microtubule-destabilizing protein 25 functions in pollen tube growth by severing actin filaments. Plant Cell 26:325–339. doi:10.1105/tpc.113.119768
Quilichini TD, Grienenberger E, Douglas CJ (2015) The biosynthesis, composition and assembly of the outer pollen wall: a tough case to crack. Phytochemistry 113:170–182. doi:10.1016/j.phytochem.2014.05.002
Ray SM, Park SS, Ray A (1997) Pollen tube guidance by the female gametophyte. Development 124:2489–2498
Ren HY, Xiang Y (2007) The function of actin-binding proteins in pollen tube growth. Protoplasma 230:171–182. doi:10.1007/s00709-006-0231-x
Romagnoli S, Cai G, Faleri C, Yokota E, Shimmen T, Cresti M (2007) Microtubule- and actin filament-dependent motors are distributed on pollen tube mitochondria and contribute differently to their movement. Plant Cell Physiol 48:345–361. doi:10.1093/pcp/pcm001
Rotman N, Rozier F, Boavida L, Dumas C, Berger F, Faure JE (2003) Female control of male gamete delivery during fertilization in Arabidopsis thaliana. Curr Biol 13:432–436
Russell SD, Jones DS (2015) The male germline of angiosperms: repertoire of an inconspicuous but important cell lineage. Front Plant Sci 6:173. doi:10.3389/fpls.2015.00173
Russell SD, Strout GW, Stramski AK, Mislan TW, Thompson RA, Schoemann LM (1996) Development polarization and morphogenesis of the generative and sperm cells of Plumbago zeylanica. 1. Descriptive cytology and three-dimensional organization. Am J Bot 83:1435–1453
Rutley N, Twell D (2015) A decade of pollen transcriptomics. Plant Reprod 28:73–89. doi:10.1007/s00497-015-0261-7
Šamaj J, Muller J, Beck M, Böhm N, Menzel D (2006) Vesicular trafficking, cytoskeleton and signalling in root hairs and pollen tubes. Trends Plant Sci 11:594–600. doi:10.1016/j.tplants.2006.10.002
Sanati Nezhad A, Packirisamy M, Geitmann A (2014) Dynamic, high precision targeting of growth modulating agents is able to trigger pollen tube growth reorientation. Plant J 80:185–195. doi:10.1111/tpj.12613
Sanders LC, Lord EM (1989) Directed movement of latex particles in the gynoecia of three species of flowering plants. Science 243:1606–1608
Sanders LC, Lord EM (1992) A dynamic role for the stylar matrix during pollen tube extension. Int Rev Cytol 140:297–318
Schrauwen JA, de Groot PF, van Herpen MM, van der Lee T, Reynen WH, Weterings KA, Wullems GJ (1990) Stage-related expression of mRNAs during pollen development in lily and tobacco. Planta 182:298–304. doi:10.1007/BF00197125
Schwacke R, Grallath S, Breitkreuz KE, Stransky E, Stransky H, Frommer WB, Rentsch D (1999) LeProT1, a transporter for proline, glycine betaine, and gamma-amino butyric acid in tomato pollen. Plant Cell 11:377–392
Schwemmie J (1968) Selective fertilization in Oenothera. Adv Genet 14:225–324
Scotland RW, Worltey AH (2003) How many species of seed plants are there? Taxon 52:101–104
Scott RJ, Spielman M, Dickinson HG (2004) Stamen structure and function. Plant Cell 16(Suppl):S46–S60. doi:10.1105/tpc.017012
Sheoran IS, Sproule KA, Olson DJH, Ross ARS, Sawhney VK (2006) Proteome profile and functional classification of proteins in Arabidopsis thaliana (Landsberg erecta) mature pollen. Sex Plant Reprod 19:185–196. doi:10.1007/s00497-006-0035-3
Sheoran IS, Ross ARS, Olson DJH, Sawhney VK (2009) Compatibility of plant protein extraction methods with mass spectrometry for proteome analysis. Plant Sci 176:99–104. doi:10.1016/j.plantsci.2008.09.015
