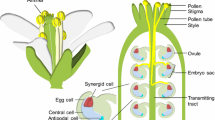
Abstract
The synergid cells are located in the female gametophyte and are essential for angiosperm reproduction. During the fertilization process, a pollen tube grows into one of the synergid cells, ceases growth, ruptures, and releases its two sperm cells into this cell. The synergid cells produce an attractant that guides the pollen tube to the female gametophyte and likely contain factors that control arrest of pollen tube growth, pollen tube discharge, and gamete fusion. The synergid cells contain an elaborated cell wall at their micropylar poles, the filiform apparatus that likely plays a role in pollen tube guidance and pollen tube reception. Recent genetic, molecular, and physiological studies in Arabidopsis, maize, and Torenia have provided insights into synergid cell development and the control of pollen tube growth by the synergid cell.


Similar content being viewed by others
References
Chaubal R, Reger BJ (1990) Relatively high calcium is localized in synergid cells of wheat ovaries. Sex Plant Reprod 3:98–102
Chaubal R, Reger BJ (1992) Calcium in the synergid cells and other regions of pearl millet ovaries. Sex Plant Reprod 5:34–46
Chaubal R, Reger BJ (1993) Prepollination degeneration in mature synergids of pearl millet: an examination using antimonite fixation to localize calcium. Sex Plant Reprod 6:225–238
Choi Y, Gehring M, Johnson L, Hannon M, Harada JJ, Goldberg RB, Jacobsen SE, Fischer RL (2002) DEMETER, a DNA glycosylase domain protein, is required for endosperm gene imprinting and seed viability in Arabidopsis. Cell 110:33–42
Christensen CA, Gorsich SW, Brown RH, Jones LG, Brown J, Shaw JM, Drews GN (2002) Mitochondrial GFA2 is required for synergid cell death in Arabidopsis. Plant Cell 14:2215–2232
Christensen CA, King EJ, Jordan JR, Drews GN (1997) Megagametogenesis in Arabidopsis wild type and the Gf mutant. Sex Plant Reprod 10:49–64
Cordts S, Bantin J, Wittich PE, Kranz E, Lorz H, Dresselhaus T (2001) ZmES genes encode peptides with structural homology to defensins and are specifically expressed in the female gametophyte of maize. Plant J 25:103–114
Davidson EH, Cameron RA, Ransick A (1998) Specification of cell fate in the sea urchin embryo: summary and some proposed mechanisms. Development 125:3269–3290
Diboll AG, Larson DA (1966) An electron microscopic study of the mature megagametophyte in Zea mays. Am J Bot 53:391–402
Dresselhaus T, Amien S, Marton M, Strecke A, Brettschneider R, Cordts S (2005) TRANSPARENT LEAF AREA1 encodes a secreted proteolipid required for anther maturation, morphogenesis, and differentiation during leaf development in maize. Plant Cell 17:730–745
Drews GN, Lee D, Christensen CA (1998) Genetic analysis of female gametophyte development and function. Plant Cell 10:5–17
Drews GN, Yadegari R (2002) Development and function of the angiosperm female gametophyte. Annu Rev Genet 36:99–124
Ephrussi A, St Johnston D (2004) Seeing is believing: the bicoid morphogen gradient matures. Cell 116:143–152
Escobar-Restrepo JM, Huck N, Kessler S, Gagliardini V, Gheyselinck J, Yang WC, Grossniklaus U (2007) The FERONIA receptor-like kinase mediates male-female interactions during pollen tube reception. Science 317:656–660
Faure J-E, Digonnet C, Dumas C (1994) An in vitro system for adhesion and fusion of maize gametes. Science 263:1598–1600
Fisher DB, Jensen WA (1969) Cotton embryogenesis: the identification, as nuclei, of the X-bodies in the degenerated synergid. Planta 84:122–133
Folsom MW, Cass DD (1990) Embryo sac development in soybean: cellularization and egg apparatus expansion. Can J Bot 68:2135–2147
Fu Y, Yuan M, Huang B-Q, Yang H-Y, Zee S-Y, O’Brien TP (2000) Changes in actin organization in the living egg apparatus of Torenia fournieri during fertilization. Sex Plant Reprod 12:315–322
Gross-Hardt R, Kagi C, Baumann N, Moore JM, Baskar R, Gagliano WB, Jurgens G, Grossniklaus U (2007) LACHESIS restricts gametic cell fate in the female gametophyte of Arabidopsis. PLoS Biol 5:e47
Guilford VB, Fisk EL (1952) Megasporogenesis and seed development in Mimulus tigrinus and Torenia fournieri. Bull Torrey Bot Club 79:6–24
Higashiyama T (2002) The synergid cell: attractor and acceptor of the pollen tube for double fertilization. J Plant Res 115:149–160
Higashiyama T, Kuroiwa H, Kawano S, Kuroiwa T (1998) Guidance in vitro of the pollen tube to the naked embryo sac of Torenia fournieri. Plant Cell 10:2019–2032
Higashiyama T, Kuroiwa H, Kawano S, Kuroiwa T (2000) Explosive discharge of pollen tube contents in Torenia fournieri. Plant Physiol 122:11–14
Higashiyama T, Kuroiwa H, Kuroiwa T (2003) Pollen-tube guidance: beacons from the female gametophyte. Curr Opin Plant Biol 6:36–41
Higashiyama T, Yabe S, Sasaki N, Nishimura Y, Miyagishima S, Kuroiwa H, Kuroiwa T (2001) Pollen tube attraction by the synergid cell. Science 293:1480–1483
Huang B-Q, Russell SD (1992) Female germ unit: organization, isolation, and function. Int Rev Cytol 140:233–292
Huang BQ, Sheridan WF (1994) Female gametophyte development in maize: microtubular organization and embryo sac polarity. Plant Cell 6:845–861
Huck N, Moore JM, Federer M, Grossniklaus U (2003) The Arabidopsis mutant feronia disrupts the female gametophytic control of pollen tube reception. Development 130:2149–2159
Hulskamp M, Schneitz K, Pruitt RE (1995) Genetic evidence for a long-range activity that directs pollen tube guidance in Arabidopsis. Plant Cell 7:57–64
Jiang X, Wang X (2004) Cytochrome C-mediated apoptosis. Annu Rev Biochem 73:87–106
Kasahara RD, Portereiko MF, Sandaklie-Nikolova L, Rabiger DS, Drews GN (2005) MYB98 is required for pollen tube guidance and synergid cell differentiation in Arabidopsis. Plant Cell 17:2981–2992
Le Q, Gutierrez-Marcos JF, Costa LM, Meyer S, Dickinson HG, Lorz H, Kranz E, Scholten S (2005) Construction and screening of subtracted cDNA libraries from limited populations of plant cells: a comparative analysis of gene expression between maize egg cells and central cells. Plant J 44:167–178
Lord EM, Russell SD (2002) The mechanisms of pollination and fertilization in plants. Annu Rev Cell Dev Biol 18:81–105
Maheshwari P (1950) An introduction to the embryology of angiosperms. McGraw-Hill, New York
Mansfield SG, Briarty LG, Erni S (1991) Early embryogenesis in Arabidopsis thaliana. I. The mature embryo sac. Can J Bot 69:447–460
Marton ML, Cordts S, Broadhvest J, Dresselhaus T (2005) Micropylar pollen tube guidance by Egg Apparatus 1 of maize. Science 307:573–576
Misra RC (1962) Contribution to the embryology of Arabidopsis thaliana. Agra Univ J Res 11:191–196
Murgia M, Huang B-Q, Tucker SC, Musgrave ME (1993) Embryo sac lacking antipodal cells in Arabidopsis thaliana (Brassicaceae). Am J Bot 80:824–838
Newcomb W (1973) The development of the embryo sac of sunflower Helianthus annuus after fertilization. Can J Bot 51:879–890
Punwani JA, Rabiger DS, Drews GN (2007) MYB98 positively regulates a battery of synergid-expressed genes encoding filiform apparatus localized proteins. Plant Cell 19:2557–2568
Ray S, Park S-S, Ray A (1997) Pollen tube guidance by the female gametophyte. Development 124:2489–2498
Rotman N, Rozier F, Boavida L, Dumas C, Berger F, Faure JE (2003) Female control of male gamete delivery during fertilization in Arabidopsis thaliana. Curr Biol 13:432–436