Shi YY, Tao WJ, Liang SP, Lu YT, Zhang L (2009) Analysis of the tip-to-base gradient of CaM in pollen tube pulsant growth using in vivo CaM-GFP system. Plant Cell Rep 28:1253–1264. doi:10.1007/s00299-009-0725-z
Shimizu KK, Okada K (2000) Attractive and repulsive interactions between female and male gametophytes in Arabidopsis pollen tube guidance. Development 127:4511–4518
Slotkin RK, Vaughn M, Borges F, Tanurdzic M, Becker JD, Feijo JA, Martienssen RA (2009) Epigenetic reprogramming and small RNA silencing of transposable elements in pollen. Cell 136:461–472. doi:10.1016/j.cell.2008.12.038
Staiger CJ, Poulter NS, Henty JL, Franklin-Tong VE, Blanchoin L (2010) Regulation of actin dynamics by actin-binding proteins in pollen. J Exp Bot 61:1969–1986. doi:10.1093/jxb/erq012
Stepper J, Shastri S, Loo TS, Preston JC, Novak P, Man P, Moore CH, Havlicek V, Patchett ML, Norris GE (2011) Cysteine S-glycosylation, a new post-translational modification found in glycopeptide bacteriocins. FEBS Lett 585:645–650. doi:10.1016/j.febslet.2011.01.023
Strasburger E (1884) Neue Untersuchungen uber den Befruchtungsvorgang bei den Phanerogamen als Grundlage fur eine Theorie der Zeugung. Gustav Fischer, Jena
Szumlanski AL, Nielsen E (2009) The Rab GTPase RabA4d regulates pollen tube tip growth in Arabidopsis thaliana. Plant Cell 21:526–544. doi:10.1105/tpc.108.060277
Takeuchi H, Higashiyama T (2011) Attraction of tip-growing pollen tubes by the female gametophyte. Curr Opin Plant Biol 14:614–621. doi:10.1016/j.pbi.2011.07.010
Takeuchi H, Higashiyama T (2012) A species-specific cluster of defensin-like genes encodes diffusible pollen tube attractants in Arabidopsis. PLoS Biol 10:e1001449. doi:10.1371/journal.pbio.1001449
Terasaka O, Niitsu T (1987) Unequal cell division and chromatin differentiation in pollen grain cells I. Centrifugal, cold and caffeine treatment. Bot Mag (Tokyo) 100:205–216
Ting JT, Wu SS, Ratnayake C, Huang AH (1998) Constituents of the tapetosomes and elaioplasts in Brassica campestris tapetum and their degradation and retention during microsporogenesis. Plant J 16:541–551
Tsukamoto T, Qin Y, Huang Y, Dunatunga D, Palanivelu R (2010) A role for LORELEI, a putative glycosylphosphatidylinositol-anchored protein, in Arabidopsis thaliana double fertilization and early seed development. Plant J 62:571–588. doi:10.1111/j.1365-313X.2010.04177.x
Tupý J (1982) Alterations in polyadenylated RNA during pollen maturation and germination. Biol Plant 24:331–340
Tupý J, Říhová L, Žárský V (1991) Production of fertile tobacco pollen from microspores in suspension culture and its storage for in situ pollination. Sex Plant Reprod 4:284–287
Twell D (2011) Male gametogenesis and germline specification in flowering plants. Sex Plant Reprod 24:149–160. doi:10.1007/s00497-010-0157-5
Twell D, Park SK, Hawkins TJ, Schubert D, Schmidt R, Smertenko A, Hussey PJ (2002) MOR1/GEM1 has an essential role in the plant-specific cytokinetic phragmoplast. Nat Cell Biol 4:711–714. doi:10.1038/ncb844
Uebler S, Dresselhaus T (2014) Identifying plant cell-surface receptors: combining ‘classical’ techniques with novel methods. Biochem Soc Trans 42:395–400. doi:10.1042/bst20130251
Ueda T, Iwashita H, Hashimoto Y, Imoto T (1996) Stabilization of lysozyme by introducing N-glycosylation signal sequence. J Biochem 119:157–161
van Bentem SD, Anrather D, Dohnal I, Roitinger E, Csaszar E, Joore J, Buijnink J, Carreri A, Forzani C, Lorkovic ZJ, Barta A, Lecourieux D, Verhounig A, Jonak C, Hirt H (2008) Site-specific phosphorylation profiling of Arabidopsis proteins by mass spectrometry and peptide chip analysis. J Proteome Res 7:2458–2470. doi:10.1021/pr8000173