Russell SD (1979) Fine structure of megagametophyte development in Zea mays. Can J Bot 57:1093–1110
Russell SD (1992) Double fertilization. Int Rev Cytol 140:357–388
Russell SD (1993) The egg cell: development and role in fertilization and early embryogenesis. Plant Cell 5:1349–1359
Russell SD (1996) Attraction and transport of male gametes for fertilization. Sex Plant Reprod 9:337–342
Sandaklie-Nikolova L, Palanivelu R, King EJ, Copenhaver GP, Drews GN (2007) Synergid cell death in Arabidopsis is triggered following direct interaction with the pollen tube. Plant Physiol 144:1753–1762
Schiott M, Romanowsky SM, Baekgaard L, Jakobsen MK, Palmgren MG, Harper JF (2004) A plant plasma membrane Ca2+ pump is required for normal pollen tube growth and fertilization. Proc Natl Acad Sci USA 101:9502–9507
Shimizu KK, Okada K (2000) Attractive and repulsive interactions between female and male gametophytes in Arabidopsis pollen tube guidance. Development 127:4511–4518
Silverstein KA, Moskal WA Jr, Wu HC, Underwood BA, Graham MA, Town CD, VandenBosch KA (2007) Small cysteine-rich peptides resembling antimicrobial peptides have been under-predicted in plants. Plant J 51:262–280
Steffen JG, Kang I-H, Macfarlane J, Drews GN (2007) Identification of genes expressed in the Arabidopsis female gametophyte. Plant J 51:281–292
van der Pluijm JE (1964) An electron microscopic investigation of the filiform apparatus in the embryo sac of Torenia fournieri. In: Linskens HF (ed) Pollen physiology and fertilization. North-Holland Publ, Amsterdam, pp 8–16
van Went JL, Willemse MTM (1984) Fertilization. In: Johri B (ed) Embryology of angiosperms. Springer, Berlin, pp 273–318
Webb MC, Gunning BES (1994) Embryo sac development in Arabidopsis. II. The cytoskeleton during megagametogenesis. Sex Plant Reprod 7:153–163
Weterings K, Russell SD (2004) Experimental analysis of the fertilization process. Plant Cell 16(Suppl):S107–S118
Willemse MTM, van Went JL (1984) The female gametophyte. In: Johri BM (ed) Embryology of angiosperms. Springer, Berlin, pp 159–196
Williams EG, Knox RB, Rouse JL (1982) Pollination sub-systems distinguished by pollen tube arrest after incompatible interspecific crosses in Rhododendron (Ericaceae). J Cell Sci 53:255–277
Williams EG, Kaul V, Rouse JL, Palser BF (1986) Overgrowth of pollen tubes in embryo sacs of Rhododendron following interspecific pollinations. Aust J Bot 34:413–423
Yadegari R, Drews GN (2004) Female gametophyte development. Plant Cell 16(Suppl):S133–S141
Yan H, Yang H-Y, Jensen W (1991) Ultrastructure of the developing embryo sac of sunflower (Helianthus annuus) before and after fertilization. Can J Bot 69:191–202
Yang H, Kaur N, Kiriakopolos S, McCormick S (2006) EST generation and analyses towards identifying female gametophyte-specific genes in Zea mays L. Planta 224:1004–1014
Yu HJ, Hogan P, Sundaresan V (2005) Analysis of the female gametophyte transcriptome of Arabidopsis by comparative expression profiling. Plant Physiol 139:1853–1869
Acknowledgments
We thank Ramin Yadegari, Jaimie Van Norman, and members of the Drews lab for critical review of this manuscript. Our work on the synergid cell is supported by a National Science Foundation grant (grant no. IOB-0542953) to G.N.D. and a National Institutes of Health Developmental Biology Training Grant (grant no. 5T32 HD07491) appointment to J.A.P.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by Thomas Dresselhaus.
Rights and permissions
About this article
Cite this article
Punwani, J.A., Drews, G.N. Development and function of the synergid cell. Sex Plant Reprod 21, 7–15 (2008). https://doi.org/10.1007/s00497-007-0059-3
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00497-007-0059-3