Vidali L, McKenna ST, Hepler PK (2001) Actin polymerization is essential for pollen tube growth. Mol Biol Cell 12:2534–2545
Vogler F, Schmalzl C, Englhart M, Bircheneder M, Sprunck S (2014) Brassinosteroids promote Arabidopsis pollen germination and growth. Plant Reprod 27:153–167. doi:10.1007/s00497-014-0247-x
Vogler F, Konrad SSA, Sprunck S (2015) Knockin’ on pollen’s door: live cell imaging of early polarization events in germinating Arabidopsis pollen. Front Plant Sci. doi:10.3389/fpls.2015.00246
Völz R, Heydlauff J, Ripper D, von Lyncker L, Groß-Hardt R (2013) Ethylene signaling is required for synergid degeneration and the establishment of a pollen tube block. Dev Cell 25:310–316. doi:10.1016/j.devcel.2013.04.001
von Besser K, Frank AC, Johnson MA, Preuss D (2006) Arabidopsis HAP2 (GCS1) is a sperm-specific gene required for pollen tube guidance and fertilization. Development 133:4761–4769. doi:10.1242/dev.02683
Wang Y, Zhang W-Z, Song L-F, Zou J-J, Su Z, Wu W-H (2008) Transcriptome analyses show changes in gene expression to accompany pollen germination and tube growth in Arabidopsis. Plant Physiol 148:1201–1211. doi:10.1104/pp.108.126375
Wei LQ, Xu WY, Deng ZY, Su Z, Xue Y, Wang T (2010) Genome-scale analysis and comparison of gene expression profiles in developing and germinated pollen in Oryza sativa. BMC Genomics 11:338. doi:10.1186/1471-2164-11-338
Williams JH (2009) Amborella trichopoda (Amborellaceae) and the evolutionary developmental origins of the angiosperm progamic phase. Am J Bot 96:144–165. doi:10.3732/ajb.0800070
Williams JH, Taylor ML, O’Meara BC (2014) Repeated evolution of tricellular (and bicellular) pollen. Am J Bot 101:559–571. doi:10.3732/ajb.1300423
Wilsen KL, Lovy-Wheeler A, Voigt B, Menzel D, Kunkel JG, Hepler PK (2006) Imaging the actin cytoskeleton in growing pollen tubes. Sex Plant Reprod 19:51–62. doi:10.1007/s00497-006-0021-9
Wilson C, Voronin V, Touraev A, Vicente O, Heberle Bors E (1997) A developmentally regulated MAP kinase activated by hydration in tobacco pollen. Plant Cell 9:2093–2100
Wolschin F, Weckwerth W (2005) Combining metal oxide affinity chromatography (MOAC) and selective mass spectrometry for robust identification of in vivo protein phosphorylation sites. Plant Methods 1(1):9
Woollard AAD, Moore I (2008) The functions of Rab GTPases in plant membrane traffic. Curr Opin Plant Biol 11:610–619. doi:10.1016/j.pbi.2008.09.010
Worrall D, Hird DL, Hodge R, Paul W, Draper J, Scott R (1992) Premature dissolution of the microsporocyte callose wall causes male sterility in transgenic tobacco. Plant Cell 4:759–771. doi:10.1105/tpc.4.7.759
Wu H, de Graaf BHJ, Mariani C, Cheung AY (2001) Hydroxyproline-rich glycoproteins in plant reproductive tissues: structure, functions and regulation. Cell Mol Life Sci 58:1418–1429
Yadegari R, Drews GN (2004) Female gametophyte development. Plant Cell 16:S133–S141. doi:10.1105/tpc.018192
Yu GH, Zou J, Feng J, Peng XB, Wu JY, Wu YL, Palanivelu R, Sun MX (2014) Exogenous γ-aminobutyric acid (GABA) affects pollen tube growth via modulating putative Ca2+-permeable membrane channels and is coupled to negative regulation on glutamate decarboxylase. J Exp Bot 65:3235–3248. doi:10.1093/jxb/eru171
Zaki MAM, Dickinson H (1991) Microspore-derived embryos in Brassica: the significance of division asymmetry in pollen mitosis I to embryogenic development. Sex Plant Reprod 4:48–55
Zhang Y, McCormick S (2010) The regulation of vesicle trafficking by small GTPases and phospholipids during pollen tube growth. Sex Plant Reprod 23:87–93. doi:10.1007/s00497-009-0118-z
Zhang ZB, Zhu J, Gao JF, Wang C, Li H, Li H, Zhang HQ, Zhang S, Wang DM, Wang QX, Huang H, Xia HJ, Yang ZN (2007) Transcription factor AtMYB103 is required for anther development by regulating tapetum development, callose dissolution and exine formation in Arabidopsis. Plant J 52:528–538. doi:10.1111/j.1365-313X.2007.03254.x
Zhang Y, He J, McCormick S (2009) Two Arabidopsis AGC kinases are critical for the polarized growth of pollen tubes. Plant J 58:474–484. doi:10.1111/j.1365-313X.2009.03792.x
Zhao HP, Ren HY (2006) Rop1Ps promote actin cytoskeleton dynamics and control the tip growth of lily pollen tube. Sex Plant Reprod 19:83–91. doi:10.1007/s00497-006-0024-6
Zhao XY, Wang Q, Li S, Ge FR, Zhou LZ, McCormick S, Zhang Y (2013) The juxtamembrane and carboxy-terminal domains of Arabidopsis PRK2 are critical for ROP-induced growth in pollen tubes. J Exp Bot 64(18):5599–5610. doi:10.1093/jxb/ert323
Zheng ZL, Yang ZB (2000) The Rop GTPase: an emerging signaling switch in plants. Plant Mol Biol 44:1–9. doi:10.1023/a:1006402628948
Zhu L, Zhang Y, Kang E, Xu Q, Wang M, Rui Y, Liu B, Yuan M, Fu Y (2013) MAP18 regulates the direction of pollen tube growth in Arabidopsis by modulating F-actin organization. Plant Cell 25:851–867. doi:10.1105/tpc.113.110528
Acknowledgments
The authors thank Barbora Honysová for drawing the Figs. 1 and 2 and Nina Lindstrøm Friggens for assistance with drawing Fig. 3 and the language editing of the manuscript. The authors acknowledge the financial support from the Czech Science Foundation Grants No. 15-22720S, 14-32292S, P305/12/2611 and 15-16050S and Ministry of Education, Youth and Sport CR project COST LD14109.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by Enrico Schleiff.
A contribution to the special issue ‘Pollen development and stress response’.
Electronic supplementary material
Below is the link to the electronic supplementary material.
497_2015_272_MOESM1_ESM.tif
Transcriptome profile of Arabidopsis cysteine-rich receptor-like protein kinases. The five CRK genes with enriched expression in pollen and in sperm cells are highlighted. (TIFF 173 kb)
497_2015_272_MOESM2_ESM.xlsx
The list of Arabidopsis protein kinases identified in the transcriptomic (Honys and Twell 2003), proteomic (Grobei et al. 2009) and phosphoproteomic (Mayank et al. 2012) datasets of mature pollen. (XLSX 1477 kb)
497_2015_272_MOESM3_ESM.xlsx
Arabidopsis cysteine-rich polypeptides of < 150 aa’s and predicted as secreted proteins outsourced from UniProt protein repository and analysed by SignalP (Petersen et al. 2011) and SecretomeP (Bendtsen et al. 2004). Dataset 2 is a list of Arabidopsis cysteine-rich receptor-like protein kinases with predicted single pass membrane helices and membrane localization. (XLSX 66 kb)
497_2015_272_MOESM4_ESM.xlsx
A list of Arabidopsis GPI-anchored proteins with predicted secretion by signalP and secretomeP as well as their putative C-terminal GPI-anchor site based on Big-Pi (Eisenhaber et al. 2003) and PredGPI prediction algorithms (Pierlioni et al. 2008). (XLSX 49 kb)
Rights and permissions
About this article
Cite this article
Hafidh, S., Fíla, J. & Honys, D. Male gametophyte development and function in angiosperms: a general concept. Plant Reprod 29, 31–51 (2016). https://doi.org/10.1007/s00497-015-0272-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00497-015-0272-4